Global Drug reference apps Market By Type (Comprehensive Clinical Databases (All-in-One), Drug Interaction Checkers, Safety and Specialty Reference Apps, Formulary and Pricing Apps and Others), By End User (Healthcare Professionals, Patients, Pharmacists, Researchers and Educators and Others), Region and Companies – Industry Segment Outlook, Market Assessment, Competition Scenario, Trends and Forecast 2026-2035
- Published date: March 2026
- Report ID: 180174
- Number of Pages: 221
- Format:
-
keyboard_arrow_up
Quick Navigation
Report Overview
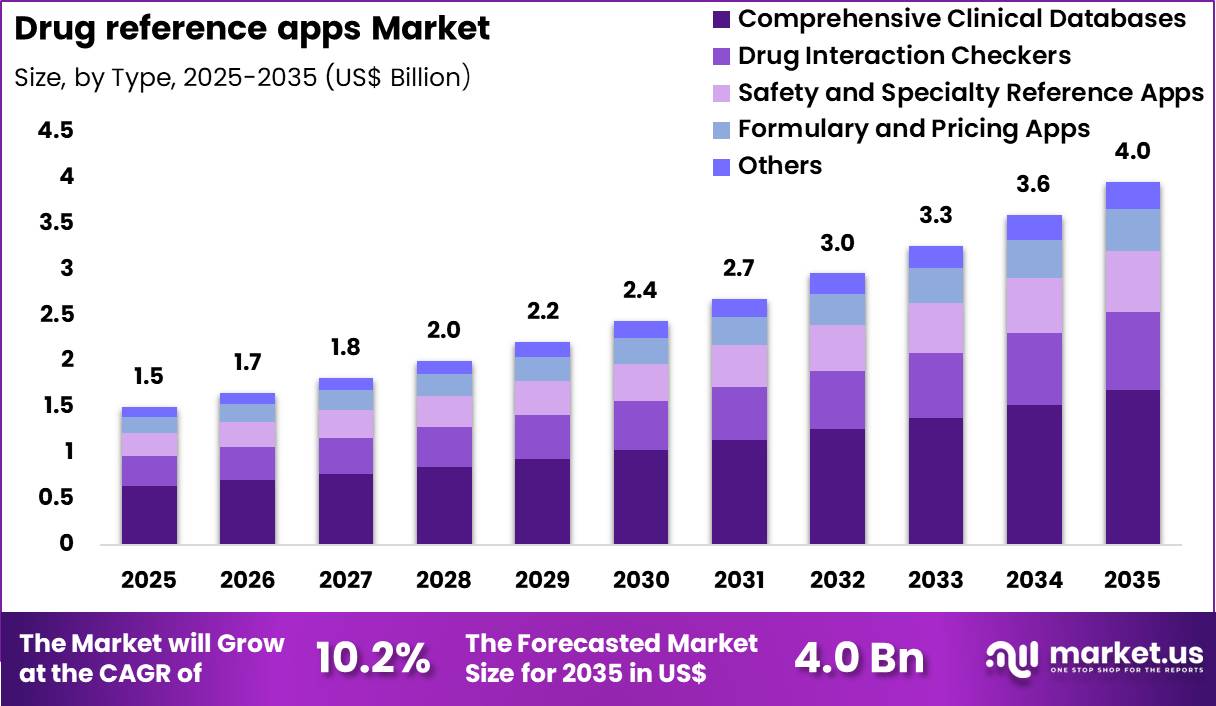
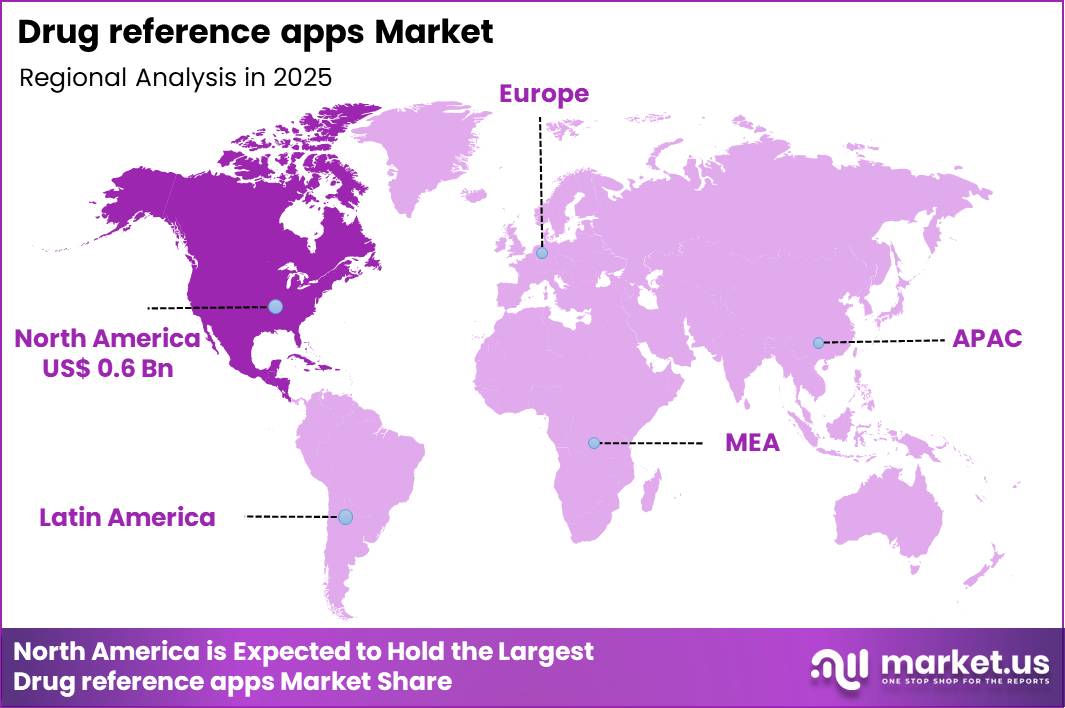
The Global Drug reference apps Market size is expected to be worth around US$ 4.0 Billion by 2035 from US$ 1.5 Billion in 2025, growing at a CAGR of 10.2% during the forecast period 2026-2035. In 2025, North America led the market, achieving over 41.2% share with a revenue of US$ 0.6 Billion.
Rising demand for accurate, instant drug information in fast-paced clinical environments propels the drug reference apps market as healthcare professionals and patients seek reliable digital tools to support safe medication decisions. Physicians increasingly consult these apps during rounds to verify dosing regimens, drug interactions, and contraindications for complex polypharmacy cases involving cardiovascular, psychiatric, or oncology therapies.
Pharmacists utilize them to perform real-time interaction checks when processing prescriptions, preventing adverse events from combinations like warfarin with NSAIDs or statins with certain antibiotics. Nurses apply drug reference apps at the bedside to confirm administration protocols, calculate infusion rates, and identify look-alike/sound-alike medications, enhancing safety in high-acuity settings.
Patients and caregivers rely on consumer-facing versions to review side effects, proper storage, and missed-dose guidance for chronic conditions such as diabetes, hypertension, and epilepsy. These applications also support emergency response by providing rapid access to overdose management protocols and antidote recommendations for poison control scenarios.
Developers pursue opportunities to integrate artificial intelligence that predicts personalized risks based on patient profiles, expanding applications in precision prescribing for geriatric and pediatric populations with altered pharmacokinetics.
Companies advance seamless connectivity with electronic health records and pharmacy systems, enabling automated alerts for allergies, renal adjustments, and pregnancy considerations during order entry. These innovations facilitate multilingual and voice-activated interfaces that improve accessibility for diverse users in multilingual care teams.
Opportunities emerge in subscription-based premium features offering off-line access and advanced formulary tools for ambulatory care. Recent trends emphasize real-time updates from regulatory databases, gamified learning modules for trainees, and integration with wearable health devices for adherence tracking, positioning drug reference apps as indispensable resources in modern medication management across clinical, retail, and home settings.
Key Takeaways
- In 2025, the market generated a revenue of US$ 1.5 Billion, with a CAGR of 10.2%, and is expected to reach US$ 4.0 Billion by the year 2035.
- The type segment is divided into comprehensive clinical databases, drug interaction checkers, safety and specialty reference apps, formulary and pricing apps and others, with comprehensive clinical databases taking the lead with a market share of 42.5%.
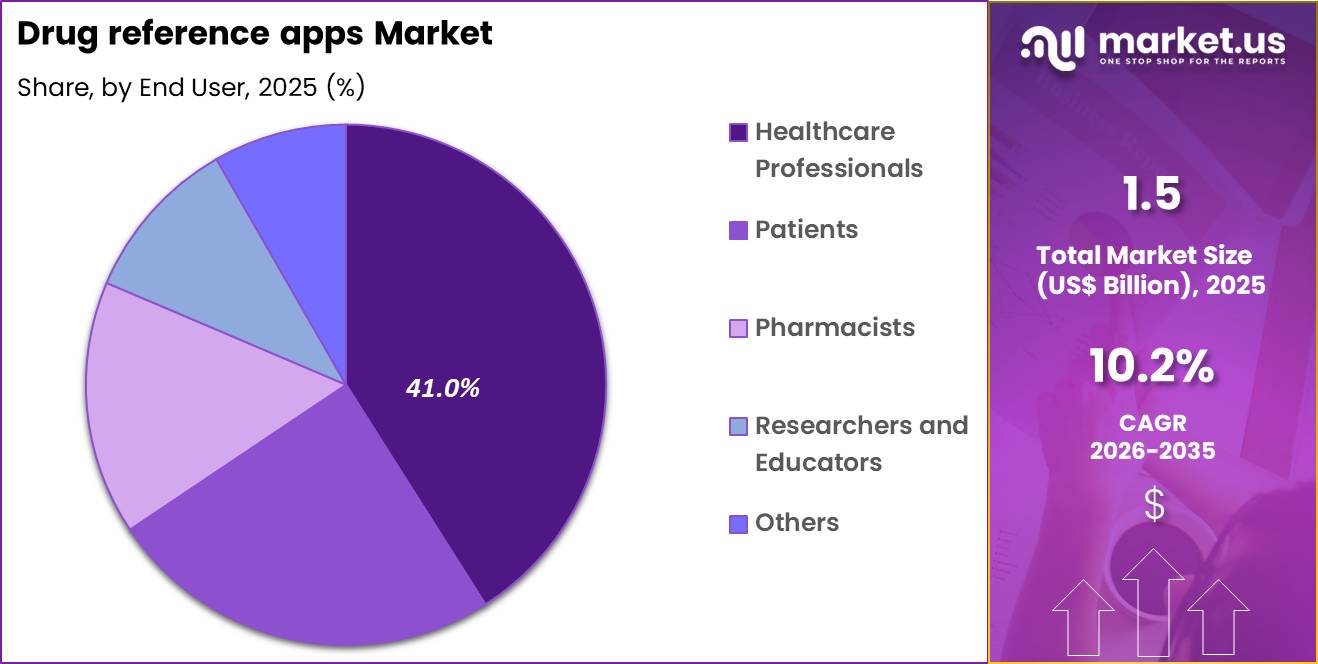
- Considering end user, the market is divided into healthcare professionals, patients, pharmacists, researchers and educators and others. Among these, healthcare professionals held a significant share of 41.0%.
- North America led the market by securing a market share of 41.2%.
Type Analysis
Comprehensive clinical databases accounted for 42.5% of growth within type and dominate the drug reference apps market due to their extensive coverage of drug monographs, dosing guidelines, contraindications, and clinical updates. Healthcare professionals increasingly rely on these platforms for evidence-based decision support at the point of care.
Segment growth is projected to strengthen as digital health adoption accelerates across hospitals and outpatient settings. Rising complexity of treatment regimens and polypharmacy management drives demand for centralized, regularly updated drug information.
Developers integrate real-time alerts, guideline updates, and clinical calculators, enhancing usability and workflow efficiency. Subscription-based revenue models and institutional licensing agreements further support expansion.
Growing emphasis on patient safety and medication error reduction reinforces adoption among clinicians. Integration with electronic health records and telehealth platforms is anticipated to create additional growth opportunities.
End-User Analysis
Healthcare professionals accounted for 41.0% of growth within end users and represent the dominant segment due to their continuous need for accurate and up-to-date drug information. Physicians, nurses, and allied health practitioners use these apps during consultations, prescribing, and treatment planning.
Segment growth is expected to continue as clinical environments prioritize digital tools that enhance efficiency and reduce errors. Increasing patient volumes and complex therapeutic protocols encourage reliance on mobile decision-support applications. Continuing medical education requirements and guideline updates further drive frequent usage.
Healthcare institutions increasingly adopt enterprise subscriptions to standardize clinical information access across departments. Rising adoption of smartphones and tablets in clinical practice supports consistent engagement. Expansion of telemedicine services is likely to increase reliance on mobile drug reference solutions for remote consultations.
Key Market Segments
By Type
- Comprehensive Clinical Databases (All-in-One)
- Drug Interaction Checkers
- Safety and Specialty Reference Apps
- Formulary and Pricing Apps
- Others
By End User
- Healthcare Professionals
- Patients
- Pharmacists
- Researchers and Educators
- Others
Drivers
Increasing adoption of mobile health tools by healthcare professionals is driving the market.
The growing reliance on smartphones for clinical decision support has substantially increased the utilization of drug reference apps among physicians, pharmacists, and nurses for instant access to medication information. Greater integration of these apps into daily workflows has led to higher usage for checking dosages, interactions, and contraindications during patient consultations.
Healthcare institutions are encouraging the use of validated apps to reduce medication errors and improve prescribing accuracy. The correlation between busy clinical environments and the need for quick, evidence-based resources further amplifies demand for these digital tools. Government health agencies are promoting digital health literacy among providers to support safe medication practices.
Drug reference apps offer comprehensive, regularly updated databases that facilitate better patient safety outcomes. National initiatives for electronic health record integration encourage seamless use of these apps in hospital systems.
Key developers are enhancing features like offline access to align with this clinical imperative. This driver fosters ongoing improvements in app usability and content reliability. In 2022, over 350,000 healthcare apps were available on global app stores, with drug reference tools forming a significant portion used by professionals.
Restraints
Concerns over app accuracy and data reliability are restraining the market.
The persistent doubts regarding the accuracy of information in some drug reference apps have limited full trust and adoption among cautious healthcare professionals. Variations in update frequency and source validation across different apps create hesitation in critical decision-making scenarios. Regulatory bodies have highlighted the risks of outdated or incomplete data leading to potential medication errors.
Healthcare providers often cross-verify app information with official sources, reducing efficiency gains. The correlation between app quality inconsistencies and clinician reluctance further constrains market growth. Government agencies stress the importance of evidence-based resources for patient safety. National guidelines for digital health tools recommend rigorous evaluation before routine use.
Key stakeholders face challenges in maintaining consistent data quality across platforms. This restraint impacts the perceived value of these apps in high-stakes clinical environments. Concerns over app accuracy and data reliability remain a primary market restraint.
Opportunities
Integration with electronic health record systems is creating growth opportunities.
The increasing interoperability between drug reference apps and hospital electronic health record systems presents avenues for seamless access to medication data during patient encounters. Governmental policies supporting digital health infrastructure encourage the development of API connections for real-time information sharing.
Healthcare networks are seeking unified platforms that combine reference tools with patient records for improved workflow efficiency. Partnerships with EHR vendors facilitate customized integrations for institutional use. The large installed base of electronic health records in hospitals magnifies prospects for embedded reference functionality.
Educational programs for clinicians promote the benefits of connected systems in daily practice. This opportunity allows developers to expand beyond standalone apps into enterprise solutions. Leading companies are prioritizing compatibility features for major EHR platforms.
Overall, EHR integration aligns with efforts to streamline clinical decision-making and reduce errors. Integration with electronic health record systems is a key growth opportunity.
Impact of Macroeconomic / Geopolitical Factors
Macroeconomic trends influence the drug reference apps market by shaping healthcare IT budgets, subscription spending, and advertising revenue streams. Persistent inflation and elevated interest rates reduce discretionary spending among smaller clinics and independent practitioners, which slows premium app adoption.
Geopolitical tensions complicate cross-border data hosting, content licensing, and cybersecurity compliance, increasing operational oversight costs. Current US tariffs on imported smartphones, tablets, and certain networking hardware indirectly raise device prices, which can limit user upgrades and slow platform optimization. These pressures challenge emerging developers that rely on rapid scaling and hardware integration.
At the same time, cost awareness drives clinicians toward digital tools that improve prescribing accuracy and reduce medication errors. Expanding telehealth services and mobile-first clinical workflows continue to increase reliance on real-time drug databases. With scalable cloud infrastructure and evidence-based content updates, the market sustains steady long-term growth potential.
Latest Trends
Launch of open-access comprehensive drug databases is a recent trend in the market.
In 2025, the introduction of open-access comprehensive drug databases has expanded free, evidence-based resources for healthcare professionals and researchers. These platforms aggregate clinically tested drug information in user-friendly formats for quick reference. Developers have emphasized transparency and regular updates to support clinical decision-making.
Academic institutions have adopted these tools for education and research purposes. Purdue University’s Center for Medication Safety launched the first comprehensive open-access database of all clinically tested drugs in May 2025. This initiative provides centralized, peer-reviewed information on medications to enhance safety and collaboration.
The trend focuses on accessibility for global users without subscription barriers. Regulatory and academic endorsements in 2025 have accelerated adoption. Industry and academic collaborations refine search functionalities for better usability. These developments aim to democratize drug information while maintaining high standards of reliability.
Regional Analysis
North America is leading the Drug reference apps Market
North America accounted for 41.2% of the drug reference apps market in 2024, as clinicians and patients increasingly turned to digital tools for quick access to medication information and clinical decision support. In the U.S., 57% of individuals reported using an app to access their health records in 2024, up from 51% in 2022, demonstrating a growing comfort with mobile health applications that support broader digital health engagement.
Widespread smartphone penetration and deep integration of electronic health record systems encouraged healthcare providers to recommend and deploy drug compendiums that link seamlessly with clinical workflows. Hospital systems and outpatient clinics adopted reference apps to reduce prescription errors and enhance drug safety, while pharmacists used them for interaction checks and dosage guidance during counseling.
Consumer awareness of medication safety and self-management also rose as patients sought reliable, up-to-date information on their prescriptions. Partnerships between app developers and academic medical centers improved content accuracy and clinical relevance, strengthening trust among professional users.
Healthcare curricula increasingly emphasized digital literacy, supporting clinician familiarity with point-of-care reference tools. Payers and insurers acknowledged the role of digital utilities in promoting adherence and reducing adverse drug events, indirectly supporting uptake. These combined trends underpinned notable regional growth in 2024.
The Asia Pacific region is expected to experience the highest CAGR during the forecast period
In Asia Pacific, expansion through the forecast period will be propelled by rapid digital transformation across healthcare systems and surging adoption of mobile health solutions among both clinicians and patients. The broader mHealth ecosystem in the region registered significant traction, with the Asia Pacific mHealth apps sector generating substantial engagement in 2023 and expanding at a robust pace into 2024, reflecting heightened uptake of medical and drug information applications as part of basic health management services.
Rising smartphone ownership and internet connectivity widened access to digital health utilities, particularly in India, China, and Southeast Asia, where growing middle-class populations prioritized convenient health-related information tools. National health initiatives in multiple countries promoted e-health literacy and supported investment in digital platforms that facilitate medication guidance and safety.
Clinicians embraced apps that help streamline prescribing practices and alert users to potential interactions, strengthening clinical confidence. Healthcare training programs incorporated digital resources into continuing education, accelerating professional adoption. Consumer health awareness campaigns encouraged patients to engage with technology for medication reminders and reference needs, especially for chronic conditions.
Interoperability improvements enabled better integration of digital tools with existing healthcare IT infrastructure, increasing utility across care settings. Cross-border collaborations facilitated localized content and language support, making these solutions more relevant in diverse markets. Taken together, these factors are positioned to sustain strong regional growth in the years ahead.
Key Regions and Countries
North America
- US
- Canada
Europe
- Germany
- France
- The UK
- Spain
- Italy
- Russia
- Netherland
- Rest of Europe
Asia Pacific
- China
- Japan
- South Korea
- India
- Australia
- New Zealand
- Singapore
- Thailand
- Vietnam
- Rest of APAC
Latin America
- Brazil
- Mexico
- Rest of Latin America
Middle East & Africa
- South Africa
- Saudi Arabia
- UAE
- Rest of MEA
Key Players Analysis
Key participants in the Drug Reference Apps market accelerate growth by strengthening clinical content accuracy, integrating AI-driven interaction checkers, and expanding subscription-based access models for hospitals and retail pharmacies. They collaborate with healthcare systems and academic institutions to embed their platforms into daily prescribing workflows and electronic medical record ecosystems.
Wolters Kluwer Health, a global healthcare information and technology company headquartered in the Netherlands, delivers evidence-based clinical decision support solutions and serves professionals across more than 180 countries.
The company focuses on continuous database updates, regulatory alignment, and mobile-first functionality to retain clinician trust. Competitors invest in multilingual capabilities and region-specific drug formularies to penetrate emerging markets.
These combined initiatives enhance user engagement, build long-term contracts, and strengthen recurring revenue streams across the Drug Reference Apps market.
Top Key Players
- Epocrates
- Medscape
- Micromedex (Merative US L.P.)
- PillPack
- Pill Identifier & Med Scanner
- Mango Health
- com
- MDCalc
- Medisafe
- MyTherapy
- Others
Recent Developments
- In September 2025, Glooko announced plans to broaden its footprint in hospital-based diabetes care through the acquisition of Monarch Medical Technologies, including its EndoTool insulin dosing software. The move is intended to strengthen inpatient glucose management capabilities, particularly as an estimated 30–40% of hospitalized patients require insulin therapy to maintain glycemic control during treatment.
- In March 2025, Wolters Kluwer incorporated its UpToDate drug reference content into Suki’s AI-powered clinical assistant platform. This integration enhances real-time clinical decision support by embedding evidence-based medication guidance directly within AI-driven physician workflows.
Report Scope
Report Features Description Market Value (2025) US$ 1.5 Billion Forecast Revenue (2035) US$ 4.0 Billion CAGR (2026-2035) 10.2% Base Year for Estimation 2025 Historic Period 2020-2024 Forecast Period 2026-2035 Report Coverage Revenue Forecast, Market Dynamics, COVID-19 Impact, Competitive Landscape, Recent Developments Segments Covered By Type (Comprehensive Clinical Databases (All-in-One), Drug Interaction Checkers, Safety and Specialty Reference Apps, Formulary and Pricing Apps and Others), By End User (Healthcare Professionals, Patients, Pharmacists, Researchers and Educators and Others) Regional Analysis North America – US, Canada; Europe – Germany, France, The UK, Spain, Italy, Russia, Netherlands, Rest of Europe; Asia Pacific – China, Japan, South Korea, India, Australia, New Zealand, Singapore, Thailand, Vietnam, Rest of APAC; Latin America – Brazil, Mexico, Rest of Latin America; Middle East & Africa – South Africa, Saudi Arabia, UAE, Rest of MEA Competitive Landscape Epocrates, Medscape, Micromedex (Merative US L.P.), PillPack, Pill Identifier & Med Scanner, Mango Health, Drugs.com, MDCalc, Medisafe, MyTherapy, Others. Customization Scope Customization for segments, region/country-level will be provided. Moreover, additional customization can be done based on the requirements. Purchase Options We have three licenses to opt for: Single User License, Multi-User License (Up to 5 Users), Corporate Use License (Unlimited User and Printable PDF)  Drug reference apps MarketPublished date: March 2026add_shopping_cartBuy Now get_appDownload Sample
Drug reference apps MarketPublished date: March 2026add_shopping_cartBuy Now get_appDownload Sample -
-
- Epocrates
- Medscape
- Micromedex (Merative US L.P.)
- PillPack
- Pill Identifier & Med Scanner
- Mango Health
- com
- MDCalc
- Medisafe
- MyTherapy
- Others










