Global MRI Contrast Agents Market By Type (Extracellular Agents, Blood Pool Agents, Organ-Specific Agents) By Application (Neurology, Oncology, Cardiology, Musculoskeletal, Gastroenterology, Others) By Route of Administration (Intravenous, Oral, Intrathecal) By End User (Hospitals, Diagnostic Imaging Centers, Ambulatory Surgical Centers, Others), By Region and Companies - Industry Segment Outlook, Market Assessment, Competition Scenario, Statistics, Trends and Forecast 2026-2035
- Published date: May 2026
- Report ID: 185633
- Number of Pages: 380
- Format:
-
keyboard_arrow_up
Quick Navigation
Report Overview
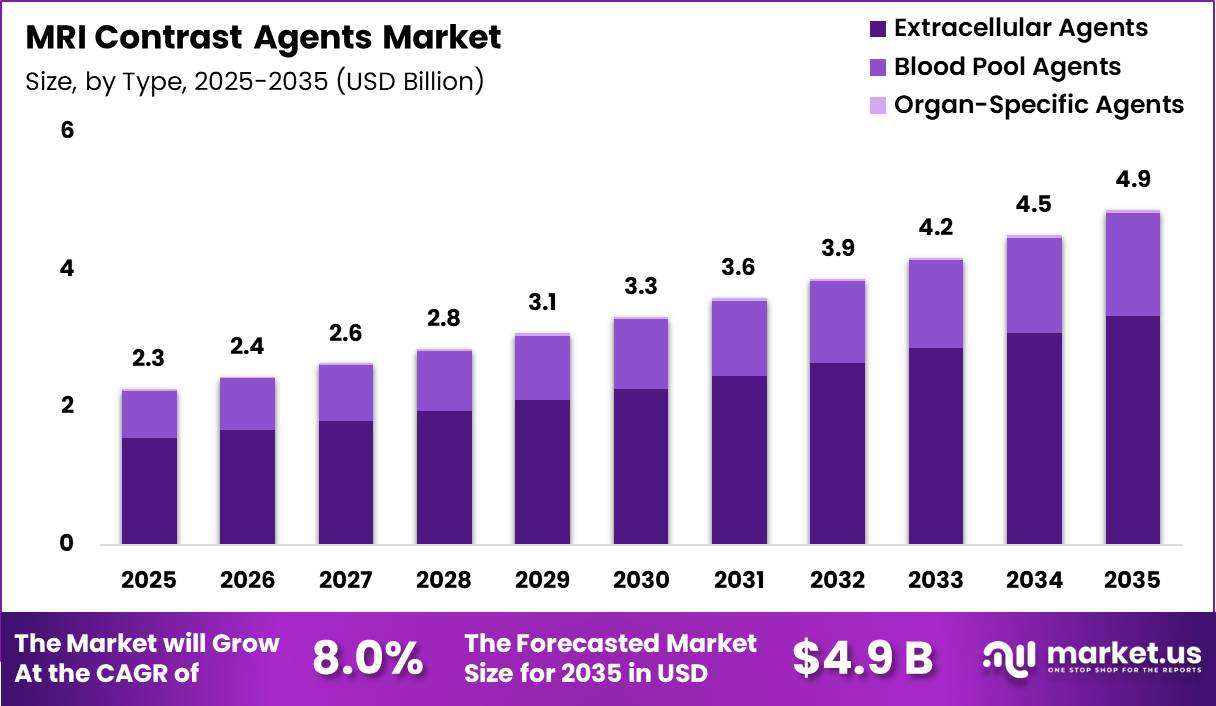
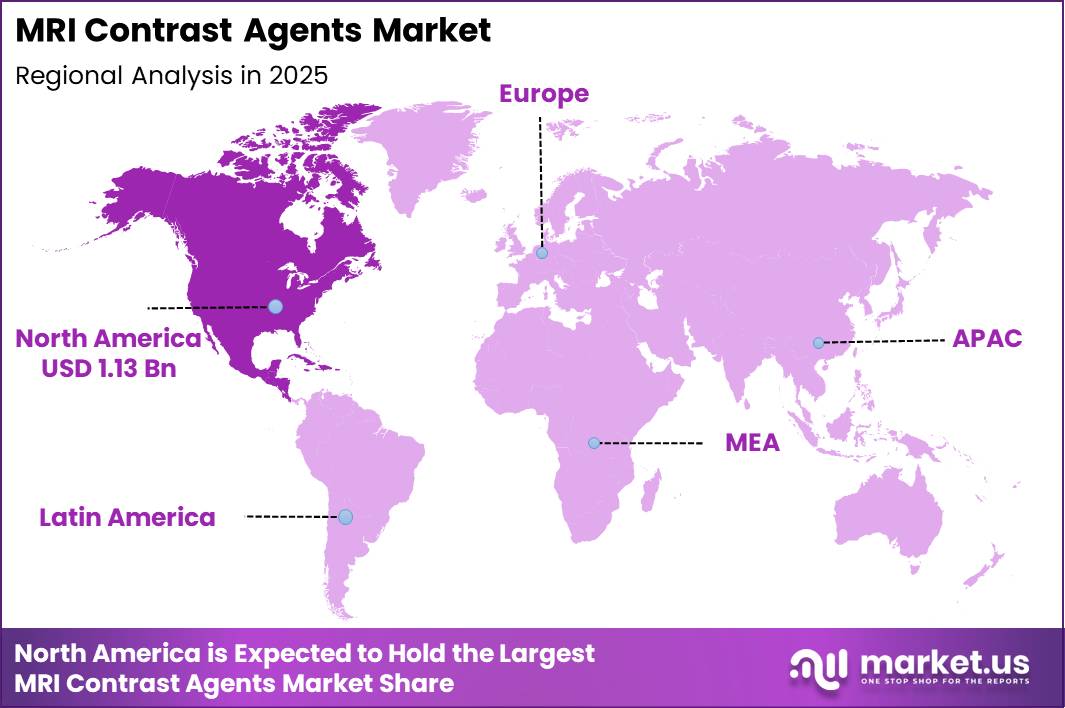
Global MRI Contrast Agents Market size is expected to be worth around US$ 4.9 Billion by 2035 from US$ 2.3 Billion in 2025, growing at a CAGR of 7.61% during the forecast period from 2026 to 2035. In 2025, North America led the market, achieving over 8.0% share with a revenue of US$ 1.13 Billion.
The MRI contrast agents market is demonstrating strong growth potential, driven by advancements in diagnostic imaging and increasing disease burden. These agents are widely utilized to enhance visualization of tumors, aneurysms, and tissue abnormalities, improving diagnostic accuracy in neurological, oncological, and cardiovascular conditions.
Continuous research and development efforts are focused on minimizing adverse reactions, reducing dosage levels, and enhancing imaging efficiency, thereby strengthening clinical adoption.
According to Johns Hopkins Medicine, nearly 30 million children and adolescents participate in sports annually, resulting in approximately 3.5 million sports-related injuries each year.
Additionally, data from Association for Safe International Road Travel indicates that 20–50 million individuals suffer injuries or disabilities due to road accidents globally. This rising incidence of trauma cases is significantly increasing the demand for advanced imaging techniques, including MRI with contrast agents.
Technological advancements, including the integration of artificial intelligence in MRI systems, have enhanced the detection of minute abnormalities, further increasing reliance on contrast-enhanced imaging. The growing preference for non-invasive diagnostic methods, combined with expanding healthcare infrastructure, particularly in emerging markets, is supporting market expansion.
Furthermore, the rising global prevalence of chronic diseases such as cancer and cardiovascular disorders—reported by World Health Organization to account for over 70% of deaths worldwide—has amplified the need for early and accurate diagnosis.
Ongoing innovation in safer agents, including macrocyclic gadolinium-based compounds, alongside increased healthcare expenditure and strategic collaborations, is expected to sustain market growth, despite regulatory scrutiny regarding safety standards.
Key Takeaways
- Market Size: Global MRI Contrast Agents Market size is expected to be worth around US$ 4.9 Billion by 2035 from US$ 2.3 Billion in 2025.
- Market Share: The market growing at a CAGR of 7.61% during the forecast period from 2026 to 2035.
- Type Analysis: extracellular agents are projected to dominate the market, accounting for approximately 68.4% of the total share in 2025.
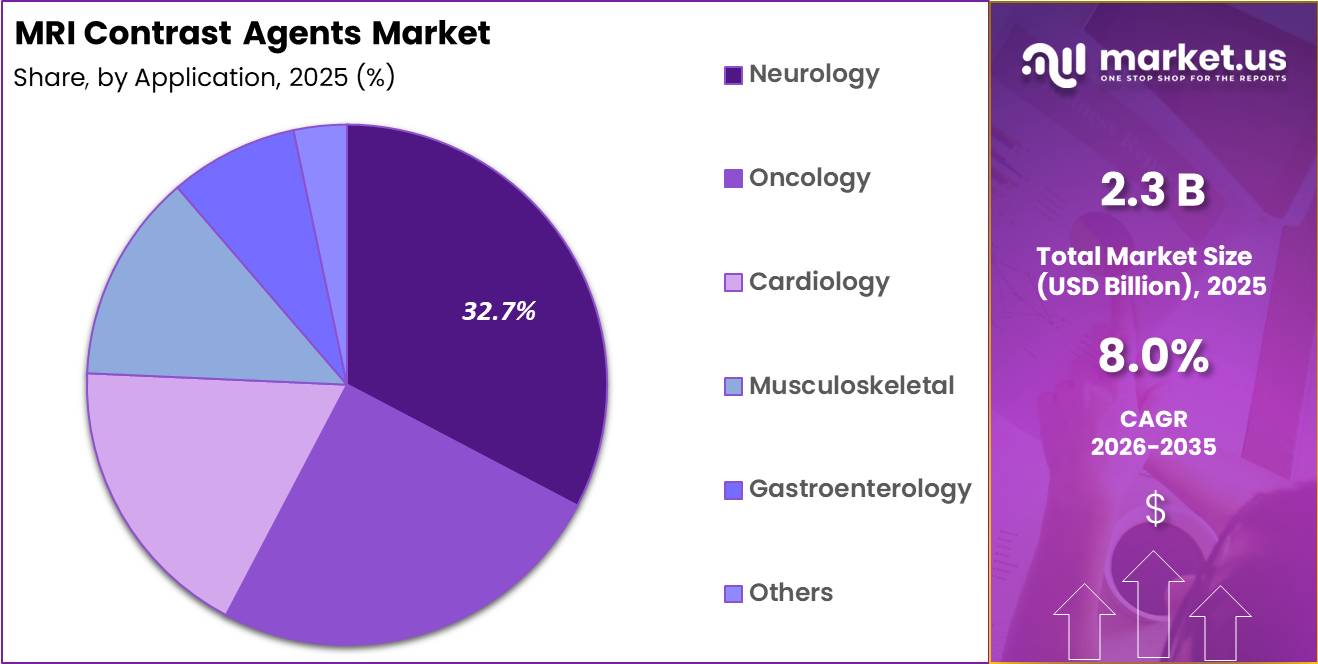
- Application Analysis: Neurology is expected to hold the largest market share, accounting for approximately 32.7%.
- Route of Administration Analysis: Intravenous administration is anticipated to dominate the segment, capturing approximately 78.4% of the market share
- End User Analysis: Hospitals are projected to lead the market, accounting for approximately 52.8%.
- Regional Analysis: In 2025, North America led the market, achieving over 8.0% share with a revenue of US$ 1.13 Billion.
Type Analysis
The MRI contrast agents market is segmented by type into extracellular agents, blood pool agents, and organ-specific agents. Among these, extracellular agents are projected to dominate the market, accounting for approximately 68.4% of the total share in 2025. This dominance can be attributed to their widespread clinical adoption, favorable safety profiles, and versatility across a broad range of imaging applications.
These agents are primarily gadolinium-based and distribute rapidly into the extracellular fluid space, making them suitable for routine diagnostic procedures. Blood pool agents, although representing a smaller share, are gaining traction due to their prolonged intravascular retention, enabling enhanced vascular imaging and improved detection of cardiovascular abnormalities.
Organ-specific agents are increasingly being utilized in targeted imaging, particularly for liver and renal diagnostics, owing to their ability to selectively accumulate in specific tissues. The growth of these segments is supported by ongoing advancements in molecular imaging and the rising demand for precision diagnostics, although concerns related to gadolinium retention may moderately influence long-term adoption trends.
Application Analysis
Based on application, the MRI contrast agents market is segmented into neurology, oncology, cardiology, musculoskeletal, gastroenterology, and others. Neurology is expected to hold the largest market share, accounting for approximately 32.7% in 2025.
This leadership is driven by the increasing prevalence of neurological disorders such as brain tumors, multiple sclerosis, and neurodegenerative diseases, where high-resolution imaging is essential for accurate diagnosis and monitoring. Oncology represents another significant segment, supported by the rising global cancer burden and the need for early tumor detection and staging.
Cardiology applications are expanding steadily due to advancements in cardiac MRI techniques and the growing incidence of cardiovascular diseases. The musculoskeletal segment benefits from increased imaging of joint disorders and sports-related injuries. Gastroenterology applications are also witnessing gradual growth, particularly in liver imaging.
Collectively, these segments are being influenced by technological improvements in MRI systems and the increasing emphasis on non-invasive diagnostic procedures.
Route of Administration Analysis
The market is segmented by route of administration into intravenous, oral, and intrathecal methods. Intravenous administration is anticipated to dominate the segment, capturing approximately 78.4% of the market share in 2025. This dominance is primarily due to its rapid systemic distribution, immediate imaging enhancement, and widespread clinical acceptance across various diagnostic applications.
Intravenous contrast agents are preferred in most MRI procedures, particularly in neurology and oncology, due to their efficiency and reliability. Oral administration, although limited in scope, is utilized in specific gastrointestinal imaging procedures where localized contrast enhancement is required.
The intrathecal route, involving direct injection into the cerebrospinal fluid, is used in specialized neurological imaging, such as detecting spinal abnormalities or cerebrospinal fluid leaks.
However, its usage remains restricted due to procedural complexity and associated risks. Overall, the preference for intravenous administration is expected to persist, supported by continuous improvements in contrast agent formulations and delivery protocols.
End User Analysis
By end user, the MRI contrast agents market is segmented into hospitals, diagnostic imaging centers, ambulatory surgical centers, and others. Hospitals are projected to lead the market, accounting for approximately 52.8% of the total share in 2025.
This dominance is driven by the high volume of MRI procedures conducted in hospital settings, availability of advanced imaging infrastructure, and the presence of skilled healthcare professionals. Diagnostic imaging centers represent a significant and growing segment, benefiting from increasing patient preference for specialized and cost-effective imaging services.
These centers often offer shorter wait times and focused expertise, contributing to their rising utilization. Ambulatory surgical centers are gradually adopting MRI technologies, particularly for preoperative and postoperative imaging, although their market share remains comparatively smaller.
The “others” category includes research institutions and specialty clinics, which contribute to market growth through clinical studies and niche applications. Overall, the expansion of healthcare infrastructure and increasing demand for diagnostic imaging are expected to sustain growth across all end-user segments.
Key Market Segments
By Type
- Extracellular Agents
- Blood Pool Agents
- Organ-Specific Agents
By Application
- Neurology
- Oncology
- Cardiology
- Musculoskeletal
- Gastroenterology
- Others
By Route of Administration
- Intravenous
- Oral
- Intrathecal
By End User
- Hospitals
- Diagnostic Imaging Centers
- Ambulatory Surgical Centers
- Others
Driving Factors
Rising MRI Utilization and Chronic Disease Burden
A major driver for MRI contrast agents is the rising use of MRI for cancer, cardiovascular and neurological imaging as populations age and chronic disease prevalence increases worldwide. The OECD reports MRI exam rates above 140 per 1,000 population in countries like the United States and Japan, compared with fewer than 10 per 1,000 in some emerging systems, indicating substantial headroom for growth as access expands.WHO estimates that noncommunicable diseases already account for about 74% of global deaths, increasing demand for high-resolution diagnostic imaging to guide treatment. In the United States, Medicare Part B imaging spending has repeatedly highlighted MRI and CT as key modalities in outpatient care, with millions of contrast-enhanced scans billed annually.
As clinical guidelines increasingly recommend MRI for early detection of multiple sclerosis, liver lesions and cardiac function, contrast-enhanced protocols become standard, reinforcing consistent demand for gadolinium-based and alternative agents within hospital and independent imaging networks.
Trending Factors
Shift Toward Safer Macrocyclic and Non-Gadolinium Agents
A dominant trend in the MRI contrast agents space is the shift from older linear gadolinium-based contrast agents (GBCAs) to macrocyclic and emerging non-gadolinium agents due to safety concerns about gadolinium retention.Since 2017, the European Medicines Agency has recommended restricting several linear GBCAs and suspending some marketing authorizations, while supporting macrocyclic agents because of their greater kinetic and thermodynamic stability. The EMA emphasized that although no clinical harm from brain gadolinium deposition has been demonstrated, precautionary measures were needed, prompting hospitals to convert formularies toward macrocyclic products.
The U.S. FDA has issued multiple Drug Safety Communications on gadolinium retention and now requires class-wide warnings, medication guides and patient-specific risk assessment, encouraging lower doses and safer formulations.
These regulatory signals have catalyzed R&D into manganese-based, iron-based and other non-gadolinium contrast systems, with numerous academic publications over the last decade outlining nanoparticle and targeted agents optimized for molecular and organ-specific MRI applications.
Restraining Factors
Regulatory Scrutiny, Safety Warnings and Liability
Regulatory scrutiny and safety-related actions constitute a key restraint on the MRI contrast agents market. In the United States, Congress passed legislation in 2022 requiring the FDA to regulate all contrast agents as drugs, regardless of mechanism, aligning them with stringent pharmaceutical quality, clinical evidence and pharmacovigilance standards.The FDA has issued class warnings on gadolinium retention and mandated patient medication guides, adding documentation and counseling workload for radiology departments and potentially limiting use in lower-risk indications.
In Europe, EMA decisions restricting or suspending multiple linear gadolinium agents have effectively removed some products from clinical practice, squeezing portfolio breadth for manufacturers and forcing substitution to specific macrocyclic brands.
These actions increase post-marketing study obligations, risk-management plans and long-term safety monitoring, raising development and compliance costs that can exceed hundreds of millions of dollars over a product life cycle. Combined with medico-legal concerns, this environment can slow adoption of novel agents and deter smaller entrants.
Opportunity
Supply Chain, Innovation and Emerging Markets
There is a significant opportunity in strengthening contrast-agent supply chains and expanding access in emerging markets while advancing safer chemistries. The U.S. government’s 100-day supply chain review highlighted vulnerabilities in pharmaceutical and imaging-related inputs, pointing out that India imports nearly 70% of its active pharmaceutical ingredients from China, creating concentration risk.Diversifying production of key drug components, including contrast media ingredients, through on-shore and near-shore manufacturing and modular production platforms is a stated policy priority that can open incentives, grants and public–private partnerships for producers.
At the same time, global imaging volumes are growing as low- and middle-income countries invest in MRI scanners under WHO and national cancer-control and noncommunicable-disease programs, where baseline MRI utilization may still be below 10 exams per 1,000 population.
Vendors that develop low-dose, macrocyclic or non-gadolinium agents with robust supply security can align with these policies and capture fast-growing procedural volumes in these under-penetrated regions.
Regional Analysis
North America maintained a leading position in the MRI contrast agents market in 2025, accounting for over 8.0% share and generating approximately US$ 1.13 billion in revenue. This dominance can be attributed to the well-established healthcare infrastructure, high adoption of advanced diagnostic imaging technologies, and strong presence of key market players.
The increasing prevalence of chronic diseases, including neurological disorders and cancer, has significantly contributed to the demand for MRI procedures across the region.
In Europe, steady market growth has been observed, supported by rising healthcare expenditure, favorable reimbursement frameworks, and expanding research activities in imaging technologies. Countries such as Germany, France, and the UK have demonstrated consistent demand due to early adoption of innovative diagnostic solutions.
The Asia-Pacific region is emerging as a high-growth market, driven by improving healthcare infrastructure, increasing patient awareness, and growing investments in medical imaging facilities. Rapid urbanization and a rising burden of lifestyle-related diseases are further accelerating market expansion in countries like China, India, and Japan.
Meanwhile, Latin America and the Middle East & Africa are experiencing gradual growth, supported by improving access to healthcare services and increasing government initiatives aimed at strengthening diagnostic capabilities. However, limited infrastructure and cost constraints may continue to restrain market penetration in these regions.
Key Regions and Countries
North America
- The US
- Canada
Europe
- Germany
- France
- The U.K.
- Italy
- Spain
- Russia & CIS
- Rest of Europe
Asia Pacific
- China
- India
- Japan
- South Korea
- ASEAN
- Australia & New Zealand
- Rest of Asia Pacific
Middle East & Africa
- GCC
- South Africa
- Rest of Middle East & Africa
Latin America
- Brazil
- Mexico
- Rest of Latin America
Key Players Analysis
The MRI contrast agents market is characterized by a highly consolidated competitive landscape, where a limited number of multinational players account for a significant share of global revenue.
Leading companies such as Bayer AG, GE HealthCare, Bracco Imaging S.p.A., and Guerbet Group collectively dominate the market, supported by strong product portfolios and extensive global distribution networks.
These firms account for a substantial proportion of market share, with the top players controlling over 70% of the global market, indicating high entry barriers and strong brand positioning.
Innovation and product differentiation are central to competitive strategies. Market leaders are heavily investing in research and development to introduce advanced gadolinium-based and macrocyclic agents with improved safety profiles and reduced toxicity risks.
For instance, Bayer AG continues to expand its portfolio through clinical advancements, while GE HealthCare leverages integrated imaging solutions combining contrast agents with AI-enabled diagnostics.
Mid-sized and emerging players, including Lantheus Medical Imaging and regional manufacturers in Asia-Pacific, are focusing on niche applications and cost-effective solutions to strengthen their market presence.
Strategic initiatives such as regulatory approvals, partnerships, and geographic expansion into emerging markets are widely adopted to sustain growth. Overall, the competitive environment remains innovation-driven, with safety, efficacy, and regulatory compliance acting as key differentiators.
Market Key Players
- Bayer AG
- Bracco Imaging S.p.A.
- GE Healthcare
- Guerbet Group
- Lantheus Medical Imaging Inc.
- Daiichi Sankyo Company Limited
- Fujifilm Holdings Corporation
- Nano Therapeutics Pvt. Ltd.
- Ixico plc
- Jodas Expoim Pvt. Ltd.
- Spago Nanomedical AB
- CMIC Holdings Co., Ltd.
- Taejoon Pharm Co., Ltd.
- Unijules Life Sciences Ltd.
- Trivitron Healthcare
- Others
Recent Developments
- Bayer AG: By March 2026, at ECR 2026, Bayer released additional subgroup data (abdominal, CNS, and MR angiography) confirming that gadoquatrane can reduce gadolinium dose by up to 60 percent while maintaining image quality, and confirmed that regulatory filings are already under review in the US, EU, Japan, and China.
- Bracco Imaging S.p.A.: In March 2025, Bracco Imaging completed the acquisition of NanoPET Pharma GmbH, expanding its portfolio of specialized contrast agents for molecular and preclinical imaging and strengthening its precision-medicine-oriented pipeline.
- GE HealthCare: In April 2026, GE HealthCare announced that the first patient was dosed in the international Phase 2/3 LUMINA trial evaluating mangaciclanol, a manganese-based MRI contrast agent designed as a potential alternative to standard gadolinium agents.
- Lantheus Medical Imaging Inc.: In February 2025, Lantheus reported successful Phase 3 data for a novel manganese-based MRI contrast agent, positioned as an alternative for patients where gadolinium use is problematic and as a response to environmental concerns around gadolinium discharge.
Report Scope
Report Features Description Market Value (2025) US$ 2.3 Billion Forecast Revenue (2035) US$ 4.9 Billion CAGR (2026-2035) 7.61% Base Year for Estimation 2025 Historic Period 2020-2024 Forecast Period 2026-2035 Report Coverage Revenue Forecast, Market Dynamics, Competitive Landscape, Recent Developments Segments Covered By Type (Extracellular Agents, Blood Pool Agents, Organ-Specific Agents) By Application (Neurology, Oncology, Cardiology, Musculoskeletal, Gastroenterology, Others) By Route of Administration (Intravenous, Oral, Intrathecal) By End User (Hospitals, Diagnostic Imaging Centers, Ambulatory Surgical Centers, Others) Regional Analysis North America – The US, Canada; Europe – Germany, France, U.K., Italy, Spain, Russia & CIS, Rest of Europe; Asia Pacific – China, India, Japan, South Korea, ASEAN, Australia & New Zealand, Rest of Asia Pacific; Middle East & Africa – GCC, South Africa, Rest of Middle East & Africa; Latin America – Brazil, Mexico, Rest of Latin America Competitive Landscape Bayer AG, Bracco Imaging S.p.A., GE Healthcare, Guerbet Group, Lantheus Medical Imaging Inc., Daiichi Sankyo Company Limited, Fujifilm Holdings Corporation, Nano Therapeutics Pvt. Ltd., Ixico plc, Jodas Expoim Pvt. Ltd., Spago Nanomedical AB, CMIC Holdings Co., Ltd., Taejoon Pharm Co., Ltd., Unijules Life Sciences Ltd., Trivitron Healthcare, Others, Customization Scope Customization for segments, region/country-level will be provided. Moreover, additional customization can be done based on the requirements. Purchase Options We have three licenses to opt for: Single User License, Multi-User License (Up to 5 Users), Corporate Use License (Unlimited User and Printable PDF) 
-
-
- Bayer AG
- Bracco Imaging S.p.A.
- GE Healthcare
- Guerbet Group
- Lantheus Medical Imaging Inc.
- Daiichi Sankyo Company Limited
- Fujifilm Holdings Corporation
- Nano Therapeutics Pvt. Ltd.
- Ixico plc
- Jodas Expoim Pvt. Ltd.
- Spago Nanomedical AB
- CMIC Holdings Co., Ltd.
- Taejoon Pharm Co., Ltd.
- Unijules Life Sciences Ltd.
- Trivitron Healthcare
- Others










