Global Bioinks Market Size, Share Analysis Report By Material (Natural Polymer-based, Synthetic Polymer-based, and Hybrid Systems), By Application (Tissue Engineering, Drug Discovery And Toxicology Testing, Organoids And Organ-on-Chip, Regenerative Medicine, and Cosmetic And Non-Animal Testing), By Region and Companies - Industry Segment Outlook, Market Assessment, Competition Scenario, Trends and Forecast 2026-2035
- Published date: May 2026
- Report ID: 185661
- Number of Pages: 336
- Format:
-
keyboard_arrow_up
Quick Navigation
Report Overview
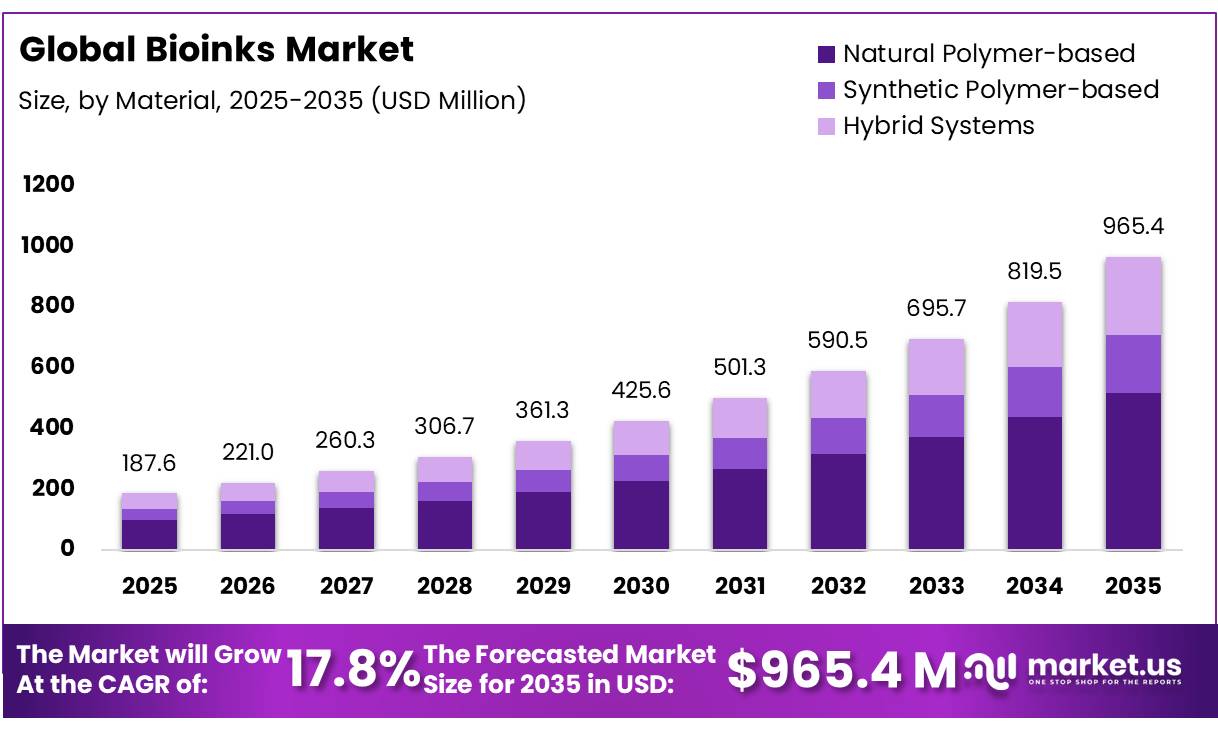
The Global Bioinks Market size is expected to be worth around USD 965.4 Million by 2035, from USD 187.6 Million in 2025, growing at a CAGR of 17.8% during the forecast period from 2026 to 2035. In 2025, Asia-Pacific held a dominant market position, capturing more than a 41.9% share, holding USD 3.9 Billion revenue.
Bioinks are specialized biomaterial formulations designed to enable 3D bioprinting of cell-laden structures that replicate aspects of native tissue architecture. These materials, typically derived from natural polymers, synthetic polymers, or hybrid combinations, provide both structural support and a biologically active environment for cell growth and differentiation.
Their use has expanded alongside advances in additive manufacturing and microphysiological systems, enabling more complex fabrication of tissue constructs and in vitro biological models. Tissue engineering remains the primary application area, where bioinks are used to create functional scaffolds for skin, cartilage, bone, and vascular tissues. Growing utilization is also observed in drug screening, toxicology testing, organoid development, and organ-on-chip platforms, reflecting increasing reliance on physiologically relevant experimental systems.
Natural polymer-based formulations dominate due to their intrinsic biocompatibility and similarity to extracellular matrix components. Progress in crosslinking techniques and hybrid material development has improved mechanical performance and print fidelity. Adoption is further supported by integration with regenerative medicine workflows and increasing demand for personalized biological constructs. Continuous material innovation and expansion of application scope continue to define the evolution of this advanced biomaterials segment.
Key Takeaways
- The global bioinks market was valued at US$187.6 million in 2025.
- The global bioinks market is projected to grow at a CAGR of 17.8% and is estimated to reach US$965.4 million by 2035.
- Based on the material, natural polymer-based bioinks dominated the market, with a market share of around 53.6%.
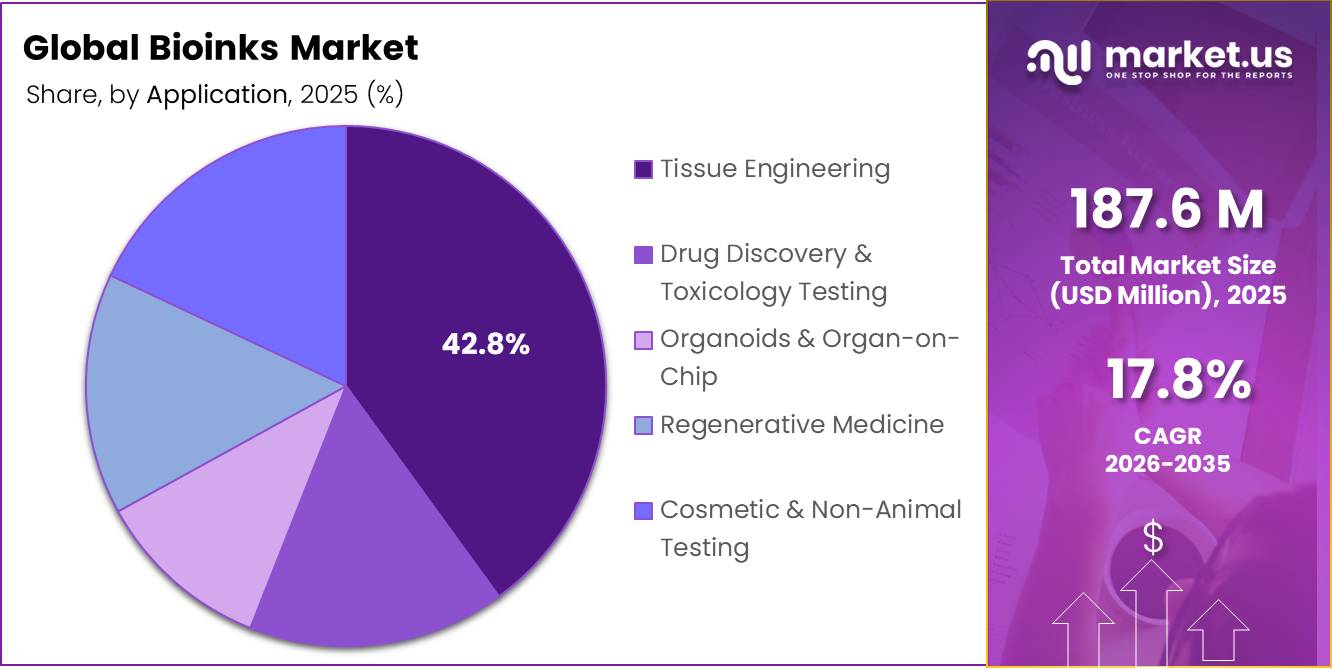
- Among the applications of the bioinks, tissue engineering held a major share in the market, 42.8% of the market share.
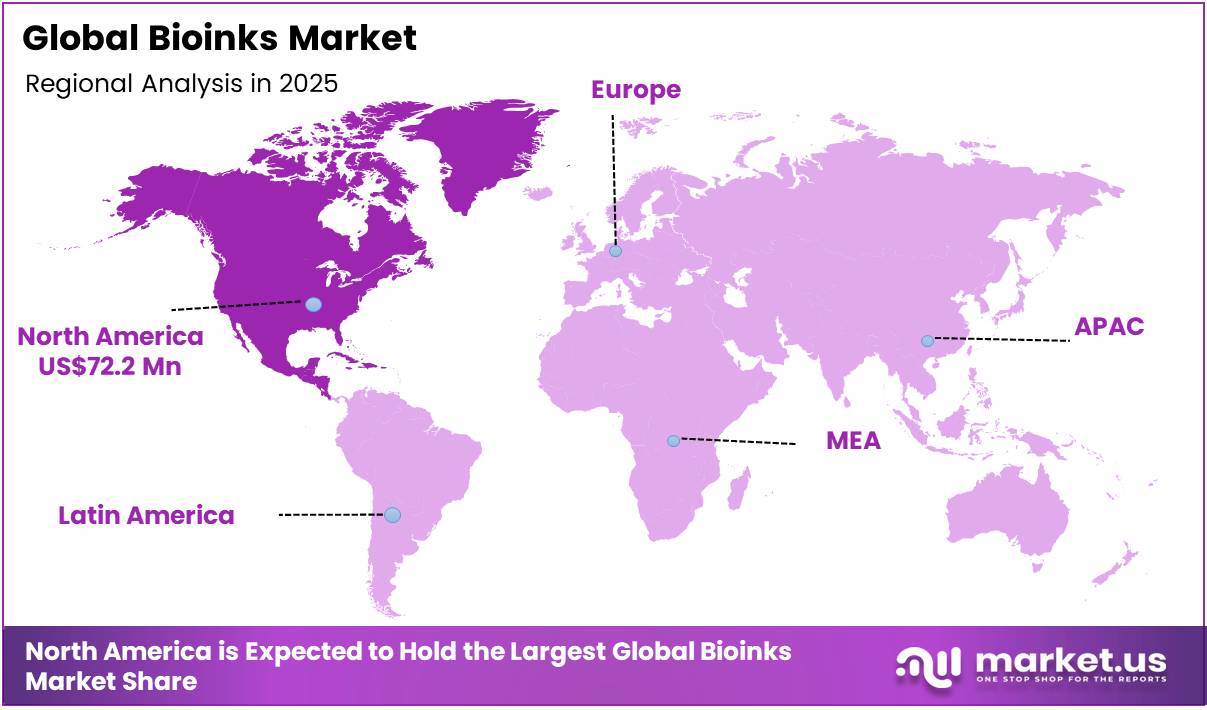
- In 2025, North America was the most dominant region in the bioinks market, accounting for around 38.5% of the total global consumption.
Material Analysis
Natural Polymer-based Bioinks Held the Largest Share in the Market.
The Natural Polymer-based segment represents the dominant share of the bioinks market, accounting for 53.60% of total material usage due to its strong biological affinity and compatibility with cellular environments. These bioinks, derived from biomacromolecules such as collagen, gelatin, alginate, fibrin, and hyaluronic acid, closely mimic the native extracellular matrix, supporting enhanced cell adhesion, proliferation, and differentiation. Their intrinsic bioactivity makes them particularly suitable for applications in tissue engineering, regenerative medicine, and organoid development, where physiological relevance is critical.
Additionally, natural polymers exhibit favorable hydrogel-forming properties under mild conditions, enabling high cell viability during extrusion and inkjet-based bioprinting processes. Despite challenges related to mechanical strength and batch variability, continuous improvements in crosslinking strategies and composite blending are strengthening their functional performance. The widespread use of naturally derived materials in research-driven bioprinting workflows continues to reinforce their leading position within the material segmentation landscape.
Application Analysis
Bioinks Are Mostly Utilized for Tissue Engineering.
The Tissue Engineering segment holds the dominant share of 42.80% within the bioinks application landscape, driven by its extensive use in developing functional biological constructs for repairing or replacing damaged tissues. Bioinks are widely utilized to fabricate scaffolds that support cell adhesion, proliferation, and differentiation, enabling the formation of structured tissue analogs such as skin, cartilage, bone, and vascular networks.
The segment benefits from increasing integration of 3D bioprinting technologies that allow precise spatial control over cell distribution and extracellular matrix composition. This has enhanced the ability to replicate native tissue architecture with improved physiological relevance. Research institutions and biomedical developers actively employ bioink-based approaches to address limitations of conventional grafting techniques, particularly in donor tissue scarcity and immune compatibility. Continuous advancements in hydrogel formulations and cell-laden constructs are further strengthening functional outcomes, reinforcing the segment’s leading position across regenerative and biofabrication-focused applications.
Key Market Segments
By Material
- Natural Polymer-based
- Synthetic Polymer-based
- Hybrid Systems
By Application
- Tissue Engineering
- Drug Discovery & Toxicology Testing
- Organoids & Organ-on-Chip
- Regenerative Medicine
- Cosmetic & Non-Animal Testing
Drivers
Rising Demand for Physiologically Relevant Tissue Models in Life Sciences Research.
The increasing reliance on physiologically relevant tissue models in life sciences research is closely tied to efforts to improve the predictive accuracy of preclinical experimentation. Conventional two-dimensional cell cultures and animal models often fail to replicate human tissue architecture, dynamic biochemical gradients, and multicellular interactions, leading to translational gaps in drug response and toxicity evaluation. Microengineered three-dimensional systems, including organoids and organ-on-chip platforms, address this limitation by enabling controlled replication of organ-level microenvironments, such as perfusion, extracellular matrix interactions, and mechanical stimulation, within bioengineered scaffolds.
Institutional studies, including those supported by the National Institutes of Health (NIH), have documented that microphysiological systems can reproduce key functional characteristics of organs such as the liver, lung, and kidney, enabling more representative assessment of pharmacokinetics and disease mechanisms compared to static cultures. The U.S. Food and Drug Administration (FDA) has also acknowledged the utility of advanced in vitro models in reducing dependence on animal testing frameworks, particularly for toxicity screening and early-stage drug evaluation. These systems are increasingly used to study human-relevant biological responses, including hepatotoxicity and inflammatory pathways that are difficult to capture in animal models.
Quantitative experimental studies demonstrate that perfused 3D bioprinted liver constructs can maintain stable metabolic activity over multi-week culture periods while expressing over 100 genes associated with drug metabolism and excretion pathways, reflecting improved functional fidelity relative to 2D systems. Integration of such models into drug screening workflows has been associated with improved detection of human-specific metabolites and compound-specific toxicity signals that may not appear in conventional assays.
Broader adoption is reinforced by microfluidic organ-on-chip systems that replicate physiological shear stress and dynamic fluid flow conditions, enabling simulation of organ–organ interactions and systemic drug distribution behaviors in controlled environments. These capabilities are increasingly utilized in oncology, toxicology, and regenerative medicine studies, where precision in modeling cellular microenvironments directly influences experimental validity and downstream translational reliability.
Restraints
Limited Standardization of Bioink Formulations Restricts Reproducibility and Scalability.
The absence of widely harmonized formulation standards for bioinks continues to affect reproducibility across 3D bioprinting and tissue engineering workflows. Bioinks typically combine natural polymers, synthetic hydrogels, or hybrid extracellular matrix (ECM)-mimicking systems, and even minor variations in polymer concentration, crosslinking density, or source material can significantly alter rheological behavior, print fidelity, and cell response. Reports highlight that natural ECM-derived materials, such as decellularized matrices, often exhibit batch-to-batch variability due to differences in tissue sourcing and processing conditions, which directly influence structural and biochemical consistency in printed constructs.
Experimental evidence shows that bioinks must balance mechanical printability with biological functionality, yet formulations optimized for structural stability may compromise cell viability and tissue maturation, while highly bioactive matrices may lack consistent mechanical integrity during fabrication. This trade-off is further amplified in extrusion-based printing systems, where variations in viscosity, shear-thinning behavior, and nozzle pressure conditions can lead to inconsistencies in filament formation and cell distribution across identical printing runs.
In organoid and organ-on-chip applications, such variability propagates into differences in organoid morphology, size distribution, and functional readouts, even under closely controlled culture environments. Studies note that minor fluctuations in matrix composition and spatial organization can significantly alter developmental outcomes, underscoring the sensitivity of 3D biological systems to material inconsistency. These compounded material and process variations collectively limit experimental reproducibility and complicate the establishment of uniform quality control benchmarks for scalable biofabrication systems.
Opportunity
Advancement of Patient-Specific and Regenerative Tissue Engineering Applications Creates Opportunities in the Magnesium Batteries Market.
The increasing orientation toward patient-specific and regenerative tissue engineering applications is strongly associated with the development of bioink-enabled fabrication of living, functional tissue constructs derived from autologous or induced pluripotent stem cell sources. Patient-derived organoids, which can replicate organ-specific cellular architecture and function in vitro, have been demonstrated to retain donor-specific genetic and phenotypic characteristics, enabling individualized disease modeling and therapeutic screening under controlled laboratory conditions. NIH-supported biomedical studies indicate that organoid systems derived from human stem cells can reproduce key structural and functional attributes of liver, intestinal, and neural tissues, supporting their use in precision biological assessment frameworks.
sBioink formulations optimized for such constructs are increasingly used in cartilage, skin, and vascular tissue engineering, where printed scaffolds can incorporate living cells and biomimetic extracellular matrices to support tissue maturation. For instance, collagen-based and decellularized ECM-based bioinks have been applied in preclinical models of skin and cartilage repair, demonstrating improved integration potential due to tissue-specific biochemical signaling.
Experimental 3D bioprinting systems have also shown the ability to fabricate vascularized tissue constructs, a critical requirement for maintaining nutrient diffusion and long-term viability in engineered tissues. This capability is particularly relevant in reconstructive applications where structural and functional restoration must align with patient-specific anatomical parameters. Collectively, these developments are reinforcing the use of bioinks in customized regenerative strategies, particularly in areas requiring defect-specific tissue reconstruction and predictive preclinical human tissue modeling.
Trends
Integration of Bioinks with Organ-on-Chip and Microphysiological Systems.
The integration of bioinks with organ-on-chip and microphysiological systems is increasingly enabling the fabrication of structured, perfused 3D tissue environments that combine engineered biomaterials with controlled microfluidic conditions. Organ-on-chip platforms are defined by microchannels lined with living cells and continuously perfused to reproduce organ-level functions such as shear stress, nutrient transport, and biochemical gradients, while bioinks provide the printable hydrogel or composite matrices required to spatially organize these cells into tissue-like architectures within such chips.
Experimental systems have demonstrated that 3D bioprinting can directly deposit multiple cell-laden bioinks into microfluidic devices, forming vascular-like networks and heterogeneous tissue regions that improve physiological relevance compared to static cultures. This integration supports more stable long-term culture conditions, as perfusion within chip systems helps maintain oxygen and metabolite exchange across printed constructs, addressing diffusion limitations observed in bulk hydrogel cultures.
Quantitative evaluations reported in organ-on-chip research indicate that such systems can better replicate in vivo-like biochemical and physical conditions, including controlled shear stress and gradient formation at microscale levels, which are critical for tissue maturation and drug-response fidelity.
Practical implementations include liver-, lung-, and vessel-on-chip models where bioprinted hydrogel matrices are used to anchor hepatocytes, endothelial cells, or epithelial layers, enabling functional assays for toxicity screening and disease modeling under dynamic flow. This convergence is further supported by sensor-integrated platforms capable of real-time monitoring of cellular responses, strengthening the utility of bioink-enabled microphysiological systems in reproducible experimental workflows.
Geopolitical Impact Analysis
Geopolitical Fragmentation and Supply Chain Volatility Reshaping Bioink Material Sourcing and Development Dynamics.
Geopolitical tensions are increasingly influencing the bioinks ecosystem through disruptions in upstream chemical supply chains, cross-border regulatory controls, and trade restrictions affecting critical raw materials used in hydrogel and polymer formulations. Bioinks rely heavily on inputs such as polyethylene glycol, gelatin derivatives, alginate, and collagen, many of which are integrated into broader bioprocessing and chemical value chains that span multiple jurisdictions. In 2025, industry assessments of life sciences supply networks indicated that polymers used in single-use bioprocessing systems accounted for approximately 55% of material composition, making them sensitive to trade-linked feedstock volatility and tariff adjustments.
Export control measures on advanced biotechnology equipment and related analytical tools have also introduced indirect constraints on bioink development workflows, particularly for high-precision characterization of rheological and cellular performance properties. These restrictions are accompanied by broader policy actions, including tariff regimes affecting medical and laboratory imports, where duties in some cases have reached double-digit percentages and, in certain categories, significantly higher levels for specific trading partners.
Supply chain fragmentation has led to increased sourcing diversification and regionalization of reagent manufacturing, with documented cases of shipping disruptions, port constraints, and raw material shortages affecting laboratory consumables and polymer feedstocks. For bioink developers, this has translated into variability in precursor consistency and longer procurement cycles for ECM-derived and synthetic hydrogel components. As a result, formulation continuity and scale-up reproducibility are increasingly dependent on localized sourcing strategies and vertically integrated material supply arrangements within life sciences manufacturing networks.
Regional Analysis
North America Held the Largest Share of the Global Bioinks Market.
In 2025, North America dominated the global bioinks market, holding about 38.5% of the total global consumption.
North America maintains a leading position in the bioinks ecosystem, primarily driven by dense integration of advanced bioprinting research, regulated biomedical innovation pathways, and strong institutional funding for regenerative medicine. This dominance is reinforced by the sustained application of bioinks in tissue engineering, drug screening, and organ-on-chip platforms, particularly within the United States, where biomedical research institutions and clinical translation programs are highly active.
Public sector involvement plays a significant role in accelerating foundational research. The U.S. National Institutes of Health (NIH) has consistently supported projects focused on organoids, biomaterials, and engineered tissues, enabling structured advancement of bioink-based systems for disease modeling and regenerative applications. Regulatory engagement by the U.S. Food and Drug Administration (FDA) further shapes adoption pathways, particularly through evolving oversight frameworks for 3D-printed medical products and preclinical testing models.
The presence of established biotechnology firms alongside academic spinouts supports the continuous refinement of hydrogel, ECM-based, and cell-laden formulations. Combined with expanding use in organ-on-chip systems and personalized tissue constructs, these factors sustain the region’s structurally advanced and research-intensive bioinks ecosystem.
Key Regions and Countries
- North America
- The US
- Canada
- Europe
- Germany
- France
- The UK
- Spain
- Italy
- Russia & CIS
- Rest of Europe
- APAC
- China
- Japan
- South Korea
- India
- ASEAN
- Rest of APAC
- Latin America
- Brazil
- Mexico
- Rest of Latin America
- Middle East & Africa
- GCC
- South Africa
- Rest of MEA
Key Players Analysis
Manufacturers of bioinks typically focus on strengthening material innovation pipelines by developing formulations with improved printability, mechanical stability, and cell compatibility, often through hybridization of natural and synthetic polymers. A key strategic emphasis lies in expanding application-specific product portfolios, particularly for organoid culture, organ-on-chip systems, and regenerative tissue engineering, where tailored rheological and biological properties are required. Companies also invest in scalable production processes to reduce batch variability and improve reproducibility, which is critical for research and translational consistency.
Collaboration with academic laboratories, biotechnology firms, and clinical research groups supports co-development of specialized bioinks aligned with emerging experimental models. In parallel, manufacturers enhance competitive positioning through integration with bioprinting hardware ecosystems and by offering validated, ready-to-use formulations that reduce end-user optimization time. Regulatory alignment, quality control standardization, and documentation support for research reproducibility further strengthen adoption across biomedical and pharmaceutical research environments.
The Major Players in The Industry
- CELLINK
- Collplant
- Advanced BioMatrix
- Allevi, Inc.
- BIO INX
- Merck KGaA
- Humabiologics, Inc.
- INNOREGEN
- Viscofan DE GmbH
- Scire Science
- Other Key Players
Report Scope
Report Features Description Market Value (2025) US$187.6 Mn Forecast Revenue (2035) US$965.4 Mn CAGR (2025-2035) 17.8% Base Year for Estimation 2025 Historic Period 2021-2024 Forecast Period 2025-2035 Report Coverage Revenue Forecast, Market Dynamics, Competitive Landscape, Recent Developments Segments Covered By Material (Natural Polymer-based, Synthetic Polymer-based, and Hybrid Systems), By Application (Tissue Engineering, Drug Discovery & Toxicology Testing, Organoids & Organ-on-Chip, Regenerative Medicine, and Cosmetic & Non-Animal Testing) Regional Analysis North America – The US & Canada; Europe – Germany, France, The UK, Spain, Italy, Russia & CIS, Rest of Europe; APAC– China, Japan, South Korea, India, ASEAN & Rest of APAC; Latin America– Brazil, Mexico & Rest of Latin America; Middle East & Africa– GCC, South Africa, & Rest of MEA Competitive Landscape CELLINK, Collplant, Advanced BioMatrix, Allevi, Inc., BIO INX, Merck KGaA, Humabiologics, Inc., INNOREGEN, Viscofan DE GmbH, Scire Science, and Other Players. Customization Scope Customization for segments, region/country-level will be provided. Moreover, additional customization can be done based on the requirements. Purchase Options We have three licenses to opt for: Single User License, Multi-User License (Up to 5 Users), Corporate Use License (Unlimited Users and Printable PDF) 
- CELLINK
- Collplant
- Advanced BioMatrix
- Allevi, Inc.
- BIO INX
- Merck KGaA
- Humabiologics, Inc.
- INNOREGEN
- Viscofan DE GmbH
- Scire Science
- Other Key Players










