Global Rapid Hepatitis Testing Market By Test Type (Serologic Antibody Assays, Molecular HCV RNA Tests, Immunoassays for HCV Core Antigen), By Indication (Hepatitis A, Hepatitis B, Hepatitis C), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, and Online Pharmacies), Region and Companies – Industry Outlook, Market Assessment, Competition Scenario, Trends and Forecast 2025–2034
- Published date: Nov 2025
- Report ID: 167792
- Number of Pages: 237
- Format:
-
keyboard_arrow_up
Quick Navigation
Report Overview
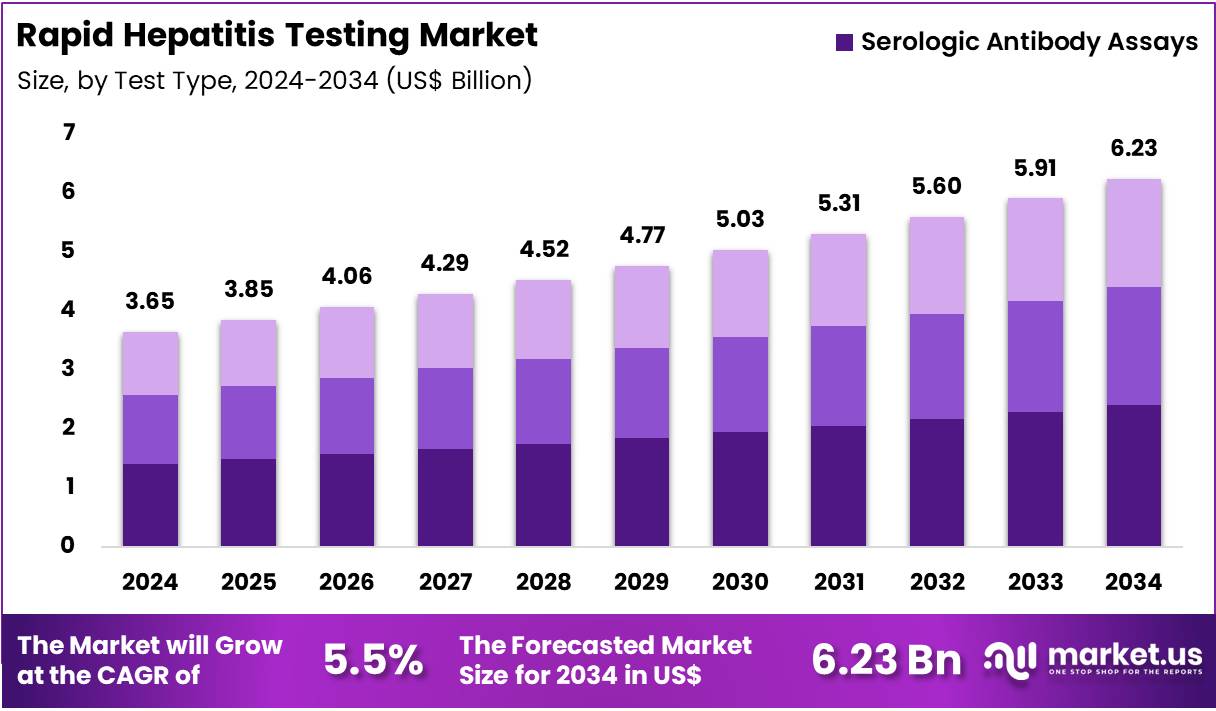
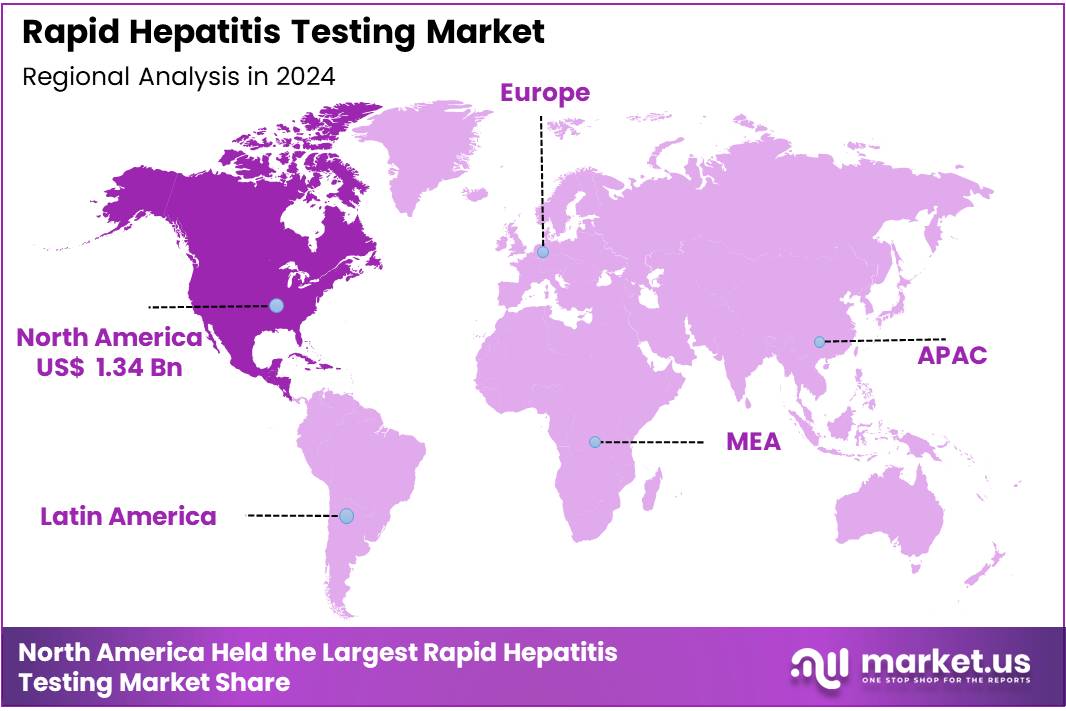
Global Rapid Hepatitis Testing Market size is expected to be worth around US$ 6.23 Billion by 2034 from US$ 3.65 Billion in 2024, growing at a CAGR of 5.5% during the forecast period 2025 to 2034. In 2024, North America led the market, achieving over 36.7% share with a revenue of US$ 1.34 Billion.
The Rapid Hepatitis Testing Market has become an essential component of infectious-disease screening frameworks worldwide, supporting early detection of hepatitis A, B, and C infections through fast, reliable, and increasingly decentralized diagnostic tools.
The market benefits from strong public-health initiatives encouraging early detection to prevent liver complications, community transmission, and late-diagnosis-related healthcare burdens. National immunization programs and hepatitis elimination targets set by bodies such as the World Health Organization continue to accelerate the adoption of rapid tests in low-resource and high-prevalence settings.
Rapid hepatitis test kits typically use finger-prick blood, serum, or oral fluid samples, providing point-of-care results within minutes. These tests deliver significant value in rural areas, outreach clinics, emergency departments, prenatal screening centers, blood-banking facilities, and harm-reduction programs. Many brands pair rapid screening with confirmatory molecular tests under standardized testing algorithms recommended by CDC, ECDC, and national disease-control programs.
As simplified diagnostics become more accessible through pharmacies and online distribution channels, rapid hepatitis testing increasingly supports routine screening among at-risk groups such as pregnant individuals, healthcare workers, travelers, dialysis patients, and people undergoing pre-surgical assessments.
Technological enhancements in immunoassay sensitivity, RNA amplification formats, and antigen-detection chemistry continue to improve accuracy and operational efficiency. Governments globally are strengthening hepatitis elimination roadmaps for 2030, increasing screening budgets, and expanding vaccination and linkage-to-care services. These collective efforts reinforce the demand for rapid, low-cost, and portable hepatitis diagnostic kits across both developed and emerging healthcare systems.
In 2024, the market expanded due to increased viral-hepatitis screening mandates and broader adoption of point-of-care diagnostic platforms. Serologic Antibody Assays accounted for the largest share within test types. Hepatitis B dominated the indication segment owing to its high global prevalence. Hospital Pharmacies represented the primary distribution channel due to clinic-based screening and high patient throughput.
Key Takeaways
- In 2024, the market generated a revenue of US$ 65 Billion, with a CAGR of 5.5%, and is expected to reach US$ 6.23 Billion by the year 2034.
- The Test Type segment is divided into Serologic Antibody Assays, Molecular HCV RNA Tests, and Immunoassays for HCV Core Antigen, with Serologic Antibody Assays taking the lead in 2024 with a market share of 38.6%
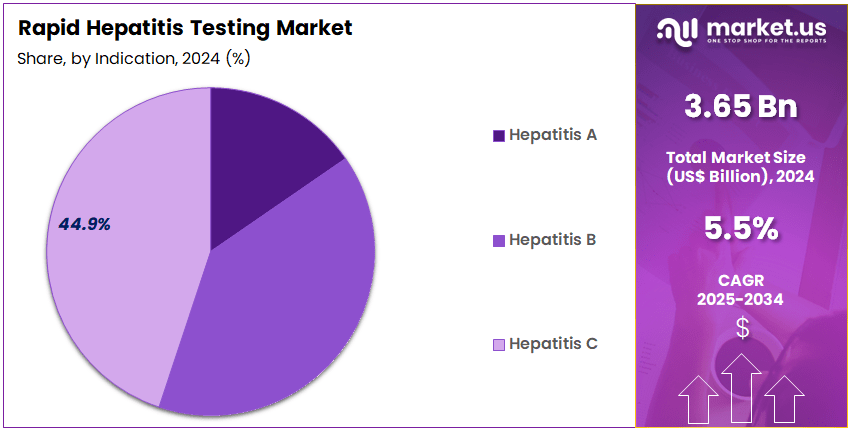
- The Indication segment is divided into Hepatitis A, Hepatitis B, and Hepatitis C, with Hepatitis C taking the lead in 2024 with a market share of 44.9%
- The Distribution Channel segment is divided into Infectious diseases, Sexually transmitted infections, and Others, with Infectious diseases taking the lead in 2024 with a market share of 79.5%.
- The End-User segment is bifurcated into Hospital Pharmacies, Retail Pharmacies, and Online Pharmacies, with Hospital Pharmacies taking the lead in 2024 with a market share of 55.2%
- North America led the market by securing a market share of 36.7% in 2024.
Test Type Analysis
Serologic Antibody Assays account for the largest share of the Rapid Hepatitis Testing Market due to their widespread use in initial screening for hepatitis A, B, and C infections. These tests detect viral-specific IgM or total antibodies and are recommended across public-health programs for early assessment of acute or past exposure.
Antibody-based rapid tests are commonly deployed in mass-screening campaigns, travel-health centers, blood-donation services, and emergency departments where immediate triage is essential. The simplicity of the assay format, minimal training requirements, and low per-test cost drive high adoption in low-resource regions where laboratory infrastructure remains limited.
These kits are frequently used in maternal-health clinics for routine antenatal hepatitis B screening, in line with WHO’s recommendation for universal HBV testing among pregnant individuals. Enhanced sensitivity improvements over the last decade have strengthened their reliability for frontline use, making them the first-line test of choice for large-scale hepatitis elimination initiatives.
Indication Analysis
Hepatitis C remains a key indication accounting for 44.9% market share in 2024 due to rising diagnosis efforts driven by the availability of highly effective antiviral therapies that offer high cure rates. Rapid anti-HCV antibody screening helps identify previously undiagnosed infections, especially among older adults, individuals with historic transfusion exposure, and high-risk populations.
Governments promoting HCV elimination targets integrate rapid tests into mobile clinics and community-testing events. The presence of simplified screening algorithms that include antibody testing followed by RNA or core-antigen confirmation accelerates linkage to treatment and contributes significantly to reducing disease burden. Increasing awareness and global commitments to HCV eradication continue to drive this segment forward.
WHO estimates reveal millions of chronic HBV cases globally, making early detection crucial to prevent liver cirrhosis and hepatocellular carcinoma. Universal birth-dose vaccination programs and antenatal HBV screening policies significantly increase the volume of rapid HBsAg testing in hospitals and primary-care centers.
Distribution Channel Analysis
Hospital Pharmacies dominated the distribution landscape with 55.2% market share due to their central role in supplying rapid hepatitis tests for inpatients, emergency cases, surgical candidates, antenatal patients, and screening groups identified through physician referrals. Hospitals frequently administer rapid tests during pre-operative workups and high-risk exposures, such as accidental needle-stick injuries among healthcare workers.
National hepatitis control programs also rely on hospital-based procurement for large-scale immunization and screening campaigns. The presence of trained personnel, laboratory support, and established care-continuum pathways reinforces hospital pharmacies as the primary channel.
Key Market Segments
By Test Type
- Serologic Antibody Assays
- Molecular HCV RNA Tests
- Immunoassays for HCV Core Antigen
By Indication
- Hepatitis A
- Hepatitis B
- Hepatitis C
By Distribution Channel
- Hospital Pharmacies
- Retail Pharmacies
- Online Pharmacies
Drivers
Expansion of decentralized and point-of-care diagnostic programs
The expansion of decentralized and point-of-care diagnostic programs is expected to strengthen the Rapid Hepatitis Testing Market as global health agencies continue shifting screening from laboratory-centric systems to community-based models. According to the World Health Organization, nearly 296 million people live with chronic hepatitis B and 58 million with chronic hepatitis C, yet more than 80 percent remain undiagnosed.
This diagnostic gap has pushed governments and NGOs to deploy rapid testing units in rural clinics, mobile health vans, prisons, antenatal care centers, and harm-reduction facilities. For example, India’s National Viral Hepatitis Control Program established district-level rapid HBV and HCV screening hubs, facilitating millions of point-of-care tests annually. Similarly, African hepatitis-elimination programs funded by global donors use rapid antibody and antigen tests where laboratory PCR machines are unavailable.
The success of HIV POC testing has also accelerated cross-adoption for hepatitis screening, with integrated community campaigns able to test over 5,000 individuals per day in certain regions. These initiatives reduce turnaround time from several days to under 30 minutes, enabling faster linkage to confirmatory testing and treatment.
Restraints
Variability in sensitivity of certain rapid antibody assays
Variability in the sensitivity of rapid antibody assays presents a significant restraint for the Rapid Hepatitis Testing Market, particularly in the detection of early-stage infections. Studies published through the US CDC and WHO indicate that some rapid HBsAg and anti-HCV antibody tests show sensitivity ranging from 85 to 98 percent depending on the manufacturer, sample type, and environmental factors. This variation can result in false negatives, especially during the window period when antibody levels remain low.
For instance, early acute hepatitis C infections are frequently missed by antibody-only tests because seroconversion can take 6 to 9 weeks. This limitation leads clinicians to prefer RNA or core-antigen confirmation, which increases overall testing costs and logistic burden in low-resource settings.
Temperature instability is another concern; many lateral-flow kits lose performance when exposed to heat above 30°C, a common challenge in rural clinics across Africa and Southeast Asia. The lack of universal quality-control frameworks across manufacturers further contributes to inconsistent analytical performance.
Opportunities
Development of integrated rapid test platforms for multiple hepatitis types
The development of integrated rapid test platforms capable of detecting multiple hepatitis types presents a strong opportunity for market expansion, especially as countries aim to streamline viral hepatitis screening. Multiplex rapid assays that combine HAV IgM, HBsAg, anti-HBc, and anti-HCV into a single cassette allow clinicians to screen large populations quickly and cost-effectively.
Global health agencies increasingly prefer multiplex tools to optimize budgets, with evidence showing that combined testing reduces per-patient diagnostic costs by up to 30 percent in mass-screening programs.
For example, field evaluations in Southeast Asia demonstrated that multiplex HBV/HCV rapid kits significantly increased screening uptake among pregnant women by reducing the number of clinic visits. Integrated platforms are particularly valuable in emergency settings where jaundice outbreaks require immediate differentiation between HAV, HBV, and other causes.
Manufacturers are also developing modular POC systems that pair rapid immunoassays with automated confirmatory antigen or RNA cartridges, enabling same-day diagnosis in resource-constrained environments. As WHO encourages countries to adopt simplified testing algorithms for meeting 2030 elimination targets, multiplex rapid tests can become central to national screening strategies.
Impact of Macroeconomic / Geopolitical Factors
Macroeconomic and geopolitical factors exert a significant influence on the Rapid Hepatitis Testing Market by shaping affordability, supply-chain continuity, screening volumes, and government health-budget allocations. During periods of economic slowdown or inflation, national healthcare spending often shifts toward essential and emergency services, affecting procurement of rapid diagnostic kits.
Rising costs of raw materials such as nitrocellulose membranes, monoclonal antibodies, and assay reagents can increase manufacturing expenses, pushing prices higher for low-income countries that depend heavily on bulk purchases for public-health screening.
Geopolitical disruptions also affect global trade flows; for example, tensions impacting shipping routes in Asia and the Middle East have previously led to delayed deliveries of diagnostic components, slowing testing programs in regions with limited inventory buffers. Countries relying on imported reagents face greater vulnerability when export controls restrict movement of medical supplies, as seen during pandemic-related border tightening where hepatitis screening declined by over 30 percent in some regions.
Migration surges driven by political instability or conflict increase demand for rapid hepatitis screening in refugee camps and border-health facilities, where overcrowded conditions elevate transmission risks. At the same time, shifts in foreign-aid priorities can alter funding for hepatitis elimination initiatives, particularly in Africa and South Asia, where donor-backed programs play a major role.
Latest Trends
Adoption of low-cost HCV core-antigen rapid assays in resource-limited settings
A growing trend in the Rapid Hepatitis Testing Market is the adoption of low-cost HCV core-antigen rapid assays, especially in low- and middle-income countries where molecular RNA testing remains unaffordable. Traditional PCR-based HCV viral-load testing costs between USD 30 and USD 100 per test, while emerging core-antigen assays often cost less than USD 10, making them significantly more accessible.
WHO has recognized HCV core-antigen testing as a viable alternative to RNA testing for confirming active infection, particularly in decentralized clinics. Countries such as Egypt and Rwanda have already integrated core-antigen diagnostics into national hepatitis-elimination pathways, enabling faster diagnosis among high-risk groups.
Field studies show that core-antigen tests can detect more than 90% of active infections, narrowing the gap between rapid antibody screening and RNA confirmation while keeping costs manageable. Mobile clinics and outreach programs increasingly use core-antigen kits to test incarcerated individuals, injection-drug users, and migrant communities, where laboratory infrastructure is limited. As manufacturers continue improving sensitivity and stability under high-temperature conditions, adoption is accelerating across Africa, South Asia, and Latin America.
Regional Analysis
North America is leading the Rapid Hepatitis Testing Market
North America represents the largest share of the Rapid Hepatitis Testing Market due to its strong public-health infrastructure, advanced diagnostic networks, and high annual testing volumes across hospitals, emergency units, blood banks, and community clinics. The region conducts extensive screening for hepatitis B and C as part of routine prenatal care, pre-operative evaluations, and harm-reduction initiatives.
For example, the US CDC recommends universal hepatitis B screening for pregnant individuals and once-in-a-lifetime hepatitis C screening for all adults, significantly increasing test demand. Large-scale testing in correctional facilities and opioid-affected communities further elevates usage of rapid antibody and antigen kits. Additionally, North America benefits from strong adoption of point-of-care diagnostics, with integrated screening programs in Native American health centers, rural clinics, and migrant health networks.
The Asia Pacific region is expected to experience the highest CAGR during the forecast period
Asia Pacific stands as the fastest-growing region due to the high prevalence of hepatitis B and C, expanding government-led elimination programs, and rapid uptake of affordable point-of-care diagnostics. Countries such as India, China, Pakistan, Vietnam, and Indonesia collectively account for a large share of the global hepatitis burden, prompting aggressive national screening efforts.
For example, India’s National Viral Hepatitis Control Program deploys rapid HBsAg and anti-HCV tests through district hospitals and outreach camps, while China expands community-level screening to support HBV vaccination and early diagnosis targets. Growing urbanization has increased demand for quick diagnostics in high-density settings where infection risks are elevated.
Key Regions and Countries
North America
- US
- Canada
Europe
- Germany
- France
- The UK
- Spain
- Italy
- Russia
- Netherland
- Rest of Europe
Asia Pacific
- China
- Japan
- South Korea
- India
- Australia
- New Zealand
- Singapore
- Thailand
- Vietnam
- Rest of APAC
Latin America
- Brazil
- Mexico
- Rest of Latin America
Middle East & Africa
- South Africa
- Saudi Arabia
- UAE
- Rest of MEA
Key Players Analysis
Key players in the market include Abbott Laboratories, Roche Diagnostics, bioMérieux, Siemens Healthineers, Bio-Rad Laboratories, Qiagen, Cepheid (Danaher), SD Biosensor, OraSure Technologies, QuidelOrtho Corporation, Chembio Diagnostics, Fujirebio, and Others.
Abbott offers widely adopted rapid HBsAg and anti-HCV test kits through its lateral-flow platforms, enabling quick point-of-care diagnosis. Its solutions support large screening programs, blood-bank testing, and decentralized care, especially in low-resource and high-burden regions. Roche strengthens rapid hepatitis detection through simplified molecular and antigen-based solutions aligned with global elimination goals.
Its near-patient RNA platforms accelerate hepatitis C confirmation, improving linkage-to-care in community clinics, outreach programs, and national screening initiatives. bioMérieux provides rapid immunoassays and diagnostic systems supporting early hepatitis A, B, and C detection. Its assays are used in hospitals, emergency care, and public-health campaigns, emphasizing reliability and accessibility in regions expanding point-of-care viral-hepatitis screening.
Top Key Players
- Abbott Laboratories
- Roche Diagnostics
- bioMérieux
- Siemens Healthineers
- Bio-Rad Laboratories
- Qiagen
- Cepheid (Danaher)
- SD Biosensor
- OraSure Technologies
- QuidelOrtho Corporation
- Chembio Diagnostics
- Fujirebio
- Others
Recent Developments
- In October 2025, Reszon Diagnostics announced the commercial launch of a rapid diagnostic test for HBsAg (hepatitis B surface antigen) for screening in serum, plasma or whole-blood. The launch complements its earlier RESZON HCV Rapid Test introduced in September 2025. The press release emphasises that 254 million people live with chronic HBV globally and only 13% are diagnosed.
- In June 2024, the US FDA permitted marketing of the Xpert HCV test and GeneXpert Xpress System for HCV RNA detection in CLIA-waived settings (e.g., urgent care, correctional), producing results in about an hour from a finger-stick blood sample. The release emphasises linking testing and treatment in one visit.
- In July 2024, WHO prequalified the OraQuick HCV self-test, extending the professional-use version. This enables individuals at home or in non-clinical settings to screen for hepatitis C self-care, expanding access especially in underserved populations.
Report Scope
Report Features Description Market Value (2024) US$ 3.65 Billion Forecast Revenue (2034) US$ 6.23 Billion CAGR (2025-2034) 5.5% Base Year for Estimation 2024 Historic Period 2020-2023 Forecast Period 2025-2034 Report Coverage Revenue Forecast, Market Dynamics, COVID-19 Impact, Competitive Landscape, Recent Developments Segments Covered By Test Type (Serologic Antibody Assays, Molecular HCV RNA Tests, Immunoassays for HCV Core Antigen), By Indication (Hepatitis A, Hepatitis B, Hepatitis C), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, and Online Pharmacies) Regional Analysis North America – US, Canada; Europe – Germany, France, The UK, Spain, Italy, Russia, Netherlands, Rest of Europe; Asia Pacific – China, Japan, South Korea, India, Australia, New Zealand, Singapore, Thailand, Vietnam, Rest of APAC; Latin America – Brazil, Mexico, Rest of Latin America; Middle East & Africa – South Africa, Saudi Arabia, UAE, Rest of MEA Competitive Landscape Abbott Laboratories, Roche Diagnostics, bioMérieux, Siemens Healthineers, Bio-Rad Laboratories, Qiagen, Cepheid (Danaher), SD Biosensor, OraSure Technologies, QuidelOrtho Corporation, Chembio Diagnostics, Fujirebio, and Others Customization Scope Customization for segments, region/country-level will be provided. Moreover, additional customization can be done based on the requirements. Purchase Options We have three licenses to opt for: Single User License, Multi-User License (Up to 5 Users), Corporate Use License (Unlimited User and Printable PDF)  Rapid Hepatitis Testing MarketPublished date: Nov 2025add_shopping_cartBuy Now get_appDownload Sample
Rapid Hepatitis Testing MarketPublished date: Nov 2025add_shopping_cartBuy Now get_appDownload Sample -
-
- Abbott Laboratories
- Roche Diagnostics
- bioMérieux
- Siemens Healthineers
- Bio-Rad Laboratories
- Qiagen
- Cepheid (Danaher)
- SD Biosensor
- OraSure Technologies
- QuidelOrtho Corporation
- Chembio Diagnostics
- Fujirebio
- Others










