Global 3D Bioprinted Human Tissue Market By Type (Skin Tissue, Bone and Cartilage, Vascular Grafts, Cardiac Tissue, Liver Tissue and Others), By Application (Regenerative Medicine & Transplantation, Pharmaceutical & Drug Discovery, Cancer Research and Others), By End Users (Medical Device Manufacturers, Hospitals and Clinical Centers, Pharmaceutical and Biotechnology Companies, Academic and Research Institutions and Others), Region and Companies – Industry Segment Outlook, Market Assessment, Competition Scenario, Trends and Forecast 2026-2035
- Published date: March 2026
- Report ID: 180779
- Number of Pages: 318
- Format:
-
keyboard_arrow_up
Quick Navigation
Report Overview
The Global 3D Bioprinted Human Tissue Market size is expected to be worth around US$ 3.9 Billion by 2035 from US$ 2.5 Billion in 2025, growing at a CAGR of 4.6% during the forecast period 2026-2035. In 2025, North America led the market, achieving over 41.3% share with a revenue of US$ 1.0 Billion.
Increasing demand for regenerative medicine and organ replacement solutions accelerates the 3D bioprinted human tissue market as researchers and clinicians seek advanced constructs that replicate native tissue architecture and function.
Scientists increasingly apply bioprinted skin equivalents in burn wound management, creating multilayered dermal-epidermal constructs that promote rapid re-epithelialization and reduce scarring in extensive thermal injuries. These tissues support cartilage repair in orthopedic applications, where bioinks seeded with chondrocytes form hyaline-like structures for focal chondral defect restoration in knee joints.

Cardiovascular researchers utilize bioprinted vascular grafts and cardiac patches to address coronary artery disease and myocardial infarction, engineering vessels with endothelialized lumens or patches containing cardiomyocytes to improve contractility and reduce scar formation.
In drug development, bioprinted liver tissue models enable accurate hepatotoxicity screening and metabolism studies, providing physiologically relevant platforms for pharmaceutical testing. Bioprinted tumor models facilitate oncology research by recreating patient-specific microenvironments that allow evaluation of personalized cancer therapies and drug resistance mechanisms.
Manufacturers pursue opportunities to develop hybrid bioprinting approaches that combine cells, biomaterials, and growth factors, expanding applications in organoid generation for intestinal and renal tissue engineering. Developers advance in vivo bioprinting techniques that deposit bioinks directly within the body under imaging guidance, enabling localized tissue regeneration in musculoskeletal and neural defects.
These innovations facilitate integration with organ-on-chip systems for dynamic, multi-tissue interactions in preclinical testing. Opportunities emerge in scalable, GMP-compliant bioinks and printing platforms that support clinical translation of bioprinted constructs. Companies invest in vascularization strategies using sacrificial channels or endothelial cells to create perfusable tissues for larger-scale implants.
In May 2025, scientists at the California Institute of Technology demonstrated an ultrasound-guided in vivo 3D printing technique capable of forming structures directly inside the body. The technology enables localized delivery of therapeutic cells and drugs at targeted sites.
In April 2025, CN Bio entered a long-term collaboration with Pharmaron to incorporate organ-on-chip technologies and bioprinted tissues into drug discovery and development programs, strengthening translational research capabilities across global pharmaceutical workflows. Recent trends emphasize patient-specific bioprinting and multi-material constructs, positioning the market for transformative impact in regenerative therapies and precision medicine.
Key Takeaways
- In 2025, the market generated a revenue of US$ 2.5 Billion, with a CAGR of 4.6%, and is expected to reach US$ 3.9 Billion by the year 2035.
- The type segment is divided into skin tissue, bone and cartilage, vascular grafts, cardiac tissue, liver tissue and others, with skin tissue taking the lead with a market share of 28.6%.
- Considering application, the market is divided into regenerative medicine & transplantation, pharmaceutical & drug discovery, cancer research and others. Among these, regenerative medicine & transplantation held a significant share of 41.8%.
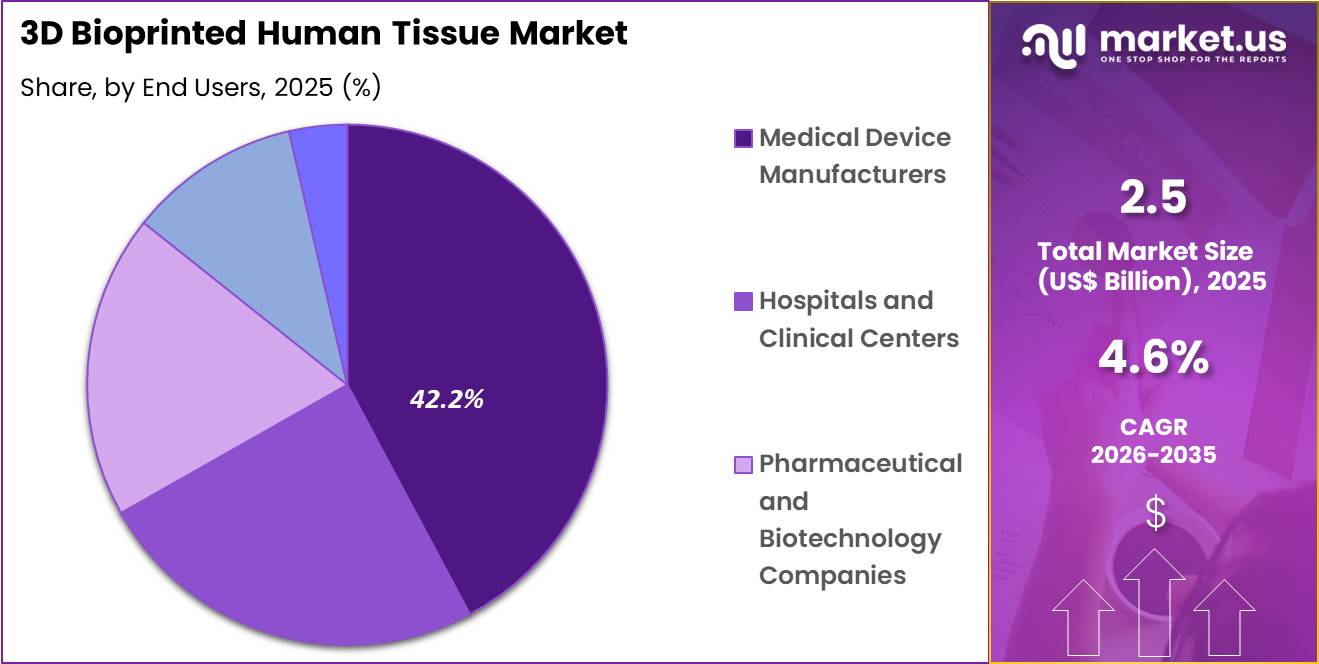
- Furthermore, concerning the end users segment, the market is segregated into medical device manufacturers, hospitals and clinical centers, pharmaceutical and biotechnology companies, academic and research institutions and others. The medical device manufacturers sector stands out as the dominant player, holding the largest revenue share of 42.2% in the market.
- North America led the market by securing a market share of 41.3%.
Type Analysis
Skin tissue accounted for 28.6% of growth within type and dominate the 3D bioprinted human tissue market due to the strong clinical and research need for skin repair, wound modeling, and burn care solutions. Skin remains one of the most accessible tissues for bioprinting because its layered structure is comparatively easier to replicate than highly vascularized internal organs.
The World Health Organization states that burns cause around 180,000 deaths annually worldwide, which highlights the persistent need for better tissue repair technologies and regenerative substitutes. Researchers and commercial developers increasingly focus on bioprinted skin to support graft development, toxicity testing, and cosmetic research.
The segment is expected to strengthen as demand rises for personalized wound treatment and lab-grown tissue alternatives that reduce dependence on conventional grafting methods.
Application Analysis
Regenerative medicine and transplantation accounted for 41.8% of growth within application and dominate the market because 3D bioprinting directly addresses the growing need for functional tissue replacement and repair. This segment gains traction as healthcare systems and research groups look for ways to reduce transplant dependency, improve tissue compatibility, and support faster healing in severe injury cases.
In the United States alone, organdonor.gov reports that more than 103,000 people are currently on the national transplant waiting list, which underscores the scale of unmet need for replacement tissues and organs.
Bioprinted tissues are anticipated to gain wider use in regenerative workflows because they support patient-specific designs and improve preclinical understanding of tissue integration. The segment is projected to expand further as tissue engineering programs align more closely with transplantation science and personalized medicine initiatives.
End-User Analysis
Medical device manufacturers accounted for 42.2% of growth within end users and dominate the 3D bioprinted human tissue market because they play a central role in commercializing tissue engineering platforms, scaffold systems, and bioprinting-enabled clinical products. These companies invest in biomaterials, printer hardware, and regulatory development to move engineered tissues from research settings toward scalable healthcare use.
FDA materials on 3D printing note that medical applications already include orthopedic and cranial implants, surgical tools, and patient-matched products, which reflects the broader momentum behind additive manufacturing in healthcare.
Device manufacturers are expected to lead this segment because they combine engineering expertise, production capability, and commercialization channels more effectively than most research institutions. Their position is likely to strengthen as hospitals and biotech firms increasingly seek industrial partners to translate bioprinted tissue concepts into usable clinical and testing solutions.
Key Market Segments
By Type
- Skin Tissue
- Bone and Cartilage
- Vascular Grafts
- Cardiac Tissue
- Liver Tissue
- Others
By Application
- Regenerative Medicine & Transplantation
- Pharmaceutical & Drug Discovery
- Cancer Research
- Others
By End Users
- Medical Device Manufacturers
- Hospitals and Clinical Centers
- Pharmaceutical and Biotechnology Companies
- Academic and Research Institutions
- Others
Drivers
Improved EBITDA in the Bioprinting segment is driving the market.
BICO Group documented notable profitability enhancements within its Bioprinting operations during the early phase of the review period. The segment’s adjusted EBITDA reached SEK 86.1 million in 2023, representing a margin of 13.0%. This marked a substantial improvement from SEK 33.3 million in 2022, equivalent to a 5.1 percent margin.
The progression reflects optimized cost structures and heightened efficiency in 3D bioprinting production processes. Key contributions arose from scaled bioink formulations and bioprinter deployments in research applications. Stakeholders observe this financial strengthening as validation of technological maturation in tissue engineering.
The uplift supports reinvestment in R&D for advanced vascularized constructs. Clinical collaborators benefit from more reliable supply of bioprinted prototypes. Such performance indicators encourage partnerships with pharmaceutical entities for drug testing models. Overall, the EBITDA trajectory underpins accelerated commercialization of 3D bioprinted human tissues.
Restraints
Decline in Bioprinting net sales is restraining the market.
BICO Group reported a contraction in its Bioprinting segment net sales for the subsequent year within the specified timeframe. Net sales totaled SEK 369.3 million in 2024. This figure declined from SEK 421.0 million in 2023, indicating a 12% reduction. Organic growth measured negative 11.9%, excluding foreign exchange effects.
The downturn stems from delayed adoption in certain academic and industrial pipelines. Manufacturers face adjusted production schedules amid softer demand forecasts. Investment in inventory management becomes critical to mitigate excess capacity.
The restraint influences confidence in scaling bioprinting infrastructure. Providers encounter challenges in forecasting long-term procurement needs. This dynamic tempers the sector’s momentum during the 2023-2024 transition.
Opportunities
NIH funding for 3D bioprinting vascularization research is creating growth opportunities.
The National Institutes of Health awarded a US$3 million grant to Penn State researchers on September 8, 2025, targeting bioprinted vascular solutions for reconstructive surgery. This initiative addresses persistent challenges in integrating blood supply within engineered tissues. Opportunities emerge for hybrid constructs combining patient-derived cells with synthetic scaffolds.
The funding enables development of perfusable networks mimicking native vasculature. Collaborators can explore scalability for clinical translation in wound healing applications. The grant fosters interdisciplinary teams spanning bioengineering and surgical disciplines. Advanced imaging techniques will validate construct functionality pre-implantation.
Such support accelerates progression toward regulatory milestones for therapeutic use. Stakeholders anticipate spillover benefits in cosmetic and trauma repair segments. This opportunity enhances the ecosystem’s capacity for innovative tissue replacements.
Impact of Macroeconomic / Geopolitical Factors
Macroeconomic conditions influence the 3D bioprinted human tissue market through research funding, biotechnology investment, and laboratory infrastructure spending. Inflation raises costs for bioinks, cell culture materials, precision printers, and laboratory utilities, which increases operational expenses for research institutions and biotech firms.
Higher interest rates reduce venture capital availability and slow funding for early stage regenerative medicine projects. Geopolitical tensions affect global supply of specialized biomaterials, microfluidic components, and high precision printing hardware, creating procurement delays.
Current US tariffs on imported bioprinting equipment and laboratory components increase acquisition costs and narrow margins for technology providers. These pressures can delay commercialization timelines and limit experimentation in smaller research labs.
At the same time, organizations strengthen domestic manufacturing partnerships and invest in local research ecosystems to reduce dependency on imports. Growing demand for personalized medicine, drug testing platforms, and regenerative therapies continues to support steady and confident market growth.
Latest Trends
FDA approval for 3D bioprinted peripheral nerve repair device is driving the market.
3D Systems announced FDA clearance on June 26, 2025, for a pioneering bioabsorbable device enabling sutureless peripheral nerve regeneration. The approval leverages proprietary bioprinting to fabricate customized nerve guides from biocompatible materials. This milestone represents the first regulatory endorsement for such 3D-printed regenerative implants.
Clinicians gain tools for precise alignment and gap bridging in traumatic injuries. The device integrates growth factor delivery to promote axonal regrowth. Partnership with TISSIUM facilitated the adhesive-free fixation mechanism. The 2025 authorization targets the $2.4 billion peripheral nerve repair sector.
Facilities anticipate streamlined procedures with reduced revision rates. Innovation in resolution and material selection sets new benchmarks. Overall, this development catalyzes adoption of bioprinted solutions in neurosurgical practices.
Regional Analysis
North America is leading the 3D Bioprinted Human Tissue Market
North America accounted for 41.3% of the 3D bioprinted human tissue market in 2025 as research institutions and biotechnology companies accelerated development of advanced tissue engineering platforms for regenerative medicine and drug testing. Strong government support for biomedical innovation has played a major role in expanding laboratory capacity for bioprinting technologies.
The National Institutes of Health allocated about USD 47.7 billion in funding for medical research in 2023, supporting extensive research programs in tissue engineering, organ regeneration, and biofabrication technologies. Universities and biotechnology startups across the United States and Canada increasingly collaborate to develop laboratory grown tissues that can support disease modeling and pharmaceutical testing.
Pharmaceutical companies are integrating engineered tissue models into preclinical research to improve drug safety evaluation and reduce reliance on animal testing. Advances in bioinks, scaffold design, and high precision printing technologies have also improved the viability and structural stability of printed tissues.
Medical research centers are establishing dedicated biofabrication laboratories to explore applications in wound healing, transplant research, and organ repair. Regulatory agencies are also engaging with innovators to evaluate the clinical potential of engineered tissues for future therapeutic use. These developments collectively strengthened the growth trajectory of tissue biofabrication technologies across North America in 2025.
The Asia Pacific region is expected to experience the highest CAGR during the forecast period
Asia Pacific is expected to experience significant expansion during the forecast period as governments increase investments in biotechnology innovation and regenerative medicine research. Several countries in the region are strengthening national strategies that support tissue engineering and advanced biomedical manufacturing.
China’s National Bureau of Statistics reported that the country’s research and development spending reached about 3.09 trillion yuan in 2022, reflecting strong commitment to scientific innovation including bioengineering technologies. Universities and biotechnology companies across China, Japan, South Korea, and Singapore are expanding laboratory research focused on engineered tissues for drug discovery and regenerative therapies.
Pharmaceutical firms in the region are adopting advanced tissue models to improve accuracy in toxicity testing and disease simulation. Academic research centers are also collaborating with international institutions to accelerate breakthroughs in biofabrication and cell engineering.
Governments are encouraging development of local biotechnology industries through funding programs and innovation parks. Rising healthcare demand and increasing interest in personalized medicine are also stimulating research in engineered tissue technologies. These developments are expected to encourage broader adoption of advanced tissue fabrication platforms across Asia Pacific in the coming years.

Key Regions and Countries
North America
- US
- Canada
Europe
- Germany
- France
- The UK
- Spain
- Italy
- Russia
- Netherland
- Rest of Europe
Asia Pacific
- China
- Japan
- South Korea
- India
- Australia
- New Zealand
- Singapore
- Thailand
- Vietnam
- Rest of APAC
Latin America
- Brazil
- Mexico
- Rest of Latin America
Middle East & Africa
- South Africa
- Saudi Arabia
- UAE
- Rest of MEA
Key Players Analysis
Key players in the 3D Bioprinted Human Tissue market strengthen growth through intensive research partnerships, expansion of bio-ink technologies, and continuous development of advanced bioprinting platforms that enable precise fabrication of living tissue models.
Companies collaborate closely with pharmaceutical firms and academic laboratories to accelerate drug discovery and disease modeling using engineered tissue constructs. Many firms also invest in automated biomanufacturing systems and scalable bioprinting tools to improve reproducibility and commercial viability of tissue engineering applications.
Organovo Holdings Inc. represents a recognized participant in the 3D Bioprinted Human Tissue market and operates as a biotechnology company headquartered in San Diego that specializes in creating functional human tissues through proprietary bioprinting technology for research and therapeutic applications.
The company develops three-dimensional tissue models that assist pharmaceutical companies in evaluating drug toxicity and biological responses more efficiently. Industry competitors such as CELLINK, Aspect Biosystems, and 3D Systems continue to launch advanced printers, expand biomaterial portfolios, and form strategic collaborations to advance regenerative medicine and tissue engineering solutions worldwide.
Top Key Players
- Allevi (3D Systems)
- Cellink Global (BICO Group)
- Cyfuse Biomedical K.K.
- EnvisionTEC Inc. (Desktop Health)
- Inventia Life Science PTY LTD
- Organovo Holdings Inc.
- Poietis
- Stratasys Ltd.
Recent Developments
- In June 2025, researchers at Stanford University reported a new technique that accelerates the creation of vascular networks within bioprinted tissues by roughly 200 times compared with earlier methods. The advancement enables faster production of large, organ-scale tissue constructs for regenerative medicine research.
- In June 2025, Stony Brook University introduced the TRACE manufacturing method, a process designed to print collagen structures that closely replicate the architecture of natural biological tissues. The approach aims to improve structural accuracy in bioprinted biomaterials.
- In February 2025, Enovis finalized the acquisition of LimaCorporate in a transaction valued at approximately EUR 800 million. The deal expands Enovis’ presence in orthopedic technologies, particularly in implants produced using advanced 3D printing methods.
Report Scope
Report Features Description Market Value (2025) US$ 2.5 Billion Forecast Revenue (2035) US$ 3.9 Billion CAGR (2026-2035) 4.6% Base Year for Estimation 2025 Historic Period 2020-2024 Forecast Period 2026-2035 Report Coverage Revenue Forecast, Market Dynamics, COVID-19 Impact, Competitive Landscape, Recent Developments Segments Covered By Type (Skin Tissue, Bone and Cartilage, Vascular Grafts, Cardiac Tissue, Liver Tissue and Others), By Application (Regenerative Medicine & Transplantation, Pharmaceutical & Drug Discovery, Cancer Research and Others), By End Users (Medical Device Manufacturers, Hospitals and Clinical Centers, Pharmaceutical and Biotechnology Companies, Academic and Research Institutions and Others) Regional Analysis North America – US, Canada; Europe – Germany, France, The UK, Spain, Italy, Russia, Netherlands, Rest of Europe; Asia Pacific – China, Japan, South Korea, India, Australia, New Zealand, Singapore, Thailand, Vietnam, Rest of APAC; Latin America – Brazil, Mexico, Rest of Latin America; Middle East & Africa – South Africa, Saudi Arabia, UAE, Rest of MEA Competitive Landscape Allevi, Cellink Global, Cyfuse Biomedical, EnvisionTEC, Inventia Life Science, Organovo Holdings, Poietis, Stratasys Ltd. Customization Scope Customization for segments, region/country-level will be provided. Moreover, additional customization can be done based on the requirements. Purchase Options We have three licenses to opt for: Single User License, Multi-User License (Up to 5 Users), Corporate Use License (Unlimited User and Printable PDF)  3D Bioprinted Human Tissue MarketPublished date: March 2026add_shopping_cartBuy Now get_appDownload Sample
3D Bioprinted Human Tissue MarketPublished date: March 2026add_shopping_cartBuy Now get_appDownload Sample -
-
- Allevi (3D Systems)
- Cellink Global (BICO Group)
- Cyfuse Biomedical K.K.
- EnvisionTEC Inc. (Desktop Health)
- Inventia Life Science PTY LTD
- Organovo Holdings Inc.
- Poietis
- Stratasys Ltd.










