Global Cosmetovigilance Market By Phase Type (Pre-clinical, Phase I, Phase II, Phase III, Phase IV), By Services Type (Pre-marketing (Clinical Safety Testing, Document Writing and Risk Management) and Post-marketing (Case Intake, Case Triage, Data Entry & Acquisition and Tracking & Reporting)), By Category (Skincare, Makeup, Haircare and Perfume & Deodorants), By Service Provider (In-house and Contract Outsourcing (Contract Research Organizations (CROs) and Business Process Outsourcing Organizations (BPOs))), Region and Companies – Industry Segment Outlook, Market Assessment, Competition Scenario, Trends and Forecast 2026-2035
- Published date: April 2026
- Report ID: 183543
- Number of Pages: 278
- Format:
-
keyboard_arrow_up
Quick Navigation
Report Overview
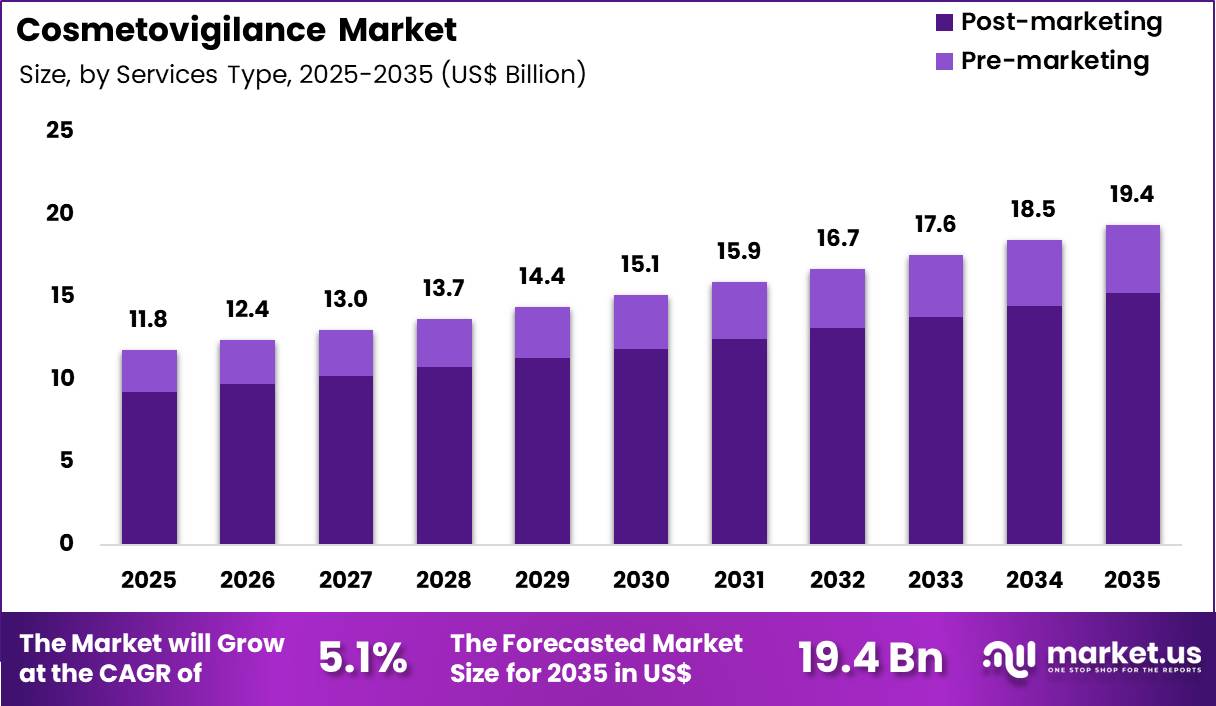
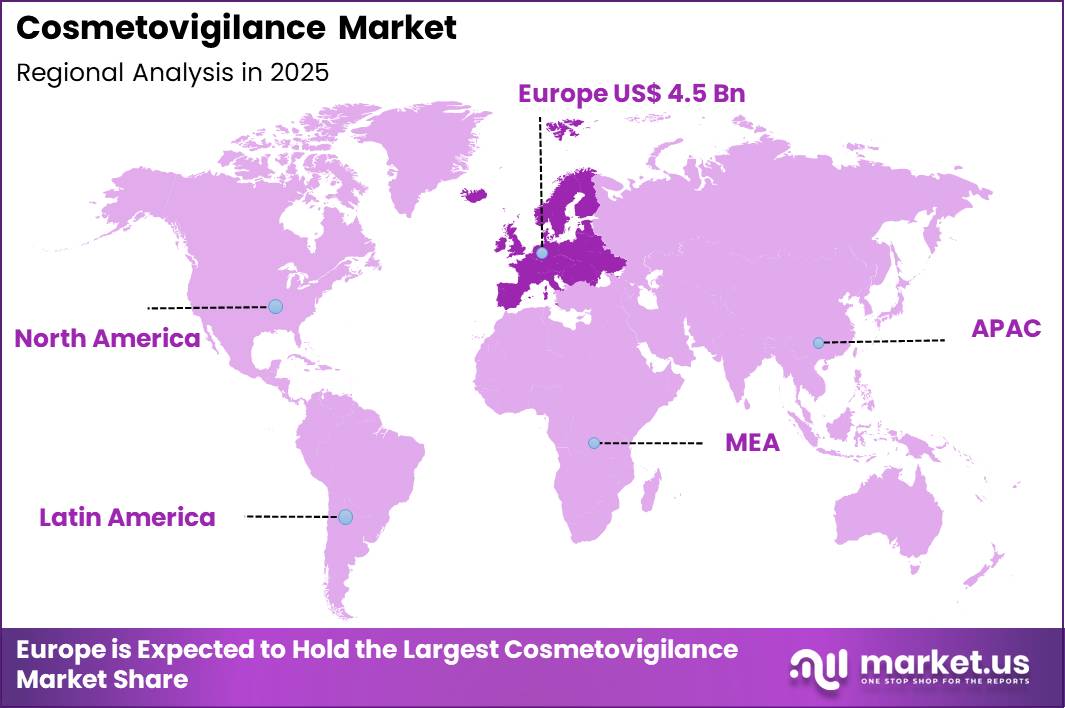
The Global Cosmetovigilance Market size is expected to be worth around US$ 19.4 Billion by 2035 from US$ 11.8 Billion in 2025, growing at a CAGR of 5.1% during the forecast period 2026-2035. In 2025, Europe led the market, achieving over 38.4% share with a revenue of US$ 4.5 Billion.
Increasing consumer use of cosmetic and personal care products, coupled with stricter global safety regulations, drives the Cosmetovigilance market as manufacturers and regulatory bodies require robust systems to monitor, report, and manage adverse events associated with these items.
Companies increasingly implement cosmetovigilance programs to track skin reactions, allergic responses, and other undesirable effects from cosmetics, sunscreens, and hair care products, enabling timely signal detection and risk mitigation.
These programs support post-market surveillance by collecting consumer-reported incidents through dedicated portals or apps, allowing brands to assess causality and implement corrective actions such as reformulation or labeling updates.
Regulatory affairs teams utilize cosmetovigilance data to comply with mandatory reporting obligations in various jurisdictions, ensuring product safety dossiers remain current and transparent.
In the cosmetics sector, safety officers apply these systems to analyze trends in low-severity events such as mild irritation or contact dermatitis, distinguishing between isolated incidents and emerging safety signals that may warrant broader investigation.
The market also serves ingredient suppliers and contract manufacturers who monitor adverse events linked to raw materials used across multiple finished products. Service providers and technology firms pursue opportunities to develop automated intake and processing platforms that handle high volumes of low-severity reports efficiently, improving workflow speed and data quality for large cosmetic portfolios.
These advancements support integration with global safety databases, facilitating cross-border signal detection and harmonized risk management. Companies invest in AI-assisted case triage and natural language processing tools that extract relevant details from unstructured consumer feedback, accelerating analysis and decision-making.
Opportunities emerge in predictive analytics that forecast potential safety issues based on usage patterns and ingredient combinations. In January 2026, AB Cube outlined its upcoming development strategy centered on IntakeEasy modules, designed to automate case intake and processing for adverse event reporting.
The solution focuses on managing large volumes of low-severity reports common in the cosmetics sector, improving efficiency in end-to-end safety workflows. Recent trends emphasize digital automation, real-time signal detection, and proactive risk management, positioning the Cosmetovigilance market as a critical enabler of consumer safety and regulatory compliance in the rapidly evolving personal care industry.
Key Takeaways
- In 2025, the market generated a revenue of US$ 11.8 Billion, with a CAGR of 5.1%, and is expected to reach US$ 19.4 Billion by the year 2035.
- The phase type segment is divided into pre-clinical, phase I, phase II, phase III, phase IV, with phase iv taking the lead with a market share of 75.1%.
- Considering services type, the market is divided into pre-marketing and post-marketing. Among these, post-marketing held a significant share of 78.5%.
- Furthermore, concerning the category segment, the market is segregated into skincare, makeup, haircare and perfume & deodorants. The skincare sector stands out as the dominant player, holding the largest revenue share of 33.1% in the market.
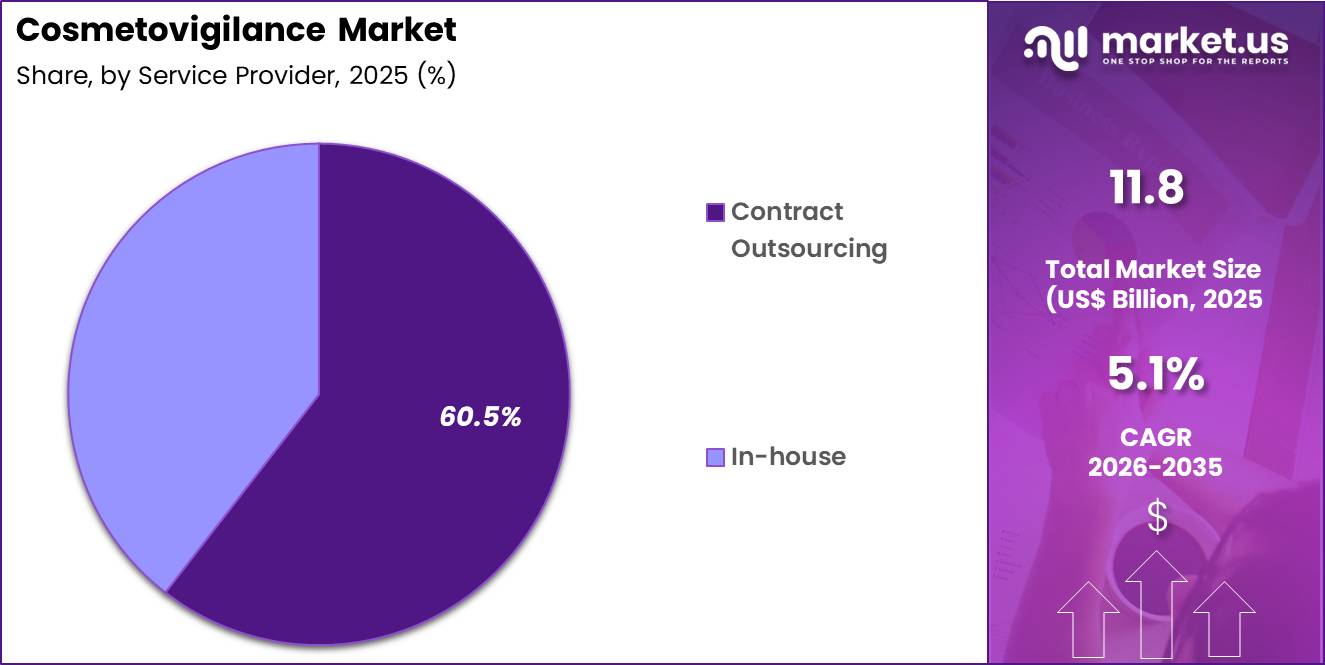
- The service provider segment is segregated into in-house and contract outsourcing, with the contract outsourcing segment leading the market, holding a revenue share of 60.5%.
- Europe led the market by securing a market share of 38.4%.
Phase Type Analysis
Phase IV accounted for 75.1% of growth within phase type and dominates the cosmetovigilance market due to the strong emphasis on post-launch safety monitoring of cosmetic products in real-world conditions. Regulatory frameworks in multiple regions require continuous surveillance of adverse effects after product commercialization, which increases reliance on phase IV activities.
Companies actively track consumer feedback, adverse event reports, and product performance to ensure compliance and brand safety. This phase is expected to expand as cosmetic product launches continue to rise across skincare, haircare, and beauty segments.
Growing consumer awareness about product safety is likely to drive higher reporting of adverse reactions. Digital platforms and reporting systems are projected to improve data collection and analysis capabilities.
The segment benefits from the need for ongoing risk assessment and regulatory documentation. As cosmetic formulations become more complex and widely used, phase IV monitoring is estimated to remain the dominant segment in this market.
Services Type Analysis
Post-marketing accounted for 78.5% of growth within services type and dominates the cosmetovigilance market due to the continuous need for monitoring product safety after commercialization. Cosmetic companies collect real-world data on adverse effects, product usage, and consumer feedback to ensure compliance with safety regulations.
Authorities in various regions mandate reporting of undesirable effects, which increases the importance of post-marketing services. This segment is expected to grow as global cosmetic consumption continues to increase.
Companies are likely to invest more in safety monitoring to protect brand reputation and avoid regulatory penalties. The segment benefits from advancements in digital reporting tools and data analytics platforms.
Social media and online reviews also contribute to real-time safety tracking. As consumer scrutiny increases and regulatory requirements become stricter, post-marketing services are anticipated to maintain their leading position in this market.
Category Analysis
Skincare accounted for 33.1% of growth within category and dominates the cosmetovigilance market due to its high usage frequency and direct contact with sensitive skin areas. Consumers use skincare products daily, which increases exposure and the likelihood of adverse reactions.
Dermatological concerns such as irritation, allergies, and sensitivity drive the need for continuous safety monitoring in this category. The segment is expected to grow as global demand for skincare products continues to rise across all age groups.
Increasing use of active ingredients and complex formulations is likely to raise the need for vigilance. Companies are projected to invest more in safety assessments to ensure product effectiveness and minimize risks.
The segment benefits from strong consumer awareness regarding skin health and product safety. As skincare remains a core segment of the beauty industry, it is estimated to retain its dominant position in the cosmetovigilance market.
Service Provider Analysis
Contract outsourcing accounted for 60.5% of growth within service provider and dominates the cosmetovigilance market due to the increasing preference of companies to rely on specialized external providers for safety monitoring and regulatory compliance.
Outsourcing allows companies to access expertise, advanced technology, and scalable solutions without building in-house infrastructure. This model is expected to grow as cosmetic companies expand globally and face diverse regulatory requirements.
Service providers are likely to offer cost-effective solutions and faster implementation of monitoring systems. The segment benefits from the need for continuous data management, reporting, and compliance support.
Outsourcing partners also provide expertise in handling large volumes of safety data and adverse event reporting. As companies focus on core product development and marketing activities, contract outsourcing is anticipated to remain the dominant service provider segment in this market.
Key Market Segments
By Phase Type
- Pre-clinical
- Phase I
- Phase II
- Phase III
- Phase IV
By Services Type
- Pre-marketing
- Clinical Safety Testing
- Document Writing
- Risk Management
- Post-marketing:
- Case Intake
- Case Triage
- Data Entry & Acquisition
- Tracking & Reporting
By Category
- Skincare
- Makeup
- Haircare
- Perfume & Deodorants
By Service Provider
- In-house
- Contract Outsourcing
- Contract Research Organizations (CROs)
- Business Process Outsourcing Organizations (BPOs)
Drivers
Increasing regulatory requirements for adverse event reporting are driving the Cosmetovigilance market.
Stringent post-market surveillance obligations have compelled cosmetic manufacturers to establish systematic monitoring and reporting frameworks for product safety.
The United States enacted the Modernization of Cosmetics Regulation Act in December 2022, mandating responsible persons to report serious adverse events to the Food and Drug Administration within 15 business days. This legislation marks the most significant expansion of FDA authority over cosmetics since 1938.
In Europe, Regulation EC 1223/2009 imposes detailed safety assessment and notification requirements through the Cosmetic Product Notification Portal. Parallel developments in China introduced a new safety-dossier mandate effective May 2025 for general cosmetics.
These synchronized global standards require companies to maintain unified databases capable of country-specific compliance extracts. Manufacturers must implement proactive signal detection rather than reactive measures. The shift elevates cosmetovigilance from an optional practice to a core operational necessity.
Compliance timelines and associated costs incentivize investment in dedicated systems and expertise. Regulatory convergence across major markets accelerates adoption of standardized processes. Consequently, these policy developments form a foundational driver sustaining market expansion during the 2022–2025 period.
Restraints
High costs of compliance and system implementation are restraining the Cosmetovigilance market.
Establishing robust cosmetovigilance programs demands significant investment in technology infrastructure, personnel training, and ongoing data management. Smaller brands and emerging market participants encounter disproportionate financial burdens when scaling safety monitoring capabilities to meet international standards.
Integration of global reporting requirements with existing quality systems adds complexity and extends project timelines substantially. Continuous updates to align with evolving regulations, including nanomaterial disclosure rules in the European Union, increase maintenance expenses over time.
Limited direct revenue attribution for safety activities discourages rapid capital allocation in cost-sensitive business environments. Administrative overhead for 15-day serious adverse event reporting under MoCRA further strains operational resources for many organizations.
These economic pressures lead some entities to delay full implementation or rely on minimal compliance approaches. Resource constraints in developing regions amplify the challenge of meeting stringent international benchmarks consistently.
Persistent gaps in standardized global protocols hinder efficiency gains across the industry. As a result, such cost-related factors impose measurable restraint on accelerated market growth throughout the 2022–2025 timeframe.
Opportunities
Expansion of outsourcing services for safety monitoring and data analytics is creating growth opportunities in the Cosmetovigilance market.
Contract service providers offer specialized platforms that combine adverse event collection, signal detection, and regulatory submission capabilities, enabling manufacturers to achieve compliance without extensive internal infrastructure development.
Opportunities arise for scalable solutions that incorporate real-world evidence and consumer-generated data from diverse channels including social media. Partnerships facilitate access to advanced analytics while reducing in-house expertise requirements for many companies.
Service models support small and medium enterprises entering regulated markets by providing cost-effective entry points with flexible scaling options. Potential exists for bundled offerings that include traceability features, blockchain verification, and cross-border database connectivity for seamless operations.
Growth in cosmetic e-commerce and complex global supply chains heightens demand for centralized monitoring that ensures rapid response to emerging safety signals. Alignment with liability insurance requirements strengthens the value proposition for proactive systems among industry participants.
These outsourced mechanisms allow organizations to focus on core product innovation while maintaining rigorous safety oversight. Overall, outsourcing avenues generate substantial prospects for market broadening and operational flexibility across various industry segments.
Impact of Macroeconomic / Geopolitical Factors
Macroeconomic conditions and geopolitical factors are influencing compliance demand, operational costs, and global expansion strategies in the cosmetovigilance market. Rising consumer awareness around product safety and increasing regulatory scrutiny are driving adoption of monitoring and reporting systems, while inflation is raising the cost of compliance tools, skilled personnel, and data management platforms.
Companies are prioritizing post-market surveillance to protect brand reputation, yet budget pressures can slow investments among smaller players. Geopolitical developments are shaping regulatory alignment and data-sharing frameworks across regions, which adds complexity for multinational cosmetic companies.
Current US tariffs have limited direct impact on cosmetovigilance services, but higher costs of imported cosmetic ingredients and packaging indirectly increase the need for stricter safety monitoring. These pressures can elevate compliance workloads and reporting requirements across the value chain.
At the same time, digitalization and automation are improving adverse event tracking and regulatory reporting efficiency. Overall, despite cost and regulatory complexity, growing emphasis on consumer safety and transparency is expected to support steady market growth.
Latest Trends
Adoption of AI-powered signal detection and digital platforms for real-time adverse event monitoring represents a recent trend in the Cosmetovigilance market.
In 2024 and 2025, companies have increasingly integrated artificial intelligence tools to process consumer complaints, social media mentions, and regulatory submissions for faster identification of potential safety signals.
The FDA launched a real-time cosmetic adverse event reporting dashboard in September 2025, providing public and industry access to daily updated data and facilitating improved transparency across reports. This development supports automated case processing and risk-based prioritization amid rising report volumes following MoCRA implementation.
Platforms now leverage natural language processing to analyze unstructured data sources, reducing manual review burdens for safety teams. The trend aligns with regulatory emphasis on proactive post-market surveillance and strict 15-day serious event timelines.
Implementations demonstrate enhanced efficiency in global compliance management through cloud-based, unified databases. Industry discussions and operational advancements in 2025 highlight AI applications in distinguishing meaningful signals from background noise within large datasets.
This evolution prioritizes data-driven decision-making over traditional manual systems. Prominent in 2024–2025, artificial intelligence integration continues to transform standards for efficient and scalable cosmetovigilance practices.
Regional Analysis
Europe is leading the Cosmetovigilance Market
Europe accounted for 38.4% of the cosmetovigilance market in 2025 as regulatory authorities and cosmetic manufacturers strengthened post-market surveillance systems to monitor product safety and adverse reactions. The region maintains one of the most stringent cosmetic regulatory frameworks, with mandatory reporting and safety assessments under established EU regulations.
According to the European Commission, over 30,000 cosmetic products are registered annually in the EU Cosmetic Products Notification Portal, reflecting a large and continuously expanding product base that requires systematic safety monitoring.
Manufacturers and distributors across Europe are increasingly implementing structured vigilance systems to track consumer complaints, adverse events, and product performance in real-world use.
Growing consumer awareness regarding ingredient safety and skin sensitivity has encouraged higher reporting of product-related reactions. Companies are investing in digital pharmacovigilance-like platforms adapted for cosmetics to ensure compliance and faster signal detection.
Regulatory authorities are also conducting periodic safety reviews and enforcing stricter compliance measures across the industry. Dermatology clinics and healthcare professionals are contributing to adverse event reporting, strengthening the overall surveillance ecosystem. These developments collectively supported steady expansion of structured cosmetic safety monitoring practices across Europe in 2025.
The Asia Pacific region is expected to experience the highest CAGR during the forecast period
Asia Pacific is expected to experience strong growth during the forecast period as cosmetic consumption rises and regulatory frameworks for product safety evolve across the region. Countries such as China, Japan, South Korea, and India are witnessing rapid expansion in cosmetic product usage driven by urbanization, rising disposable income, and increasing beauty awareness.
The World Health Organization highlights the growing importance of consumer safety monitoring in personal care products as global usage continues to rise across diverse populations. Governments across the region are strengthening regulatory guidelines for cosmetic safety, including adverse event reporting and product traceability requirements.
Cosmetic companies are increasingly adopting structured safety monitoring systems to comply with evolving regulations and maintain consumer trust. Dermatological consultations and clinical reporting are also improving identification of product-related adverse effects.
Digital platforms and mobile applications are enabling faster collection of consumer feedback and safety data. Regional manufacturers are investing in quality assurance systems and ingredient transparency initiatives. These developments are expected to accelerate adoption of cosmetovigilance practices across Asia Pacific in the coming years.
Key Regions and Countries
North America
- The US
- Canada
Europe
- Germany
- France
- The U.K.
- Italy
- Spain
- Russia & CIS
- Rest of Europe
Asia Pacific
- China
- India
- Japan
- South Korea
- ASEAN
- Australia & New Zealand
- Rest of Asia Pacific
Middle East & Africa
- GCC
- South Africa
- Rest of Middle East & Africa
Latin America
- Brazil
- Mexico
- Rest of Latin America
Key Players Analysis
Key participants in the Cosmetovigilance Market expand growth by developing advanced safety monitoring platforms, strengthening regulatory compliance frameworks, and collaborating with cosmetic brands to track adverse effects and ensure product safety across global markets.
Companies invest in data analytics, AI-driven signal detection, and digital reporting systems that enable faster identification of potential product-related risks. They also focus on integrating safety databases with regulatory systems and enhancing consumer feedback channels to improve transparency and response time.
UL Solutions represents a prominent participant in the Cosmetovigilance Market and operates as a global safety science company that provides testing, inspection, and compliance services for consumer products, including cosmetics and personal care items. The company emphasizes risk assessment, regulatory expertise, and safety validation services to support product lifecycle management.
Industry competitors continue to expand pharmacovigilance capabilities into cosmetics, strengthen global compliance strategies, and invest in digital surveillance tools to enhance product safety monitoring and sustain market growth.
Top Key Players
- Poseidon CRO
- AxeRegel
- PharSafer
- AB Cube
- Aixial Group
- Di Renzo
- Accenture
- OC Vigilance
- SKILLPHARMA S.R.L.
- Proclinical
Recent Developments
- In March 2026, Accenture introduced its Reinvention Services initiative alongside a leadership restructuring. The program emphasizes deeper integration of artificial intelligence and data-driven processes within life sciences and consumer goods operations, strengthening its outsourcing capabilities for global cosmetovigilance and regulatory support.
- In March 2026, Aixial Group expanded its operational footprint across the Nordic region and Portugal, reinforcing its positioning as a comprehensive contract research organization. The company is focusing on incorporating real-world evidence into post-marketing studies to help brands better assess long-term safety and performance of cosmetic ingredients.
Report Scope
Report Features Description Market Value (2025) US$ 11.8 Billion Forecast Revenue (2035) US$ 19.4 Billion CAGR (2026-2035) 5.1% Base Year for Estimation 2025 Historic Period 2020-2024 Forecast Period 2026-2035 Report Coverage Revenue Forecast, Market Dynamics, COVID-19 Impact, Competitive Landscape, Recent Developments Segments Covered By Phase Type (Pre-clinical, Phase I, Phase II, Phase III, Phase IV), By Services Type (Pre-marketing (Clinical Safety Testing, Document Writing and Risk Management) and Post-marketing (Case Intake, Case Triage, Data Entry & Acquisition and Tracking & Reporting)), By Category (Skincare, Makeup, Haircare and Perfume & Deodorants), By Service Provider (In-house and Contract Outsourcing (Contract Research Organizations (CROs) and Business Process Outsourcing Organizations (BPOs))) Regional Analysis North America – The US, Canada; Europe – Germany, France, U.K., Italy, Spain, Russia & CIS, Rest of Europe; Asia Pacific – China, India, Japan, South Korea, ASEAN, Australia & New Zealand, Rest of Asia Pacific; Middle East & Africa – GCC, South Africa, Rest of Middle East & Africa; Latin America – Brazil, Mexico, Rest of Latin America Competitive Landscape Poseidon CRO, AxeRegel, PharSafer, AB Cube, Aixial Group, Di Renzo, Accenture, OC Vigilance, SKILLPHARMA S.R.L., Proclinical. Customization Scope Customization for segments, region/country-level will be provided. Moreover, additional customization can be done based on the requirements. Purchase Options We have three licenses to opt for: Single User License, Multi-User License (Up to 5 Users), Corporate Use License (Unlimited User and Printable PDF) 
-
-
- Poseidon CRO
- AxeRegel
- PharSafer
- AB Cube
- Aixial Group
- Di Renzo
- Accenture
- OC Vigilance
- SKILLPHARMA S.R.L.
- Proclinical










