Global Western Blotting Market By Product Type (Reagent Kits, Electrophoresis Blotting Systems and Transfer Membranes), By Application (Scientific Research, Medical Diagnostics, Food & Beverages, Agricultural Application and Others), By End User (Pharmaceutical & Biotechnology Companies, Diagnostic Laboratories and Research Institutions), Region and Companies – Industry Segment Outlook, Market Assessment, Competition Scenario, Trends and Forecast 2026-2035
- Published date: Feb 2026
- Report ID: 177804
- Number of Pages: 224
- Format:
-
keyboard_arrow_up
Quick Navigation
Report Overview
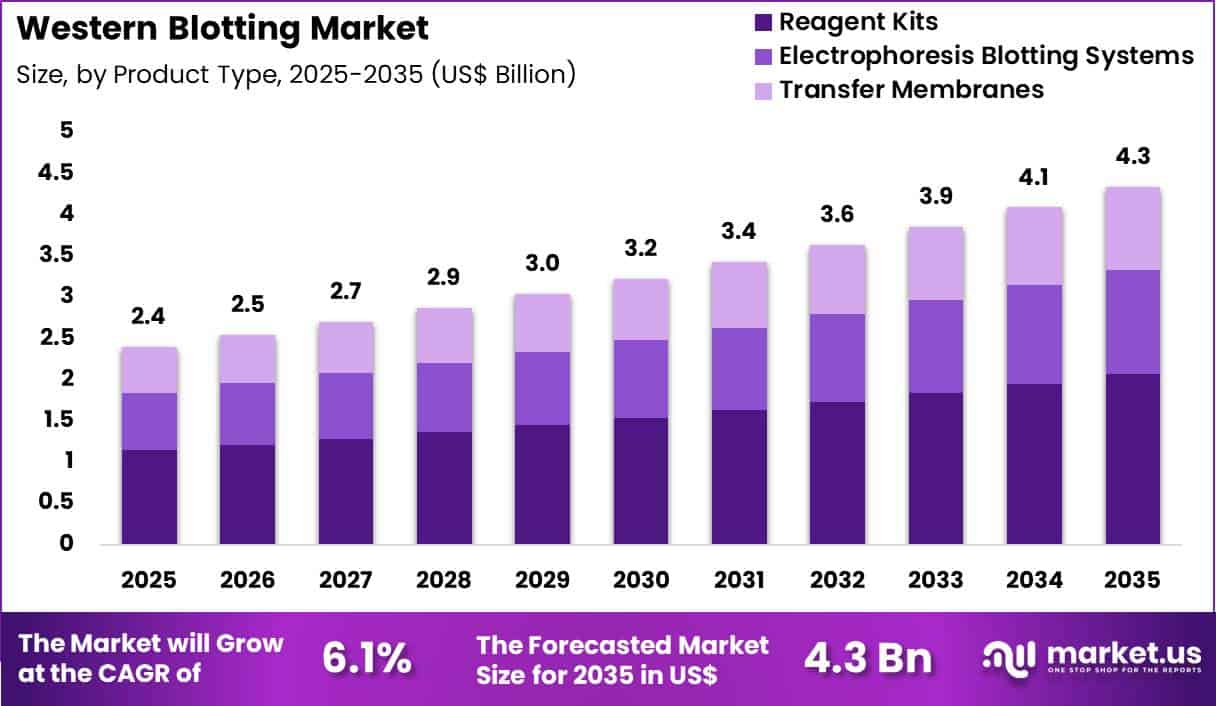
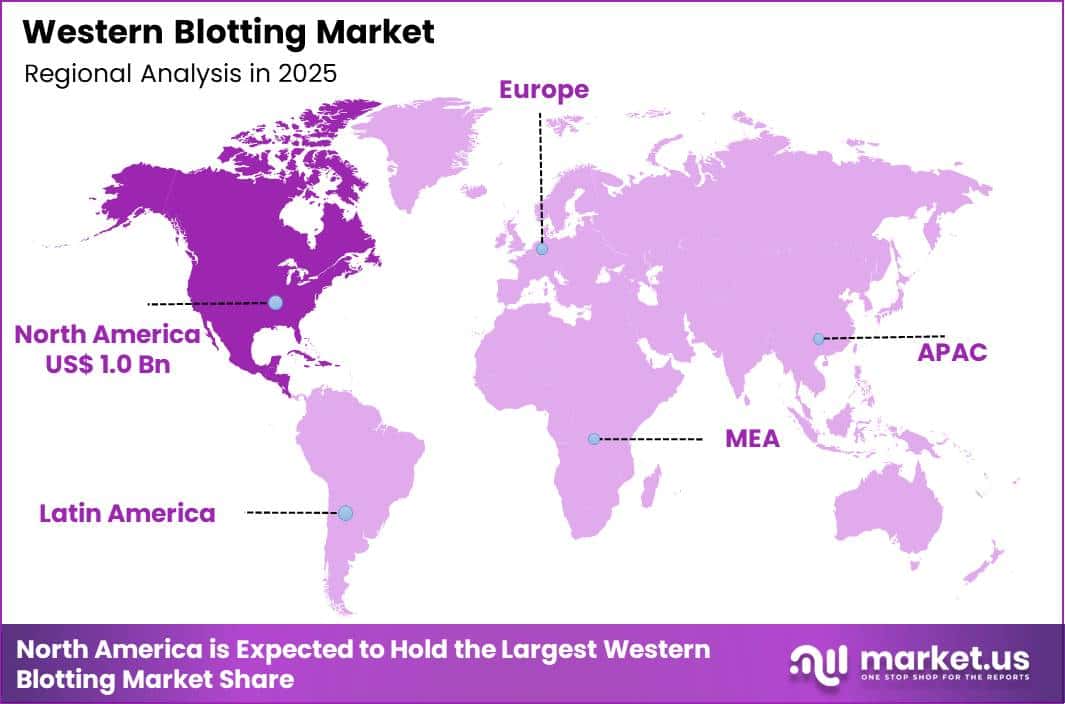
Global Western Blotting Market size is expected to be worth around US$ 4.3 Billion by 2035 from US$ 2.4 Billion in 2025, growing at a CAGR of 6.1% during the forecast period 2026-2035. In 2025, North America led the market, achieving over 42.4% share with a revenue of US$ 1.0 Billion.
Growing demand for precise protein detection and quantification in life sciences research propels the Western blotting market as laboratories require reliable methods to validate protein expression, post-translational modifications, and molecular interactions.
Researchers increasingly apply Western blotting in cancer biology to confirm oncoprotein levels and phosphorylation states in cell lines treated with targeted inhibitors, guiding mechanistic studies and drug efficacy assessments. These techniques support immunology investigations by detecting cytokine production and signaling pathway activation in immune cell populations, informing vaccine development and autoimmune disease models.
Neuroscientists utilize Western blotting to quantify neurodegenerative markers such as tau and amyloid-beta aggregates in brain tissue lysates, advancing understanding of Alzheimer’s and Parkinson’s pathology. Virologists employ the method to verify viral protein expression and host response factors during infection studies, facilitating antiviral compound screening.
Pharmaceutical developers rely on Western blotting for quality control of recombinant proteins, ensuring correct molecular weight and purity in biologics manufacturing pipelines. Manufacturers pursue opportunities to develop automated Western blotting systems that streamline gel electrophoresis, transfer, and detection steps, expanding applications in high-throughput screening for biomarker validation and antibody specificity testing.
Developers advance chemiluminescent and fluorescent detection reagents with enhanced sensitivity, broadening utility in low-abundance protein analysis across proteomics workflows. These innovations facilitate multiplexed Western blotting that simultaneously probes multiple targets, accelerating pathway mapping in signal transduction research.
Opportunities emerge in digital imaging platforms with quantitative software that improve reproducibility and data integrity for regulatory submissions. Companies invest in capillary-based Western systems that eliminate manual gel handling, reducing variability in clinical research settings.
Recent trends emphasize integration with mass spectrometry for orthogonal validation and AI-assisted band quantification, positioning Western blotting as an indispensable tool in translational research and biopharmaceutical development.
Key Takeaways
- In 2025, the market generated a revenue of US$ 4 billion, with a CAGR of 6.1%, and is expected to reach US$ 4.3 billion by the year 2035.
- The product type segment is divided into reagent kits, electrophoresis blotting systems and transfer membranes, with reagent kits taking the lead with a market share of 47.6%.
- Considering application, the market is divided into scientific research, medical diagnostics, food & beverages, agricultural application and others. Among these, scientific research held a significant share of 52.4%.
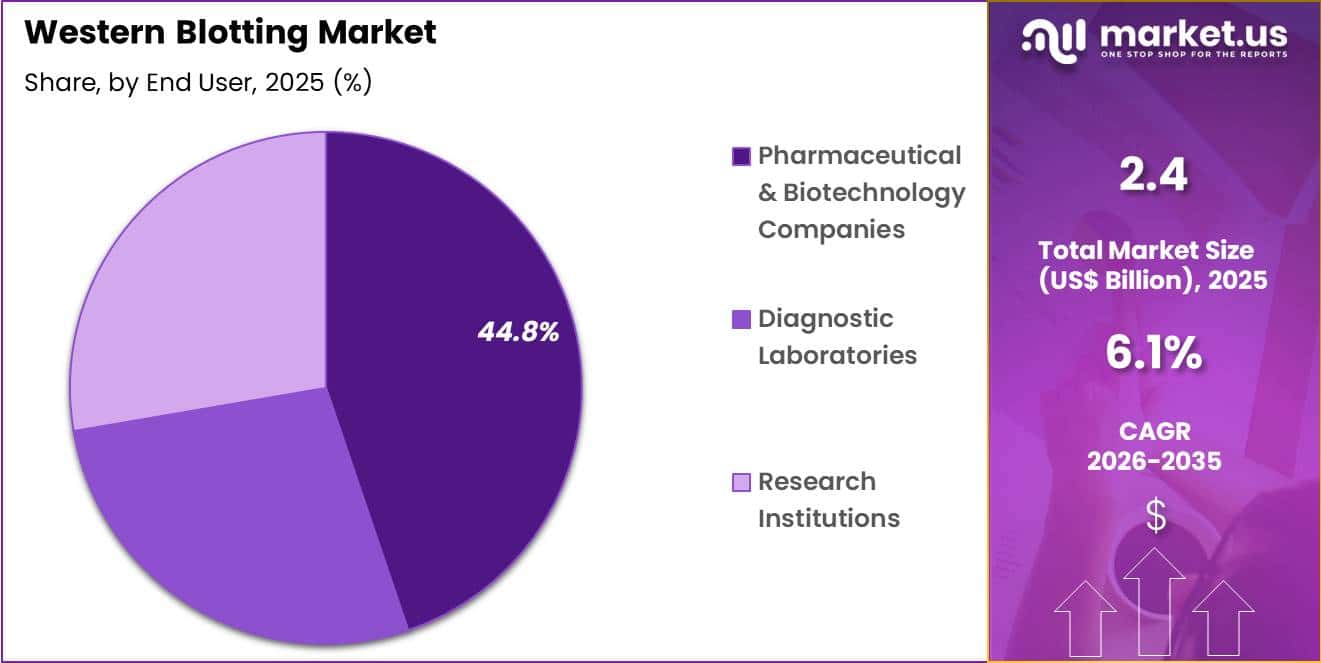
- Furthermore, concerning the end user segment, the market is segregated into pharmaceutical & biotechnology companies, diagnostic laboratories and research institutions. The pharmaceutical & biotechnology companies sector stands out as the dominant player, holding the largest revenue share of 44.8% in the market.
- North America led the market by securing a market share of 42.4%.
Product Type Analysis
Reagent kits contributed 47.6% of growth within product type and led the western blotting market due to their recurring consumption and essential role in protein detection workflows. Laboratories depend on antibodies, buffers, substrates, and detection reagents for every blotting experiment, which creates continuous demand.
Researchers favor ready-to-use kits because they improve reproducibility and reduce preparation errors. Expanding proteomics and biomarker validation studies increase assay volumes across academic and commercial settings.
Growth strengthens as manufacturers introduce enhanced chemiluminescent and fluorescent detection chemistries that improve sensitivity. Standardized kit formats support multi-site studies and regulated environments.
Pharmaceutical pipelines focused on protein-based therapeutics further increase validation testing. Funding for molecular biology research sustains procurement cycles. The segment is expected to remain dominant as protein expression analysis continues to serve as a core laboratory technique.
Application Analysis
Scientific research generated 52.4% of growth within application and emerged as the leading segment due to the technique’s foundational role in molecular and cellular biology. Western blotting supports protein identification, quantification, and post-translational modification analysis across diverse study areas.
Universities and research institutes rely on this method for pathway validation and experimental confirmation. Increasing exploration of disease mechanisms elevates demand for protein-level analysis tools.
Growth accelerates as life science research funding expands in genomics and proteomics integration. Multi-omics studies require protein validation to complement sequencing data. Training curricula reinforce western blotting as a standard laboratory skill. Publication-driven research culture sustains consistent usage. The segment is anticipated to maintain leadership as scientific discovery continues to depend on reliable protein characterization techniques.
End-User Analysis
Pharmaceutical and biotechnology companies accounted for 44.8% of growth within end user and dominated the western blotting market due to their extensive involvement in drug discovery and development.
These organizations use western blotting to validate targets, confirm protein expression, and assess therapeutic effects. Preclinical and clinical development programs require repeated protein analysis, which increases reagent consumption. Quality control processes further reinforce routine application.
Growth continues as biologics and targeted therapies expand across pipelines. Regulatory documentation often requires molecular validation data, which supports technique adoption. Internal research teams prioritize reliable and well-established assays to reduce development risk. Strategic investment in R&D infrastructure increases equipment and reagent demand. The segment is projected to remain the primary growth driver as pharmaceutical innovation continues to rely on protein-level validation.
Key Market Segments
By Product Type
- Reagent Kits
- Electrophoresis Blotting Systems
- Transfer Membranes
By Application
- Scientific Research
- Medical Diagnostics
- Food & Beverages
- Agricultural Application
- Others
By End User
- Pharmaceutical & Biotechnology Companies
- Diagnostic Laboratories
- Research Institutions
Drivers
Increasing government funding for biomedical research is driving the market.
Government allocations to biomedical research have substantially expanded the infrastructure for protein analysis techniques, including western blotting, in academic and institutional laboratories worldwide. This funding supports the procurement of consumables and instruments essential for validating protein expression in diverse experimental models.
The National Institutes of Health (NIH) plays a pivotal role in financing projects that rely on western blotting for mechanistic studies in disease pathways. In fiscal year 2024, the NIH budget totaled $47.4 billion, enabling thousands of grants that incorporate protein detection methodologies.
Such investments facilitate high-throughput screening and validation in oncology, neuroscience, and infectious disease research. Western blotting remains a gold standard for confirming protein size and abundance, benefiting from this financial backing. International agencies mirror this support, broadening the technique’s application in global collaborative initiatives.
The sustained funding cycle ensures continuous demand for reliable western blotting reagents and equipment. This driver reinforces the technique’s position in fundamental and translational science. Overall, enhanced budgetary commitments propel advancements in laboratory capabilities dependent on western blotting.
Restraints
Time-consuming manual procedures are restraining the market.
The labor-intensive steps involved in traditional western blotting protocols, from gel electrophoresis to detection, deter high-throughput applications in time-sensitive research environments. Manual handling increases the risk of variability and errors, affecting reproducibility in experimental results. Laboratories with limited personnel find it challenging to scale western blotting for large cohort studies.
The multi-day workflow contrasts with faster alternatives like ELISA or mass spectrometry for certain analytes. Optimization of transfer and blocking conditions demands expertise, complicating standardization across users. This restraint encourages shift toward automated platforms, reducing reliance on conventional methods.
Quality control for antibodies and membranes adds to procedural delays. Despite accuracy, the technique’s demands limit its use in rapid diagnostic development. Industry responses include semi-automated systems, but full transition remains gradual. Consequently, operational inefficiencies hinder broader integration in modern workflows.
Opportunities
Rising pharmaceutical R&D investments are creating growth opportunities.
The escalation in pharmaceutical research and development expenditures opens pathways for western blotting in drug target validation and biomarker discovery. Increased funding for biologics and personalized medicine amplifies the need for precise protein quantification tools in preclinical stages. Collaborations between pharma entities and academic labs facilitate technology transfer for advanced blotting applications.
Regional incentives for biotech innovation support the deployment of western blotting in emerging therapeutic pipelines. The focus on orphan drugs and rare diseases heightens demand for specialized protein analysis in small-scale studies.
Training initiatives for R&D staff promote proficient use of blotting techniques in compound screening. This opportunity enables suppliers to offer customized solutions for high-value pharmaceutical projects. Key firms are aligning product development with pharma needs for reproducible protein data.
Overall, R&D momentum aligns with precision medicine goals requiring robust analytical methods. Strategic partnerships can accelerate market penetration in lucrative drug development sectors.
Impact of Macroeconomic / Geopolitical Factors
Global economic shifts affect the Western blotting market by shaping grant availability, institutional budgets, and corporate research priorities. When inflation rises and borrowing costs remain high, universities and smaller biotech firms delay capital purchases and limit spending on premium reagents and imaging upgrades.
Geopolitical friction interrupts cross border trade in antibodies, nitrocellulose membranes, buffers, and detection substrates, which creates supply gaps and pricing fluctuations. Current US tariffs on selected laboratory instruments and imported consumables increase acquisition costs for distributors and end users, which narrows profit margins and forces tighter procurement reviews.
These pressures slow expansion plans and strain research timelines in cost conscious environments. At the same time, tariff exposure motivates regional manufacturing, contract production partnerships, and broader supplier qualification strategies.
Ongoing demand for protein validation in translational research and biologics development continues to anchor usage levels. With operational discipline and stronger supply resilience, the market can navigate volatility and maintain forward momentum.
Latest Trends
Adoption of compact benchtop imaging systems is a recent trend in the market.
In 2024, the introduction of compact imaging systems has streamlined western blot documentation by offering benchtop solutions with enhanced sensitivity and multitasking capabilities. These devices incorporate LED illumination and CMOS detection for chemiluminescent and fluorescent blots without darkroom requirements.
Manufacturers emphasize user-friendly software for image acquisition and analysis in space-constrained laboratories. Clinical research benefits from portable imagers that support GLP-compliant documentation. Bio-Rad launched the ChemiDoc Go Imaging System in 2024, a compact benchtop device for gel and blot imaging with multiple detection modes. This system facilitates high-resolution capture in standard lab settings.
The trend addresses needs for efficient workflows in academic and small-scale research facilities. Regulatory compliance features ensure traceability in validated environments. Industry collaborations optimize sensitivity for low-abundance proteins. These innovations reduce footprint while maintaining performance standards. This evolution enhances accessibility for diverse users in protein research.
Regional Analysis
North America is leading the Western Blotting Market
North America held a 42.4% share of the Western Blotting market in 2024, reflecting sustained demand from academic research, biopharma development, and clinical laboratories. Research institutions expanded protein expression and validation studies in oncology, immunology, and neuroscience, which increased routine use of blotting reagents and imaging systems.
Biotech companies relied on protein analysis to confirm target engagement and biomarker expression during preclinical and early clinical programs. Diagnostic laboratories continued to apply immunoblot assays for confirmatory testing in infectious and autoimmune conditions. Advanced imaging platforms and automation improved throughput and reproducibility across research centers.
Strong grant funding and established laboratory infrastructure further supported adoption. A clear indicator of the research intensity behind this growth comes from the National Science Foundation, which reported federal obligations for life sciences research exceeding USD 45 billion in fiscal year 2023, reinforcing continued investment in protein analysis workflows.
The Asia Pacific region is expected to experience the highest CAGR during the forecast period
The Western Blotting market in Asia Pacific is expected to expand steadily during the forecast period as regional research ecosystems strengthen and biotechnology pipelines mature. Governments increase support for molecular biology and translational medicine, which drives demand for reliable protein detection methods.
Universities and research institutes expand postgraduate training in proteomics and cell biology, widening the user base for laboratory reagents and systems. Growing pharmaceutical manufacturing and biosimilar development programs require robust validation of protein expression. Public health laboratories also enhance confirmatory testing capacity for infectious diseases.
Local suppliers improve reagent accessibility and reduce procurement timelines. A verifiable signal of rising research commitment appears in 2023 data from Japan’s Ministry of Education, Culture, Sports, Science and Technology, which allocated over JPY 4 trillion toward science and technology budgets, underscoring sustained governmental backing for advanced life science research across the region.
Key Regions and Countries
North America
- US
- Canada
Europe
- Germany
- France
- The UK
- Spain
- Italy
- Russia
- Netherland
- Rest of Europe
Asia Pacific
- China
- Japan
- South Korea
- India
- Australia
- New Zealand
- Singapore
- Thailand
- Vietnam
- Rest of APAC
Latin America
- Brazil
- Mexico
- Rest of Latin America
Middle East & Africa
- South Africa
- Saudi Arabia
- UAE
- Rest of MEA
Key Players Analysis
Key competitors in the Western blotting market pursue growth by enhancing throughput, sensitivity, and automation in protein separation and detection systems that help life science labs accelerate discovery workflows. They also broaden offerings with ready-to-use reagents, high-affinity antibodies, and integrated imaging platforms that reduce hands-on time and support reproducible results across diverse research applications.
Firms strengthen customer value by embedding digital analysis tools and cloud-enabled data management that improve interpretation and collaboration across research teams. Strategic alliances with academic institutions and biotech innovators deepen adoption in translational studies and pharmaceutical development programs.
Bio-Rad Laboratories Inc. exemplifies a diversified life science technology company with a robust portfolio spanning electrophoresis systems, western blot reagents, and imaging solutions, supported by extensive global distribution and technical support networks.
The company advances performance through disciplined R&D investment, targeted acquisitions that expand capabilities, and customer-centric commercialization that aligns evolving scientific needs with market-relevant innovations.
Top Key Players
- Bio-Rad Laboratories
- Thermo Fisher Scientific
- Merck KGaA
- GE HealthCare
- Danaher Corporation
- Abcam
- PerkinElmer
- LI-COR Biosciences
- Advansta
- Azure Biosystems
Recent Developments
- In January 2025, Roche Diagnostics outlined a strategic roadmap to introduce 75 new diagnostic assays by 2029. The planned pipeline includes assay formats relevant to protein detection techniques such as western blot–related applications, reinforcing the company’s commitment to expanding its diagnostics portfolio.
- In August 2024, Bio-Rad Laboratories launched the ChemiDoc Go Imaging System, designed for imaging gels and western blot membranes. The addition strengthens Bio-Rad’s life-science instrumentation offerings by providing researchers with a compact solution for protein analysis workflows.
- In July 2024, Thermo Fisher Scientific finalized its USD 3.1 billion acquisition of Olink Holding. The transaction expands Thermo Fisher’s life-science capabilities by incorporating more than 5,300 validated protein biomarkers into its proteomics and research solutions portfolio.
Report Scope
Report Features Description Market Value (2025) US$ 2.4 Billion Forecast Revenue (2035) US$ 4.3 Billion CAGR (2026-2035) 6.1% Base Year for Estimation 2025 Historic Period 2020-2024 Forecast Period 2026-2035 Report Coverage Revenue Forecast, Market Dynamics, COVID-19 Impact, Competitive Landscape, Recent Developments Segments Covered By Product Type (Reagent Kits, Electrophoresis Blotting Systems and Transfer Membranes), By Application (Scientific Research, Medical Diagnostics, Food & Beverages, Agricultural Application and Others), By End User (Pharmaceutical & Biotechnology Companies, Diagnostic Laboratories and Research Institutions) Regional Analysis North America – US, Canada; Europe – Germany, France, The UK, Spain, Italy, Russia, Netherlands, Rest of Europe; Asia Pacific – China, Japan, South Korea, India, Australia, New Zealand, Singapore, Thailand, Vietnam, Rest of APAC; Latin America – Brazil, Mexico, Rest of Latin America; Middle East & Africa – South Africa, Saudi Arabia, UAE, Rest of MEA Competitive Landscape Bio-Rad Laboratories, Thermo Fisher Scientific, Merck KGaA, GE HealthCare, Danaher Corporation, Abcam, PerkinElmer, LI-COR Biosciences, Advansta, Azure Biosystems Customization Scope Customization for segments, region/country-level will be provided. Moreover, additional customization can be done based on the requirements. Purchase Options We have three licenses to opt for: Single User License, Multi-User License (Up to 5 Users), Corporate Use License (Unlimited User and Printable PDF) 
-
-
- Bio-Rad Laboratories
- Thermo Fisher Scientific
- Merck KGaA
- GE HealthCare
- Danaher Corporation
- Abcam
- PerkinElmer
- LI-COR Biosciences
- Advansta
- Azure Biosystems










