Global Intrauterine Insemination Devices Market By Product Type (Curved Catheters, Flexible Catheters, Straight Catheters and Media/ Sperm Wash), By End-User (Hospitals and IVF Clinics), Region and Companies – Industry Segment Outlook, Market Assessment, Competition Scenario, Trends and Forecast 2026-2035
- Published date: Feb 2026
- Report ID: 179173
- Number of Pages: 247
- Format:
-
keyboard_arrow_up
Quick Navigation
Report Overview
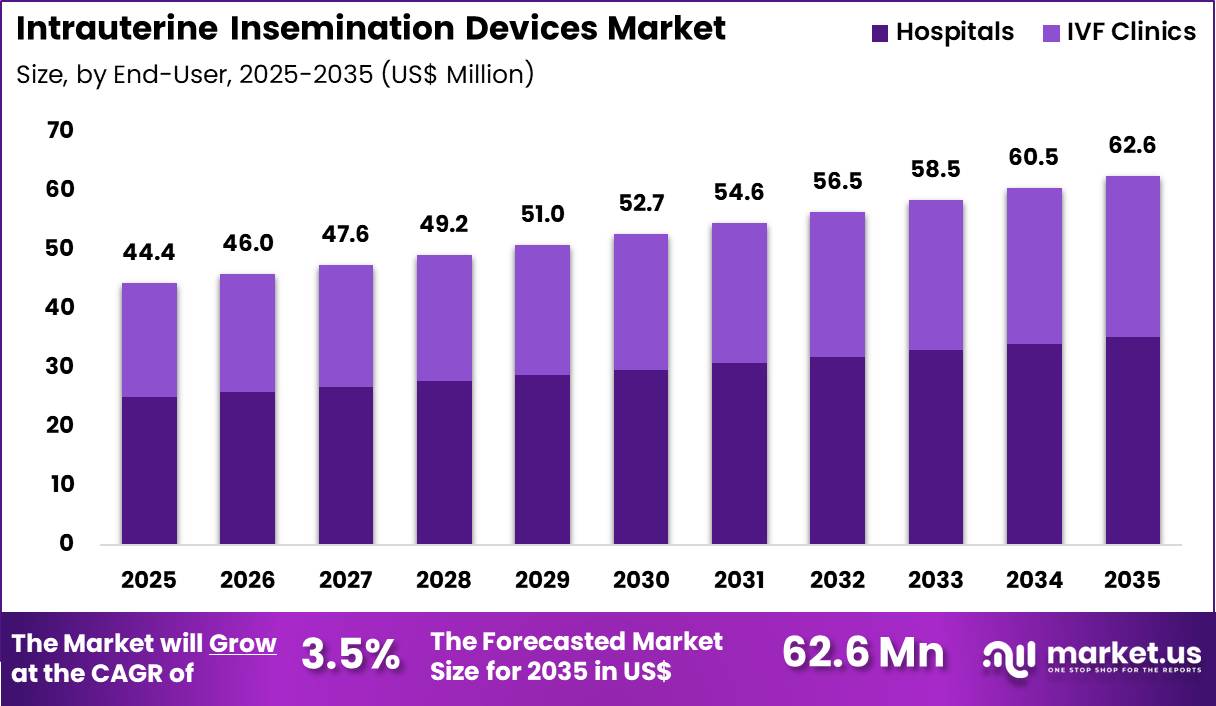
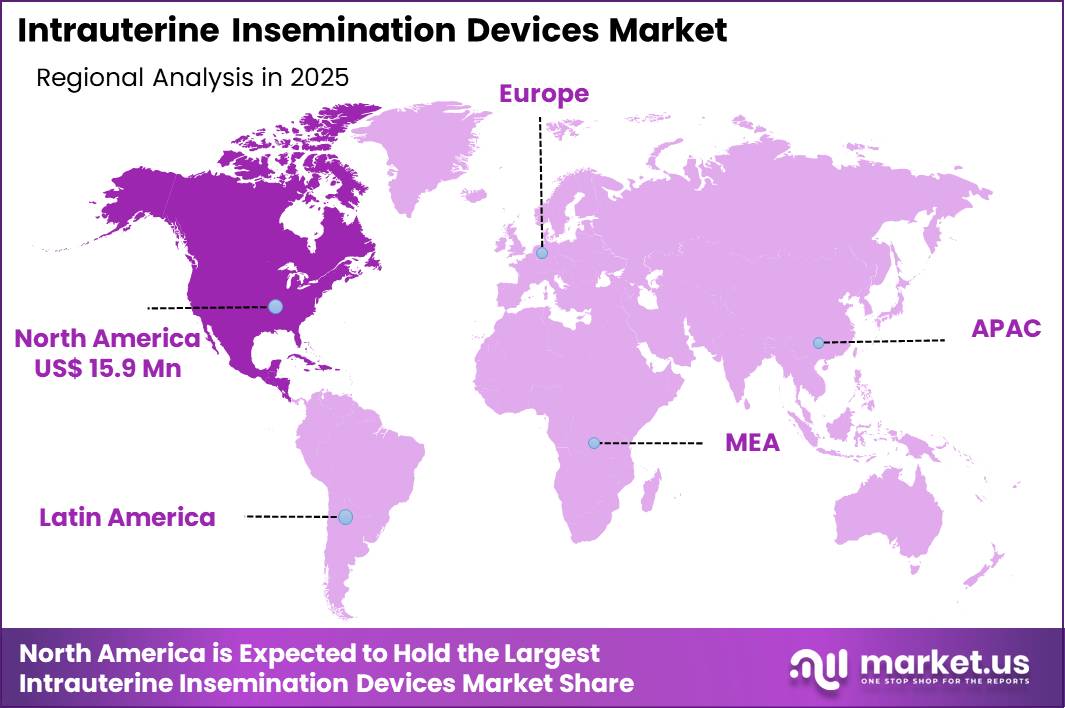
The Global Intrauterine Insemination Devices Market size is expected to be worth around US$ 62.6 Million by 2035 from US$ 44.4 Million in 2025, growing at a CAGR of 3.5% during the forecast period 2026-2035. In 2025, North America led the market, achieving over 35.8% share with a revenue of US$ 15.9 Billion.
Increasing infertility rates due to lifestyle factors, delayed childbearing, and environmental influences propel the intrauterine insemination devices market as couples and individuals seek accessible, less invasive fertility solutions.
Clinicians increasingly utilize flexible catheters and insemination kits to deliver washed sperm directly into the uterine cavity, optimizing conception chances for patients with ovulatory disorders or unexplained infertility. These devices support applications in donor insemination for single women or same-sex couples, providing precise sperm placement that enhances success rates without complex procedures.
Reproductive endocrinologists apply advanced syringes and cannulas during timed cycles, addressing male factor infertility by bypassing cervical barriers and concentrating motile sperm near fallopian tubes. Intrauterine insemination devices also enable therapeutic insemination in endometriosis cases, where controlled delivery minimizes discomfort and improves endometrial receptivity.
Manufacturers pursue opportunities to integrate ultrasound guidance and smart sensors into devices, expanding applications in real-time monitoring that ensures accurate placement and reduces multiple attempts. Developers advance disposable, pre-sterilized kits with ergonomic designs, broadening utility in outpatient clinics where efficiency and infection control remain paramount.
These innovations facilitate customized tubing lengths and tip configurations for diverse anatomical needs, optimizing outcomes in polycystic ovary syndrome treatments. Opportunities emerge in hybrid systems combining insemination with fertility tracking apps, appealing to tech-savvy users.
Companies invest in biocompatible materials that minimize irritation, enhancing patient comfort. Recent trends emphasize sustainable, eco-friendly packaging and AI-assisted procedure planning, positioning the market for growth in patient-centered fertility care.
Key Takeaways
- In 2025, the market generated a revenue of US$ 44.4 Million, with a CAGR of 3.5%, and is expected to reach US$ 62.6 Million by the year 2035.
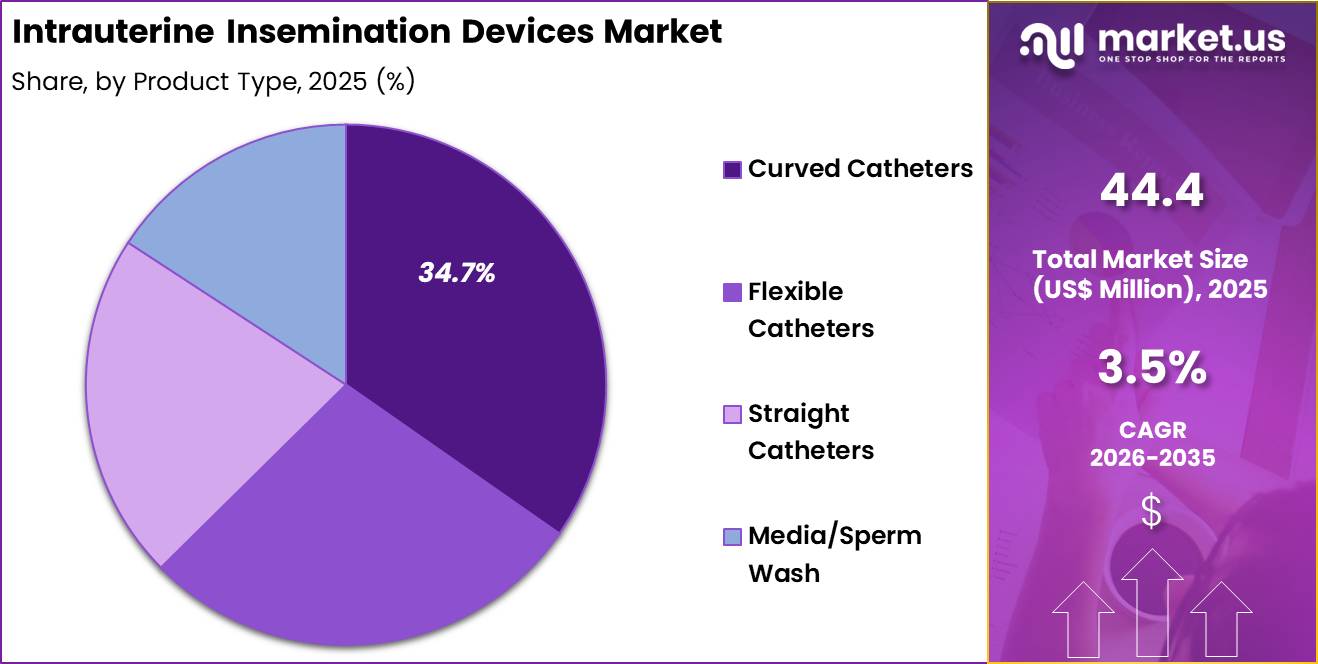
- The product type segment is divided into curved catheters, flexible catheters, straight catheters and media/ sperm wash, with curved catheters taking the lead with a market share of 34.7%.
- Considering end-user, the market is divided into hospitals and IVF clinics. Among these, hospitals held a significant share of 56.4%.
- North America led the market by securing a market share of 35.8%.
Product Type Analysis
Curved catheters accounted for 34.7% of growth within product type and led the intrauterine insemination devices market due to improved anatomical adaptability and procedural accuracy. Clinicians prefer curved designs because they facilitate smoother navigation through the cervical canal. Reduced trauma and better placement precision enhance patient comfort and procedural success rates. Rising infertility prevalence increases the number of insemination cycles performed annually.
Growth strengthens as reproductive specialists focus on optimizing clinical outcomes with minimally invasive tools. Technological improvements enhance catheter flexibility and tip softness, which supports safer insertion.
Hospitals and fertility centers adopt curved catheters to standardize procedures and reduce complications. Increasing awareness of assisted reproductive technologies expands patient volume. The segment is projected to maintain leadership as providers continue prioritizing comfort, accuracy, and procedural efficiency.
End-User Analysis
Hospitals contributed 56.4% of growth within end-user and dominated the intrauterine insemination devices market due to integrated fertility and obstetrics services. Large hospitals manage comprehensive reproductive health programs, which include diagnostic evaluation and treatment under one roof. Multidisciplinary expertise enables better patient monitoring and complication management. Growing investments in reproductive health infrastructure support equipment procurement.
Growth accelerates as urban populations seek fertility treatments in tertiary care facilities. Insurance coverage expansion in several regions increases hospital-based insemination procedures. Hospitals implement standardized clinical protocols that ensure consistent device utilization.
Strong referral networks enhance patient inflow for assisted reproduction services. The segment is anticipated to remain dominant as hospitals continue to serve as primary centers for advanced fertility treatment and reproductive care.
Key Market Segments
By Product Type
- Curved Catheters
- Flexible Catheters
- Straight Catheters
- Media/ Sperm Wash
By End-User
- Hospitals
- IVF Clinics
Drivers
Rising demand for assisted reproductive technologies is driving the market.
The expanding utilization of assisted reproductive technologies has significantly increased the demand for intrauterine insemination devices as a cost-effective initial treatment for infertility. Greater public awareness of fertility options and improved access to reproductive specialists have led to higher procedure volumes.
Fertility clinics are incorporating IUI as a first-line intervention for couples with unexplained infertility or mild male factor issues. The correlation between delayed childbearing and reduced natural conception rates further amplifies the need for IUI equipment. Government health programs are supporting fertility services to address demographic challenges.
Intrauterine insemination devices enable controlled sperm delivery, improving conception probabilities in clinical practice. National fertility registries indicate consistent growth in IUI cycles performed annually.
Leading manufacturers are developing advanced catheters with enhanced flexibility and precision. This driver encourages innovation in disposable insemination kits for improved safety. Increased infertility rates driven by lifestyle changes and medical conditions are a primary market driver.
Restraints
Limited insurance coverage for fertility treatments is restraining the market.
Inadequate insurance reimbursement for intrauterine insemination procedures creates substantial financial barriers for many patients seeking treatment. Most private and public payers classify IUI as elective, resulting in high out-of-pocket expenses for multiple cycles. Fertility clinics face challenges maintaining affordability while covering device and medication costs.
The correlation between coverage restrictions and treatment discontinuation rates limits overall market penetration. Government policies in many regions provide minimal support for fertility services, affecting patient access.
Providers must navigate complex billing requirements that delay reimbursement and strain practice finances. This restraint particularly impacts middle-income couples who cannot afford repeated attempts. Industry advocacy for expanded coverage has achieved limited progress. Despite proven efficacy, financial limitations slow broader adoption of IUI devices. Insufficient insurance coverage for fertility treatments remains a key market restraint.
Opportunities
Rising medical tourism for fertility treatments is creating growth opportunities.
The growth of medical tourism in fertility care offers substantial potential for intrauterine insemination devices in countries providing affordable, high-quality services. Governmental initiatives in select nations promote reproductive tourism through specialized clinics and streamlined visa processes. International patients seek cost-effective IUI treatments combined with advanced laboratory support.
Partnerships between local fertility centers and global device suppliers facilitate technology transfer and regulatory alignment. The large number of cross-border patients in popular destinations magnifies demand for reliable IUI equipment.
Educational programs for international patients promote standardized treatment protocols. This opportunity enables manufacturers to expand distribution through medical tourism networks. Leading companies are developing portable kits suitable for clinic use in tourist destinations.
Overall, medical tourism growth aligns with efforts to improve global fertility care access. Rising medical tourism for fertility treatments is a key growth opportunity.
Impact of Macroeconomic / Geopolitical Factors
Broader economic trends shape the intrauterine insemination devices market by influencing patient spending power, fertility clinic cash flow, and insurer reimbursement decisions. Persistent cost inflation increases expenses for sterile catheters, syringes, laboratory media, and packaging materials, which raises overall treatment pricing.
Elevated borrowing costs constrain capital investments in new reproductive health facilities and laboratory upgrades. Geopolitical uncertainty disrupts cross border trade in medical plastics and precision components, adding sourcing complexity and delivery risk.
Current US tariffs on imported medical supplies and device parts increase procurement costs and narrow supplier margins. These pressures can reduce procedure accessibility in price sensitive regions and slow capacity expansion.
At the same time, providers pursue local sourcing and lean inventory strategies to strengthen supply resilience. Continued growth in fertility awareness and delayed family planning trends supports stable and sustainable market momentum.
Latest Trends
Development of automated sperm preparation systems is a recent trend in the market.
In 2024, the introduction of automated sperm processing devices has improved consistency and efficiency in intrauterine insemination preparation. These systems reduce manual handling errors and standardize sperm concentration for better insemination outcomes.
Manufacturers have prioritized user-friendly interfaces and rapid processing times for clinic use. Clinical evaluations in 2024 confirmed comparable or improved pregnancy rates with automated methods. Technological advancements in sperm processing and intrauterine insemination techniques are improving success rates and clinical outcomes.
This progress addresses variability in traditional manual washing procedures. The trend focuses on integration with existing laboratory workflows for seamless adoption. Regulatory clearances in 2024 for automated systems have accelerated clinical integration. Industry collaborations optimize protocols for diverse semen samples. These innovations aim to enhance procedural reliability while reducing technician workload in fertility centers.
Regional Analysis
North America is leading the Intrauterine Insemination Devices Market
North America accounted for 35.8% of the Intrauterine Insemination Devices market, reflecting consistent procedural uptake and advanced reproductive care infrastructure in 2024. Rising maternal age and increasing cases of ovulatory disorders and male factor infertility have encouraged more couples to pursue early clinical intervention.
The Society for Assisted Reproductive Technology reported 389,993 ART cycles performed in the United States in 2022, underscoring the scale of fertility treatment activity that supports insemination-based procedures. Fertility clinics continue to expand capacity and invest in precision catheters and optimized sperm preparation systems to improve pregnancy outcomes.
Broader insurance coverage across several US states has strengthened patient affordability and treatment continuity. Growing acceptance of assisted conception among single women and same-sex couples has widened the eligible patient pool.
Physicians emphasize less invasive and cost-effective treatment pathways before progressing to advanced IVF cycles. These demographic, clinical, and reimbursement drivers have supported steady market expansion across the region in 2024.
The Asia Pacific region is expected to experience the highest CAGR during the forecast period
Asia Pacific is projected to experience notable growth during the forecast period as fertility rates decline and governments encourage assisted reproduction to counter demographic imbalance. South Korea’s Statistics Korea reported a total fertility rate of 0.72 in 2023, one of the lowest globally, intensifying national focus on reproductive support services.
Urbanization and career prioritization continue to delay first pregnancies across China, Japan, and India, increasing demand for clinical fertility solutions. Regional health ministries are expanding subsidies and insurance frameworks to make insemination-based treatments more accessible. Private fertility networks are scaling operations beyond metropolitan centers to address unmet demand in emerging cities.
Clinicians are adopting refined catheter technologies and laboratory protocols to improve procedural efficiency and patient comfort. Cross-border reproductive care in destinations such as Thailand and Malaysia is attracting regional patients seeking affordable options. Strong demographic pressure, policy incentives, and expanding clinic networks are expected to sustain robust growth across Asia Pacific in the coming years.
Key Regions and Countries
North America
- US
- Canada
Europe
- Germany
- France
- The UK
- Spain
- Italy
- Russia
- Netherland
- Rest of Europe
Asia Pacific
- China
- Japan
- South Korea
- India
- Australia
- New Zealand
- Singapore
- Thailand
- Vietnam
- Rest of APAC
Latin America
- Brazil
- Mexico
- Rest of Latin America
Middle East & Africa
- South Africa
- Saudi Arabia
- UAE
- Rest of MEA
Key Players Analysis
Leading participants in the intrauterine insemination devices market strengthen growth by optimizing catheter flexibility, atraumatic tip design, and controlled sperm delivery mechanisms that enhance procedural precision and patient comfort. They also expand commercial traction by offering bundled reproductive kits, lab consumables, and workflow guidance that support fertility clinics in standardizing treatment protocols.
Companies invest in clinician education initiatives and evidence-driven marketing to reinforce confidence in device reliability and improve repeat utilization rates. Strategic distribution agreements with IVF centers and women’s health networks help secure recurring procedural volumes across developed and emerging markets.
CooperSurgical, Inc., a subsidiary of CooperCompanies, operates as a specialized women’s health medical device provider with a comprehensive fertility portfolio and a strong global sales infrastructure that aligns product innovation with reproductive medicine needs.
The company advances its market position through targeted R&D programs, collaborative engagement with fertility specialists, and a commercialization strategy focused on consistent clinical performance and long-term partnership development.
Top Key Players
- Cook Medical
- CooperSurgical
- Medgyn Products
- Kitazato Corporation
- Rocket Medical
- Laboratoire CCD
- Gynetics Medical Products
- Hamilton Thorne
- Wallace Instruments
- Fertility Focus
Recent Developments
- In 2025, CooperCompanies reported that its CooperSurgical segment, which includes the Wallace IUI catheter line, generated US$ 1.35 billion in annual revenue. According to recent financial disclosures, its fertility business saw a 6% revenue increase in the third quarter, supported by a strategic reorganization to improve clinical support in the US.
- In 2026, Vitrolife Group disclosed that its full-year 2025 sales reached approximately US$ 323 million. As per the company’s February 2026 report, its consumables division—covering insemination and lab devices—achieved 8% organic growth, driven by a 10% increase in demand across the Asia Pacific region.
Report Scope
Report Features Description Market Value (2025) US$ 44.4 Million Forecast Revenue (2035) US$ 62.6 Million CAGR (2026-2035) 3.5% Base Year for Estimation 2025 Historic Period 2020-2024 Forecast Period 2026-2035 Report Coverage Revenue Forecast, Market Dynamics, COVID-19 Impact, Competitive Landscape, Recent Developments Segments Covered By Product Type (Curved Catheters, Flexible Catheters, Straight Catheters and Media/ Sperm Wash), By End-User (Hospitals and IVF Clinics) Regional Analysis North America – US, Canada; Europe – Germany, France, The UK, Spain, Italy, Russia, Netherlands, Rest of Europe; Asia Pacific – China, Japan, South Korea, India, Australia, New Zealand, Singapore, Thailand, Vietnam, Rest of APAC; Latin America – Brazil, Mexico, Rest of Latin America; Middle East & Africa – South Africa, Saudi Arabia, UAE, Rest of MEA Competitive Landscape Cook Medical, CooperSurgical, Medgyn Products, Kitazato Corporation, Rocket Medical, Laboratoire CCD, Gynetics Medical Products, Hamilton Thorne, Wallace Instruments, Fertility Focus Customization Scope Customization for segments, region/country-level will be provided. Moreover, additional customization can be done based on the requirements. Purchase Options We have three licenses to opt for: Single User License, Multi-User License (Up to 5 Users), Corporate Use License (Unlimited User and Printable PDF)  Intrauterine Insemination Devices MarketPublished date: Feb 2026add_shopping_cartBuy Now get_appDownload Sample
Intrauterine Insemination Devices MarketPublished date: Feb 2026add_shopping_cartBuy Now get_appDownload Sample -
-
- Cook Medical
- CooperSurgical
- Medgyn Products
- Kitazato Corporation
- Rocket Medical
- Laboratoire CCD
- Gynetics Medical Products
- Hamilton Thorne
- Wallace Instruments
- Fertility Focus










