Global Endoscope Reprocessing Market By Product Type (Automatic Endoscope Reprocessors, Endoscope Tracking Systems, Consumables and Detergents, Others) By Application (Disinfection, Sterilization) By End User (Hospitals, Ambulatory Surgical Centers, Specialty Clinics, Others), Region and Companies – Industry Segment Outlook, Market Assessment, Competition Scenario, Trends and Forecast 2026-2035
- Published date: May 2026
- Report ID: 185779
- Number of Pages: 206
- Format:
-
keyboard_arrow_up
Quick Navigation
Report Overview
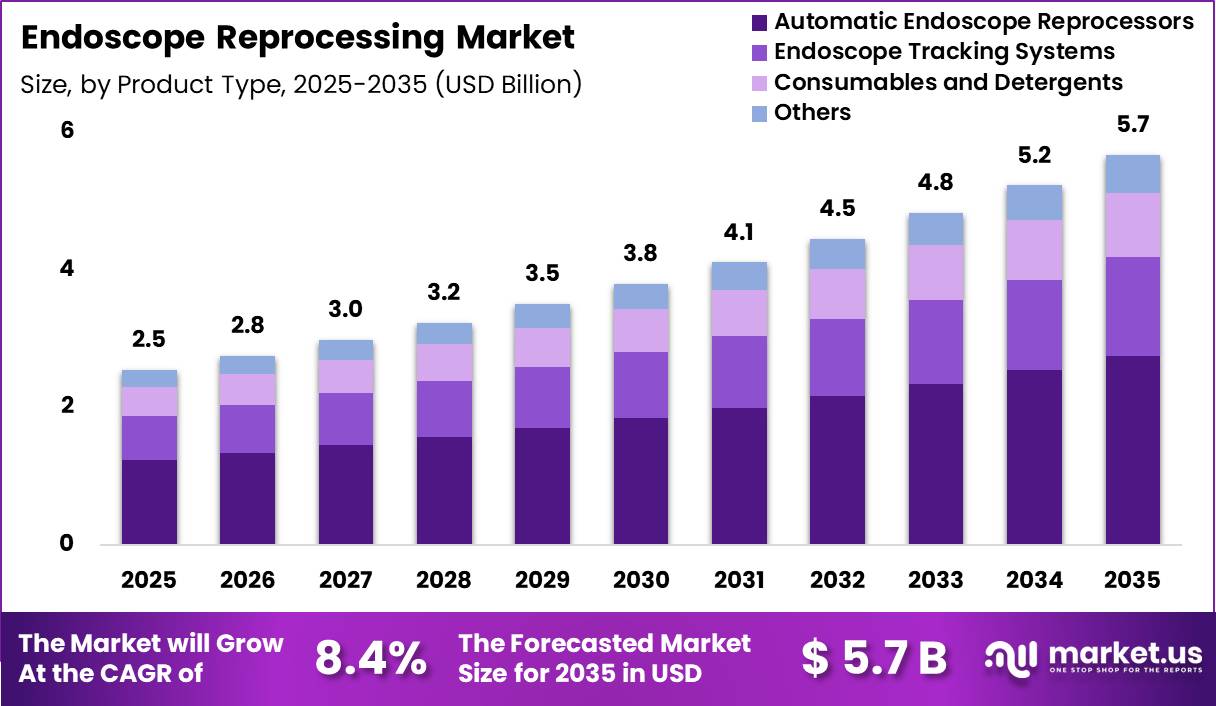
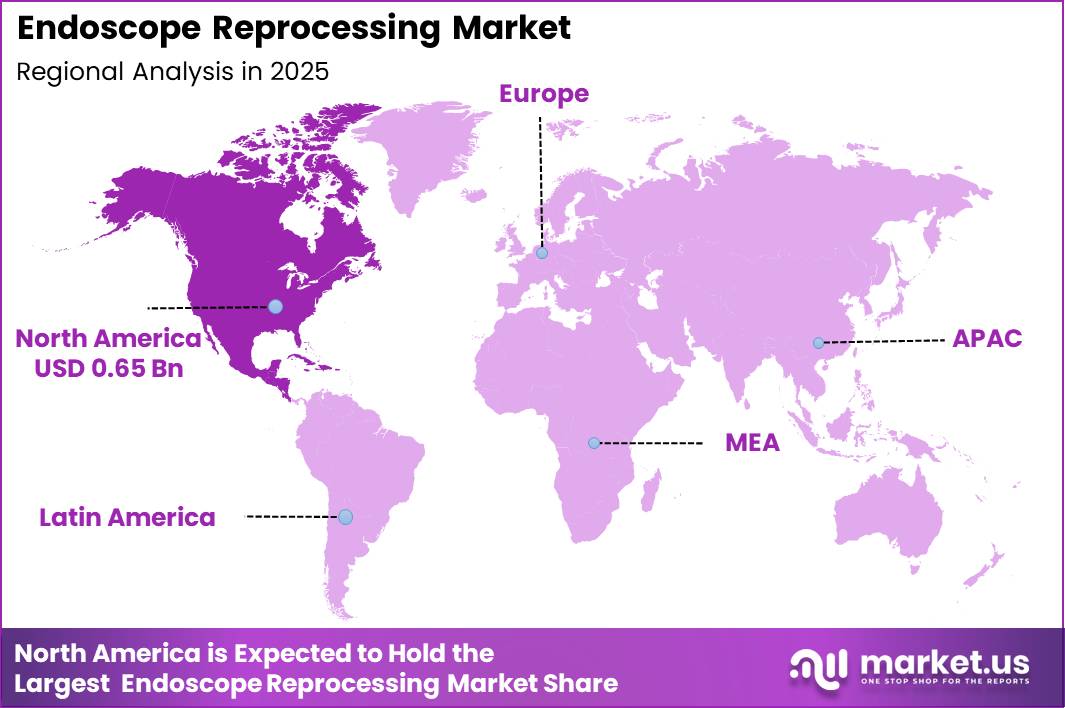
Global Endoscope Reprocessing Market size is expected to be worth around US$ 5.7 Billion by 2035 from US$ 2.5 Billion in 2025, growing at a CAGR of 8.4% during the forecast period from 2026 to 2035. In 2025, North America led the market, achieving over 38.2% share with a revenue of US$ 0.65 Billion.
The Endoscope reprocessing market is gaining strategic importance within the global healthcare industry due to the rising volume of minimally invasive diagnostic and therapeutic procedures and the increasing emphasis on infection prevention in hospitals and ambulatory surgical centers.
Endoscope reprocessing refers to the cleaning, high-level disinfection, sterilization, drying, and storage of reusable endoscopes to prevent cross-contamination between patients. According to the U.S. Food and Drug Administration (FDA), more than 500,000 Endoscopic Retrograde Cholangiopancreatography (ERCP) procedures using duodenoscopes are performed annually in the United States alone.
The market is being driven by growing concerns regarding healthcare-associated infections (HAIs) linked to improperly reprocessed endoscopes. The FDA has repeatedly highlighted that the complex design of duodenoscopes and reusable flexible endoscopes can hinder effective cleaning and disinfection, even when manufacturers’ instructions are followed correctly.
Interim FDA surveillance studies identified contamination rates of approximately 4% to 6% in older duodenoscope models after reprocessing, prompting healthcare authorities to encourage the adoption of advanced automated endoscope reprocessors, disposable components, and enhanced sterilization technologies.
In addition, the increasing prevalence of gastrointestinal disorders, colorectal cancer, pancreatic diseases, and respiratory conditions has significantly increased endoscopy procedure volumes worldwide. The World Health Organization (WHO) estimates that healthcare-associated infections affect hundreds of millions of patients annually across the globe, strengthening the need for stringent medical device reprocessing protocols in healthcare facilities.
Furthermore, regulatory agencies such as the FDA and the Centers for Disease Control and Prevention (CDC) are continuously issuing updated guidelines and validation requirements for reusable medical devices, supporting investments in automated cleaning systems, disinfectants, drying cabinets, and endoscope tracking solutions.
Technological advancements, including disposable endcaps, single-use endoscopes, and automated high-level disinfection systems capable of achieving a 99.9999% microbial reduction standard, are further supporting market expansion across developed and emerging healthcare markets.
Key Takeaways
- Market Size: Global Endoscope Reprocessing Market size is expected to be worth around US$ 5.7 Billion by 2035 from US$ 2.5 Billion in 2025.
- Market Share: The market growing at a CAGR of 8.4% during the forecast period from 2026 to 2035.
- Product Type Analysis: Automatic Endoscope Reprocessors dominated the market and accounted for 48.5% of the global market share in 2025.
- Application Analysis: In 2025, the Disinfection segment held the dominant market position and accounted for 56.2% of the total market share.
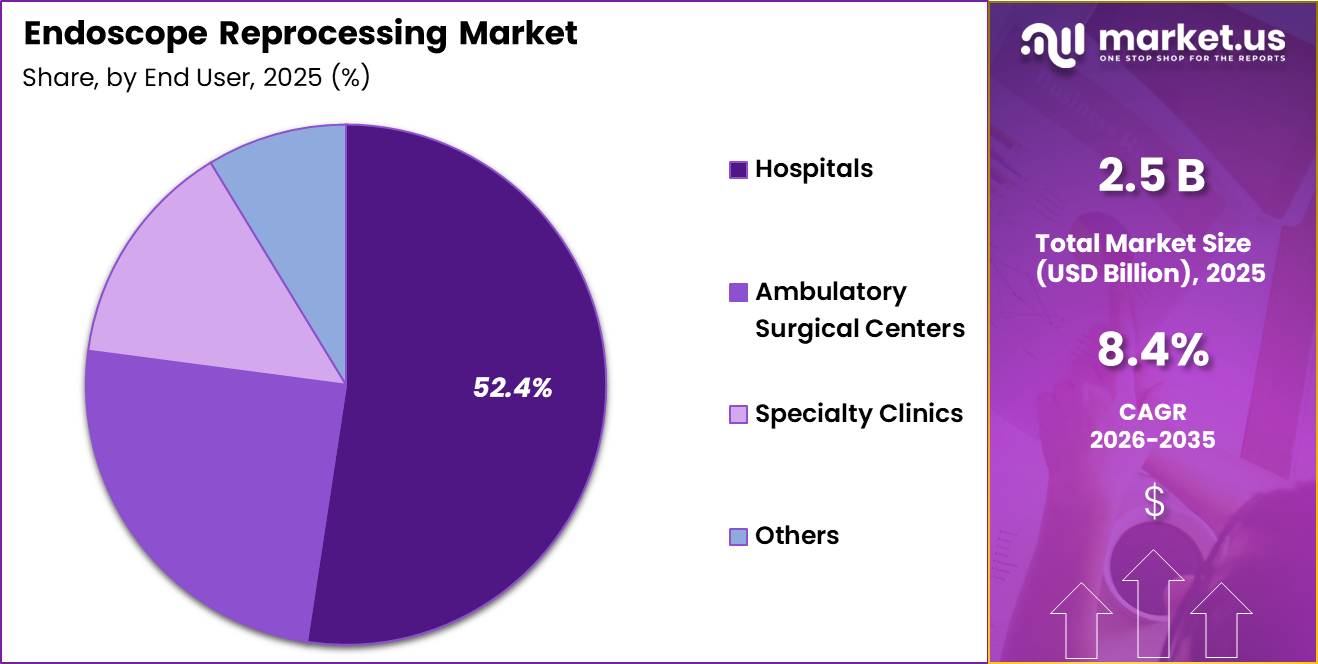
- End User Analysis: Hospitals emerged as the leading segment and captured 52.4% of the global market share in 2025.
- Regional Analysis: In 2025, North America led the market, achieving over 38.2% share with a revenue of US$ 0.65 Billion.
Product Type Analysis
The product type segment of the Endoscope Reprocessing Market is categorized into Automatic Endoscope Reprocessors (AERs), Endoscope Tracking Systems, Consumables and Detergents, and Others. Among these, Automatic Endoscope Reprocessors dominated the market and accounted for 48.5% of the global market share in 2025.
The dominance of this segment can be attributed to the increasing adoption of automated reprocessing solutions across healthcare facilities to reduce manual handling errors, improve workflow efficiency, and ensure compliance with stringent infection prevention standards. The growing number of endoscopic procedures worldwide has further accelerated the deployment of AER systems in hospitals and ambulatory centers.
Endoscope Tracking Systems are witnessing notable growth due to the rising focus on traceability, documentation accuracy, and patient safety. These systems assist healthcare providers in monitoring reprocessing cycles and maintaining regulatory compliance.
Consumables and Detergents also represent a significant share, supported by recurring demand for disinfectants, enzymatic cleaners, and sterilization chemicals required during reprocessing procedures. The Others segment includes drying cabinets, storage systems, and testing products that contribute to enhanced endoscope hygiene management.
Application Analysis
Based on application, the Endoscope Reprocessing Market is segmented into Disinfection and Sterilization. In 2025, the Disinfection segment held the dominant market position and accounted for 56.2% of the total market share. The segment growth is primarily driven by the widespread use of high-level disinfection procedures for flexible endoscopes, particularly in gastrointestinal and pulmonary applications.
Disinfection methods are widely preferred due to their cost-effectiveness, operational efficiency, and ability to eliminate harmful microorganisms while maintaining device integrity. Increasing awareness regarding healthcare-associated infections (HAIs) and stricter regulatory guidelines for endoscope hygiene are further supporting segment expansion.
The Sterilization segment is also experiencing steady growth owing to rising demand for advanced infection control practices in critical care and surgical environments. Sterilization technologies are increasingly being adopted for reusable medical devices that require complete microbial elimination.
Technological advancements in low-temperature sterilization systems and the growing adoption of minimally invasive procedures are contributing to segment development. Furthermore, healthcare facilities are increasingly investing in comprehensive reprocessing protocols to enhance patient safety and reduce contamination risks associated with improperly cleaned endoscopic equipment.
End User Analysis
Based on end user, the Endoscope Reprocessing Market is segmented into Hospitals, Ambulatory Surgical Centers (ASCs), Specialty Clinics, and Others. Hospitals emerged as the leading segment and captured 52.4% of the global market share in 2025.
The dominance of hospitals is primarily attributed to the high volume of endoscopic procedures performed in hospital settings, along with the presence of advanced reprocessing infrastructure and trained healthcare personnel. Hospitals are increasingly investing in automated reprocessing technologies and infection prevention systems to comply with strict healthcare regulations and improve patient safety outcomes.
Ambulatory Surgical Centers represent a rapidly growing segment due to the increasing preference for outpatient minimally invasive procedures and cost-efficient treatment settings. The growing establishment of ASCs across developed and emerging economies is supporting demand for compact and efficient endoscope reprocessing systems.
Specialty Clinics are also contributing significantly to market growth, particularly in gastroenterology, urology, and pulmonology practices where endoscopic procedures are routinely conducted. The Others segment includes diagnostic centers and research institutions that utilize endoscope reprocessing products to maintain hygiene standards and operational efficiency within clinical environments.
Key Market Segments
By Product Type
- Automatic Endoscope Reprocessors
- Endoscope Tracking Systems
- Consumables and Detergents
- Others
By Application
- Disinfection
- Sterilization
By End User
- Hospitals
- Ambulatory Surgical Centers
- Specialty Clinics
- Others
Driving Factors
Rising Endoscopy Volumes and Infection Risk
A key driver for the endoscope reprocessing market is the rapid growth in endoscopic procedure volumes combined with documented infection risks from contaminated scopes. In Finland alone, upper gastrointestinal endoscopies reached an annual rate of 9.30 procedures per 1,000 inhabitants, increasing by 2.6% per year over the study period, indicating sustained growth in demand for safe reprocessing solutions.Flexible gastrointestinal and bronchoscopic endoscopes have been implicated in nosocomial outbreaks; in one U.S. hospital, Pseudomonas aeruginosa colonization or infection occurred in 6.7% of 240 ERCP patients and 8.9% of 1,109 other upper GI endoscopy patients due to reprocessing failures.
CDC guidance stresses that every flexible endoscope must undergo meticulous cleaning and high-level disinfection or sterilization between patients, directly stimulating investment in standardized, automated reprocessing platforms. Together, rising procedure volumes and quantified outbreak data are pushing hospitals and regulators to prioritize robust endoscope reprocessing workflows and technologies.
Trending Factors
Shift to Automation, Surveillance, and Safer Designs
A prominent trend in endoscope reprocessing is the move toward automated systems, enhanced surveillance, and redesigned scopes that reduce contamination risk. The U.S. FDA’s post-market duodenoscope sampling showed high-concern organism contamination rates of 4–6% for older fixed-endcap models, compared with only 1.1% for a newer design, driving interest in devices that are easier to reprocess and in disposable components.FDA safety communications from 2015 onward explicitly recommend supplemental measures such as microbiological culturing, repeat high-level disinfection, and in some settings sterilization, encouraging facilities to adopt automated, trackable reprocessing protocols.
At the same time, infection-control literature describes multi-step reprocessing workflows (pre-cleaning, leak testing, cleaning, high-level disinfection, drying, storage), highlighting cleaning as the most vulnerable step and pushing demand for automated washers, drying cabinets, and real-time quality checks. These regulatory and clinical pressures are normalizing automation, data logging, and human-factors-tested instructions as standard features in reprocessing suites.
Restraining Factors
Persistent Contamination, Complexity, and Cost
Persistent contamination rates, process complexity, and cost pressures act as major restraints on the endoscope reprocessing market. Despite adherence to guidelines, FDA analyses still found contamination with high-concern organisms in up to 6% of sampled older-generation duodenoscopes, indicating that even validated protocols may not fully eliminate risk.Historical CDC investigations documented outbreaks where 6.7% of ERCP patients and 8.9% of upper GI endoscopy patients acquired Pseudomonas aeruginosa due to failures in automated reprocessors, underscoring vulnerability to human error, design flaws, and equipment malfunction.
Reprocessing is also labor-intensive, requiring multiple sequential steps and specialized staff; this complexity increases training needs and operational costs, particularly for smaller hospitals.
Furthermore, regulatory expectations for microbiologic surveillance, enhanced drying, and documentation increase capital and consumable expenditures, which can limit adoption of advanced automated systems in low- and middle-income settings where budget constraints remain acute.
Opportunity
Single-Use, Emerging Markets, and Capacity Gaps
There is significant opportunity for growth in safer technologies, emerging regions, and capacity expansion to meet rising endoscopy demand. FDA now explicitly supports transition to fully disposable duodenoscopes or those with disposable endcaps, creating space for vendors of single-use scopes, compatible reprocessing chemicals, and hybrid workflows that reduce residual contamination risk from complex elevator mechanisms.Systematic reviews show overall bronchoscope contamination rates near 8.7%, and a U.S. Senate investigation linked at least 250 life-threatening infections worldwide to endoscopes between 2012 and 2015, quantifying the potential benefits of safer designs and adjunct reprocessing innovations.
At the same time, device manufacturers highlight large gaps in endoscopy capacity; for example, Japan has about 282 endoscopists per million people versus roughly 72 per million in India, implying substantial latent demand for diagnostic and therapeutic endoscopy infrastructure and associated reprocessing equipment in emerging markets. Vendors that combine robust infection control with cost-efficient solutions can capture this growth.
Regional Analysis
North America dominated the endoscope reprocessing market in 2025, accounting for more than 38.2% of the global market share and generating approximately US$ 0.65 billion in revenue. The regional market growth is primarily supported by the high volume of endoscopic procedures, advanced healthcare infrastructure, and stringent infection prevention regulations across the United States and Canada.
The strong presence of leading healthcare facilities and increasing investments in automated endoscope reprocessors (AERs), drying cabinets, and contamination surveillance systems further contribute to regional expansion.
Europe represented the second-largest regional market due to strict sterilization guidelines implemented by healthcare authorities and rising awareness regarding healthcare-associated infections (HAIs). Countries such as Germany, France, and the United Kingdom are increasingly adopting automated cleaning technologies to improve patient safety and regulatory compliance.
The Asia Pacific region is projected to witness the fastest growth during the forecast period owing to expanding healthcare infrastructure, rising hospital admissions, and increasing adoption of minimally invasive procedures in China, India, and Japan. Growing healthcare expenditure and improving infection control standards are expected to support market growth across emerging economies.
Key Regions and Countries
North America
- The US
- Canada
Europe
- Germany
- France
- The U.K.
- Italy
- Spain
- Russia & CIS
- Rest of Europe
Asia Pacific
- China
- India
- Japan
- South Korea
- ASEAN
- Australia & New Zealand
- Rest of Asia Pacific
Middle East & Africa
- GCC
- South Africa
- Rest of Middle East & Africa
Latin America
- Brazil
- Mexico
- Rest of Latin America
Key Players Analysis
The endoscope reprocessing market is characterized by the presence of several established medical device and infection prevention companies focused on product innovation, automation, and regulatory compliance.
Key players such as Olympus Corporation, STERIS plc, Getinge AB, Cantel Medical Corp., and Advanced Sterilization Products maintain strong market positions through extensive product portfolios and global distribution networks. These companies are actively investing in automated endoscope reprocessors (AERs), drying cabinets, detergents, and tracking systems to improve workflow efficiency and reduce contamination risks.
Strategic initiatives such as mergers, acquisitions, and partnerships are widely adopted to strengthen technological capabilities and geographic presence. For instance, STERIS plc expanded its infection prevention portfolio through the acquisition of Cantel Medical, enhancing its position in the endoscope reprocessing segment.
In addition, manufacturers are emphasizing compliance with stringent guidelines issued by regulatory authorities such as the FDA and CDC, which has accelerated the adoption of high-level disinfection systems and advanced monitoring solutions.
Furthermore, increasing healthcare-associated infection concerns and the growing number of minimally invasive procedures are encouraging companies to develop cost-effective, automated, and user-friendly reprocessing technologies. Continuous innovation and rising hospital investments are expected to intensify competition within the global market.
Market Key Players
- Cantel Medical Corporation
- Olympus Corporation
- STERIS plc
- Getinge Group
- Wassenburg Medical B.V.
- ASP
- Belimed Group
- Custom Ultrasonics, Inc.
- ARC Group
- Shinva Medical Instrument Co., Ltd.
- Borer Chemie AG
- Soluscope SAS
- Endo-Technik GmbH
- Steelco S.p.A.
- Hoya Group
- Others
Recent Developments
- Olympus Corporation (May 2025 ): Olympus launched the ScopeLocker Air endoscope drying cabinet, designed to improve infection prevention by providing continuous airflow through internal channels and around the scope to support guideline‑compliant drying and storage. This move strengthens Olympus’s value proposition beyond visualization, into the “last meter” of safe endoscope turnaround in high‑volume endoscopy units.
- Wassenburg Medical B.V.(April 2026): Wassenburg announced its relocation to a new “future‑proof” headquarters and production facility in Elst, the Netherlands, with the move effective in 2026. The expansion supports higher manufacturing capacity and a broader international footprint for its portfolio across pre‑cleaning, cleaning and disinfection, drying, storage, transport, and traceability of flexible endoscopes.
Report Scope
Report Features Description Market Value (2025) US$ 2.5 Billion Forecast Revenue (2035) US$ 5.7 Billion CAGR (2026-2035) 8.4% Base Year for Estimation 2025 Historic Period 2020-2024 Forecast Period 2026-2035 Report Coverage Revenue Forecast, Market Dynamics, Competitive Landscape, Recent Developments Segments Covered By Product Type (Automatic Endoscope Reprocessors, Endoscope Tracking Systems, Consumables and Detergents, Others) By Application (Disinfection, Sterilization) By End User (Hospitals, Ambulatory Surgical Centers, Specialty Clinics, Others) Regional Analysis North America – The US, Canada; Europe – Germany, France, U.K., Italy, Spain, Russia & CIS, Rest of Europe; Asia Pacific – China, India, Japan, South Korea, ASEAN, Australia & New Zealand, Rest of Asia Pacific; Middle East & Africa – GCC, South Africa, Rest of Middle East & Africa; Latin America – Brazil, Mexico, Rest of Latin America Competitive Landscape Cantel Medical Corporation, Olympus Corporation, STERIS plc, Getinge Group, Wassenburg Medical B.V., ASP, Belimed Group, Custom Ultrasonics, Inc., ARC Group, Shinva Medical Instrument Co., Ltd., Borer Chemie AG, Soluscope SAS, Endo-Technik GmbH, Steelco S.p.A., Hoya Group, Others, Customization Scope Customization for segments, region/country-level will be provided. Moreover, additional customization can be done based on the requirements. Purchase Options We have three licenses to opt for: Single User License, Multi-User License (Up to 5 Users), Corporate Use License (Unlimited User and Printable PDF)  Endoscope Reprocessing MarketPublished date: May 2026add_shopping_cartBuy Now get_appDownload Sample
Endoscope Reprocessing MarketPublished date: May 2026add_shopping_cartBuy Now get_appDownload Sample -
-
- Cantel Medical Corporation
- Olympus Corporation
- STERIS plc
- Getinge Group
- Wassenburg Medical B.V.
- ASP
- Belimed Group
- Custom Ultrasonics, Inc.
- ARC Group
- Shinva Medical Instrument Co., Ltd.
- Borer Chemie AG
- Soluscope SAS
- Endo-Technik GmbH
- Steelco S.p.A.
- Hoya Group
- Others










