Global Endobronchial Ultrasound Biopsy Device Market By Product Type (EBUS Bronchoscopes, EBUS Needles, EBUS Workstations and Processors, Others) By Usability (Reusable, Single-use) By Application (Lung Cancer Diagnosis and Staging, Infectious Disease Diagnosis, Mediastinal Lesion Biopsy, Others) By End User (Hospitals, Ambulatory Surgical Centers, Specialty Clinics), Region and Companies – Industry Segment Outlook, Market Assessment, Competition Scenario, Trends and Forecast 2026-2035
- Published date: May 2026
- Report ID: 185766
- Number of Pages: 264
- Format:
-
keyboard_arrow_up
Quick Navigation
Report Overview
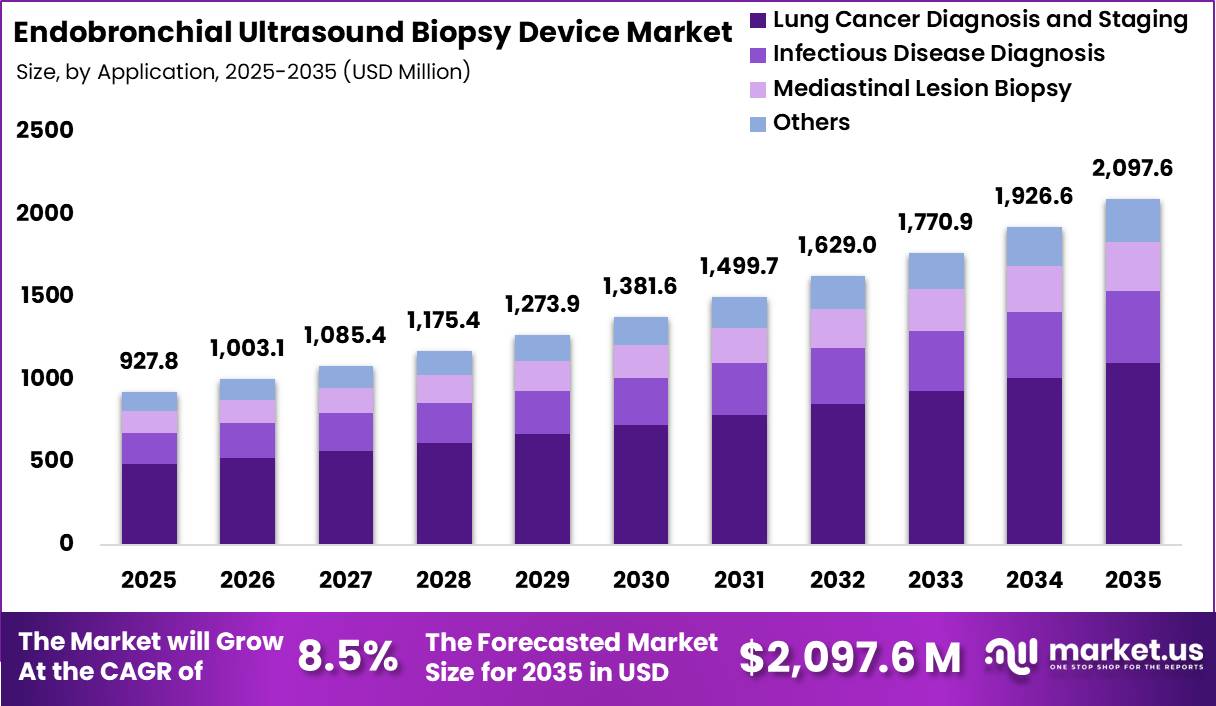
Global Endobronchial Ultrasound Biopsy Device Market size is expected to be worth around US$ 2,097.6 Million by 2035 from US$ 927.8 Million in 2025, growing at a CAGR of 8.5% during the forecast period from 2026 to 2035. In 2025, North America led the market, achieving over 39.8% share with a revenue of US$ 369.6 Million.
The global endobronchial ultrasound biopsy device market is experiencing steady growth due to the increasing burden of lung cancer, tuberculosis, and other respiratory diseases worldwide.Endobronchial ultrasound (EBUS) biopsy devices are minimally invasive diagnostic tools used to examine airways and collect tissue samples from lymph nodes and lung lesions. These devices are widely adopted for diagnosing and staging lung cancer because they provide real-time imaging with improved diagnostic accuracy and reduced surgical complications.
According to the U.S. Centers for Disease Control and Prevention (CDC), lung cancer remains the third most common cancer in the United States, with approximately 218,893 new lung cancer cases reported in 2022 and 131,584 deaths recorded in 2023.
The growing number of lung cancer patients is significantly increasing the demand for advanced bronchoscopic and biopsy procedures globally. In addition, the World Health Organization (WHO) reported that nearly 10.8 million people were affected by tuberculosis globally in 2023, highlighting the rising need for accurate pulmonary diagnostic technologies.
The market is also benefiting from technological advancements such as ultrasound-guided transbronchial needle aspiration (EBUS-TBNA), portable bronchoscopy systems, and integration of artificial intelligence in imaging analysis.
These innovations improve procedure precision, shorten diagnosis time, and enhance patient safety. Healthcare providers are increasingly preferring minimally invasive diagnostic methods, as EBUS procedures reduce hospital stays, lower complication risks, and improve patient recovery rates.
Hospitals, specialty pulmonary clinics, and cancer centers represent the major end users of EBUS biopsy devices. North America currently dominates the market due to advanced healthcare infrastructure, high screening rates, and growing adoption of minimally invasive pulmonary procedures.
Meanwhile, Asia-Pacific is expected to witness significant growth driven by rising smoking rates, increasing respiratory disease prevalence, expanding healthcare infrastructure, and improving access to advanced diagnostic technologies.
Key Takeaways
- Market Size: Global Endobronchial Ultrasound Biopsy Device Market size is expected to be worth around US$ 2,097.6 Million by 2035 from US$ 927.8 Million in 2025.
- Market Share: The market growing at a CAGR of 8.5% during the forecast period from 2026 to 2035.
- Product Type Analysis: In 2025, North America led the market, achieving over 39.8% share with a revenue of US$ 369.6 Million.
- Usability Analysis: EBUS bronchoscopes segment accounted for 42.8% of the market share in 2025.
- Application Analysis: The reusable segment dominated the market with a 68.1% share in 2025.
- End User Analysis: lung cancer diagnosis and staging segment accounted for 52.5% of the market share in 2025.
- Regional Analysis: In 2025, North America led the market, achieving over 39.8% share with a revenue of US$ 369.6 Million.
Product Type Analysis
The product type segment of the Endobronchial Ultrasound Biopsy Device Market is led by EBUS bronchoscopes, which accounted for 42.8% of the market share in 2025. The dominance of this segment is attributed to the increasing clinical adoption of minimally invasive bronchoscopy procedures for lung cancer diagnosis and mediastinal lymph node assessment.
EBUS bronchoscopes provide real-time ultrasound imaging and enhanced visualization capabilities, improving procedural accuracy and diagnostic yield. Technological advancements such as high-definition imaging, improved maneuverability, and compatibility with robotic bronchoscopy systems have further accelerated segment growth.
The EBUS needles segment represents another significant category and includes aspiration needles, biopsy forceps, and cytology brushes. Rising demand for accurate tissue sampling and molecular testing in oncology diagnostics is supporting the adoption of these consumables. Increasing preference for EBUS-guided transbronchial needle aspiration procedures is also contributing to segment expansion.
EBUS workstations and processors are witnessing steady growth owing to advancements in imaging software, data integration systems, and ultrasound processing technologies. The others segment, comprising accessories and ancillary devices, continues to gain traction due to increasing procedural volumes across hospitals and specialty pulmonary centers.
Usability Analysis
Based on usability, the Endobronchial Ultrasound Biopsy Device Market is segmented into reusable and single-use devices. The reusable segment dominated the market with a 68.1% share in 2025. This dominance is primarily associated with the high adoption of reusable EBUS bronchoscopes and biopsy instruments in hospitals and large healthcare facilities due to their long-term cost efficiency and operational reliability.
Healthcare providers continue to prefer reusable systems because of their ability to support high procedural volumes while reducing per-procedure costs over extended usage cycles. In addition, continuous advancements in sterilization technologies and infection control protocols have strengthened confidence in reusable devices.
The single-use segment is expected to witness notable growth during the forecast period owing to increasing concerns regarding cross-contamination and hospital-acquired infections.
Disposable EBUS needles and bronchoscopic accessories are gaining traction in outpatient and ambulatory settings where procedural efficiency and infection prevention remain critical priorities. Furthermore, regulatory emphasis on patient safety and growing awareness regarding sterile diagnostic procedures are supporting adoption of single-use products.
Rising demand for minimally invasive pulmonary diagnostic procedures, coupled with expanding bronchoscopy volumes globally, is expected to support growth across both usability categories. However, reusable devices are anticipated to maintain market leadership due to their widespread integration within established healthcare infrastructure and favorable cost economics for large-scale clinical operations.
Application Analysis
Based on application, the Endobronchial Ultrasound Biopsy Device Market is dominated by the lung cancer diagnosis and staging segment, which accounted for 52.5% of the market share in 2025. The segment’s leadership is primarily driven by the increasing global incidence of lung cancer and the growing adoption of minimally invasive diagnostic techniques.
EBUS-guided procedures are widely utilized for mediastinal lymph node evaluation, tumor staging, and tissue sampling, offering higher diagnostic accuracy and lower procedural complications compared to traditional surgical biopsies. The increasing use of molecular profiling and personalized oncology treatment approaches has further accelerated demand for EBUS biopsy procedures in cancer diagnostics.
The infectious disease diagnosis segment is witnessing steady growth due to the rising prevalence of respiratory infections such as tuberculosis and granulomatous diseases. EBUS-guided sampling enables clinicians to obtain accurate tissue specimens for microbiological and pathological analysis, improving diagnostic efficiency in pulmonary infections.
The mediastinal lesion biopsy segment is also expanding significantly owing to increasing detection of thoracic abnormalities through advanced imaging modalities. EBUS devices are increasingly preferred for evaluating mediastinal masses and lymphadenopathy because they reduce the need for invasive surgical procedures.
The others segment includes applications in inflammatory disease assessment and pulmonary lesion evaluation, which continue to contribute to overall market growth.
End User Analysis
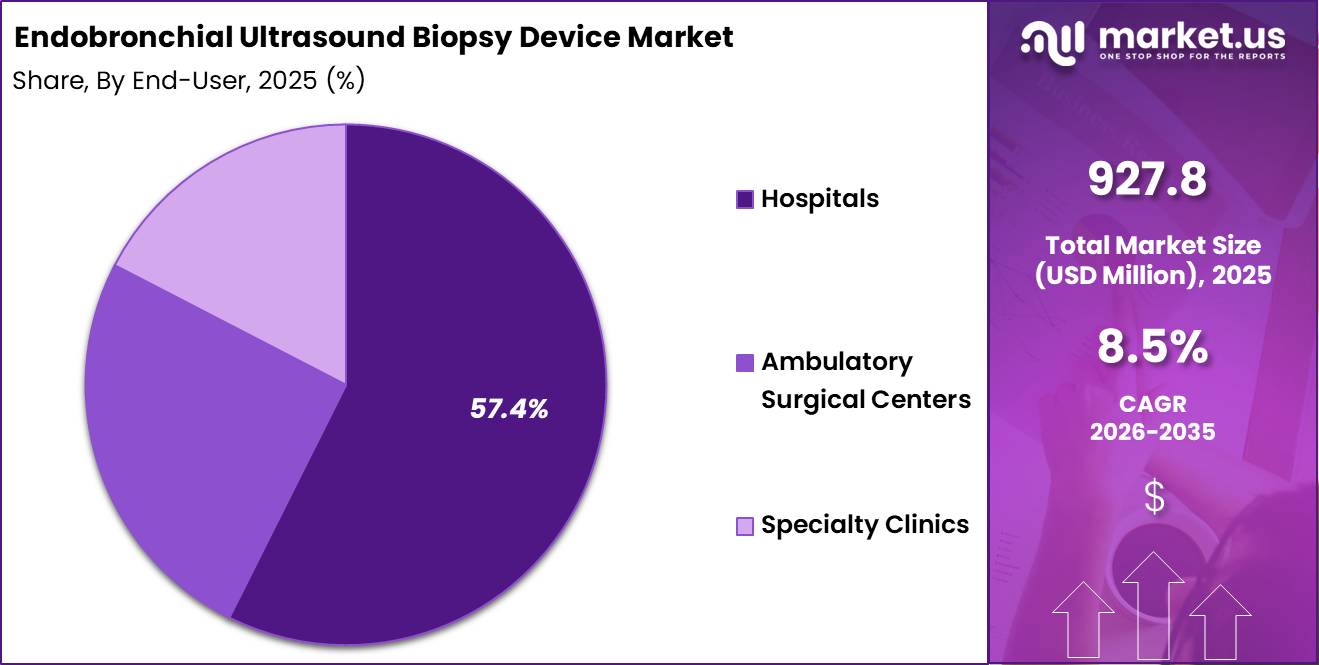
Based on end user, the Endobronchial Ultrasound Biopsy Device Market is led by hospitals, which accounted for 57.4% of the market share in 2025. The dominance of hospitals is attributed to the availability of advanced bronchoscopy infrastructure, skilled pulmonologists, and integrated diagnostic facilities.
Hospitals perform a high volume of EBUS-guided procedures for lung cancer staging, mediastinal lesion evaluation, and respiratory disease diagnosis. In addition, favorable reimbursement policies, growing investments in minimally invasive diagnostic technologies, and increasing adoption of robotic-assisted bronchoscopy systems are supporting segment expansion.
Ambulatory surgical centers are expected to register notable growth during the forecast period due to rising demand for cost-effective outpatient diagnostic procedures.
These facilities offer shorter patient stay durations, lower procedural costs, and improved operational efficiency, making them increasingly attractive for EBUS-guided biopsy procedures. Technological advancements in portable ultrasound systems and disposable accessories are further enhancing adoption in ambulatory settings.
Specialty clinics also represent an important segment, particularly in pulmonary and oncology-focused care centers. Increasing patient preference for specialized respiratory diagnostic services and the growing availability of advanced EBUS platforms in private clinics are contributing to segment growth. Expanding awareness regarding early lung cancer detection and minimally invasive diagnostics is expected to support continued adoption across all end-user categories.
Key Market Segments
By Product Type
- EBUS Bronchoscopes
- EBUS Needles
- Aspiration Needles
- Biopsy Forceps
- Cytology Brushes
- EBUS Workstations and Processors
- Others
By Usability
- Reusable
- Single-use
By Application
- Lung Cancer Diagnosis and Staging
- Infectious Disease Diagnosis
- Mediastinal Lesion Biops
- Others
By End User
- Hospitals
- Ambulatory Surgical Centers
- Specialty Clinics
Driving Factors
The increasing global burden of lung cancer is a major driver for the Endobronchial Ultrasound (EBUS) Biopsy Device Market. According to the World Health Organization, lung cancer accounted for approximately 2.5 million new cases and 1.8 million deaths globally in 2022, making it the leading cause of cancer-related mortality worldwide.
The rising incidence of smoking, air pollution exposure, and occupational respiratory disorders has significantly increased demand for accurate and minimally invasive diagnostic technologies. EBUS-guided biopsy procedures are increasingly preferred over conventional surgical mediastinoscopy because they offer real-time imaging, reduced procedural complications, and faster patient recovery.
In addition, healthcare systems are emphasizing early cancer detection programs to improve survival rates. The growing adoption of bronchoscopy-based diagnostic pathways across hospitals and pulmonary care centers is supporting market expansion.
Increasing awareness regarding early-stage lung cancer diagnosis, combined with technological advancements in ultrasound-guided bronchoscopic imaging, is further accelerating device utilization. Expanding healthcare infrastructure and rising investments in respiratory disease diagnostics are expected to sustain long-term market growth globally.
Trending Factors
A significant trend shaping the Endobronchial Ultrasound Biopsy Device Market is the growing adoption of single-use bronchoscopic devices and advanced infection-control technologies. Healthcare providers are increasingly focusing on reducing hospital-acquired infections associated with reusable endoscopic instruments.
According to the U.S. Food and Drug Administration, reusable endoscopes and bronchoscopes require extensive reprocessing and sterilization procedures after every patient use to minimize contamination risks. Studies published through PubMed indicate that reusable bronchoscopes have been linked to infection outbreaks due to inadequate reprocessing practices and procedural complexity.
Consequently, healthcare facilities are increasingly adopting disposable EBUS accessories and single-use flexible bronchoscopes to improve patient safety and operational efficiency. Another emerging trend is the integration of artificial intelligence-assisted imaging and robotic bronchoscopy systems, which improve lesion targeting and biopsy precision.
The increasing shift toward outpatient pulmonary procedures and minimally invasive diagnostics is also encouraging innovation in portable ultrasound processors and compact EBUS systems. These technological advancements are expected to enhance procedural accuracy and clinical workflow efficiency across respiratory care settings.
Restraining Factors
High procedural and equipment costs remain a major restraint for the Endobronchial Ultrasound Biopsy Device Market, particularly across low- and middle-income healthcare systems. EBUS procedures require specialized bronchoscopes, ultrasound processors, biopsy needles, and highly trained pulmonologists, increasing the overall operational expenditure for healthcare facilities.
In addition, reusable bronchoscopes require rigorous cleaning, disinfection, and sterilization procedures after each use. According to the U.S. Food and Drug Administration, inadequate reprocessing of reusable medical devices may lead to microbial contamination and healthcare-associated infections, creating additional compliance and maintenance burdens for hospitals.
The complexity and time required for high-level disinfection procedures further increase labor and infrastructure costs. Furthermore, the limited availability of skilled respiratory specialists in developing regions restricts widespread procedural adoption. Several healthcare facilities in emerging economies continue to rely on conventional diagnostic methods due to budget limitations and lack of advanced bronchoscopy infrastructure.
Reimbursement variability for minimally invasive pulmonary diagnostic procedures also affects adoption rates across certain regions. These factors collectively create barriers to market penetration, particularly in resource-constrained healthcare environments where capital-intensive diagnostic technologies remain less accessible.
Opportunity
Growing emphasis on early lung cancer screening and minimally invasive respiratory diagnostics presents substantial opportunities for the Endobronchial Ultrasound Biopsy Device Market. According to the World Health Organization, lung cancer remains the most common cause of cancer mortality globally, with approximately 2.5 million new cases reported in 2022.
Increasing awareness regarding the benefits of early detection is encouraging governments and healthcare providers to expand pulmonary screening initiatives. The rising use of low-dose computed tomography screening programs is expected to increase the number of patients requiring confirmatory biopsy procedures, thereby driving demand for EBUS-guided diagnostics.
In countries such as India, healthcare institutions have reported increasing lung cancer admissions and rising late-stage diagnoses, highlighting the need for improved diagnostic accessibility. Furthermore, advancements in robotic bronchoscopy, artificial intelligence-based lesion navigation, and portable imaging systems are creating opportunities for more precise and efficient tissue sampling.
Expanding healthcare infrastructure in emerging economies and growing investments in oncology diagnostics are also expected to support market expansion. Increasing preference for outpatient minimally invasive procedures is likely to accelerate adoption of technologically advanced EBUS biopsy platforms across hospitals and specialty pulmonary centers.
Regional Analysis
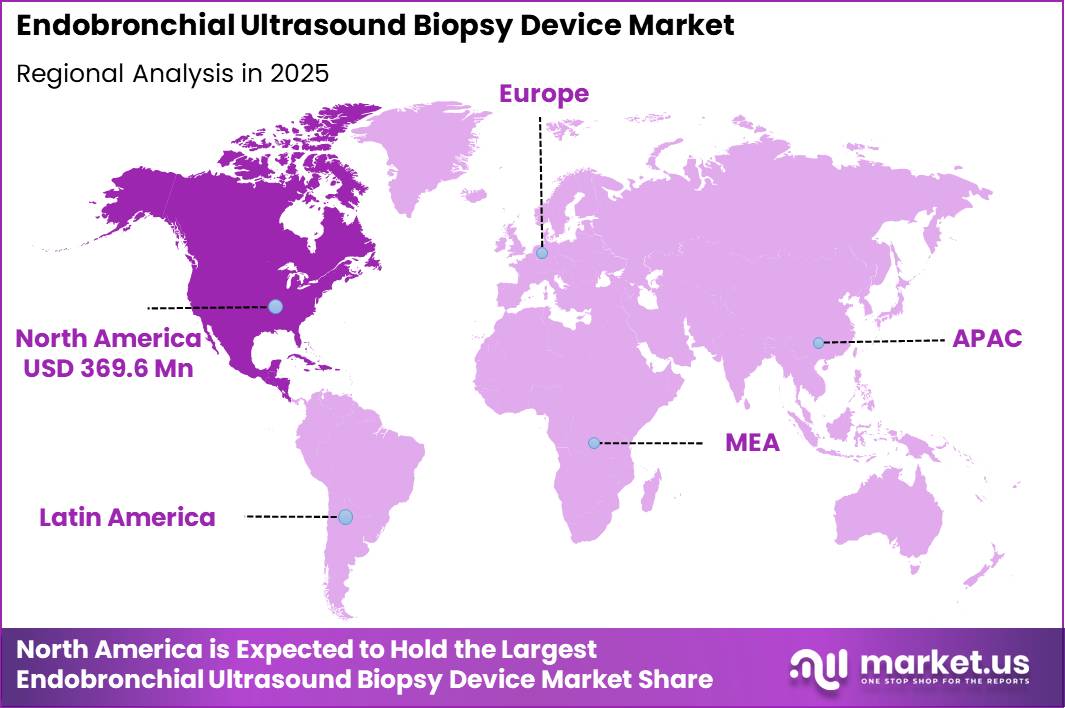
North America dominated the Endobronchial Ultrasound (EBUS) Biopsy Device Market in 2025, accounting for over 39.8% of the global market share and generating revenue of approximately US$ 369.6 million.
The regional market growth can be attributed to the strong presence of advanced healthcare infrastructure, increasing prevalence of lung cancer and respiratory disorders, and rising adoption of minimally invasive diagnostic procedures across the United States and Canada. The growing emphasis on early-stage cancer detection and precision diagnostics has significantly accelerated the demand for EBUS biopsy devices in hospitals, specialty clinics, and diagnostic centers.
Furthermore, favorable reimbursement frameworks, high healthcare expenditure, and continuous technological advancements in bronchoscopy and ultrasound imaging systems have strengthened market expansion across the region. The presence of leading medical device manufacturers and increasing investments in pulmonary care have also contributed to product innovation and wider accessibility of EBUS-guided procedures.
In addition, the growing geriatric population, which is more susceptible to chronic respiratory diseases, continues to support long-term market demand. Academic research institutions and healthcare organizations across North America are increasingly focusing on improving diagnostic accuracy and patient outcomes, further enhancing the adoption of endobronchial ultrasound biopsy technologies throughout the region.
Key Regions and Countries
North America
- The US
- Canada
Europe
- Germany
- France
- The U.K.
- Italy
- Spain
- Russia & CIS
- Rest of Europe
Asia Pacific
- China
- India
- Japan
- South Korea
- ASEAN
- Australia & New Zealand
- Rest of Asia Pacific
Middle East & Africa
- GCC
- South Africa
- Rest of Middle East & Africa
Latin America
- Brazil
- Mexico
- Rest of Latin America
Key Players Analysis
The Endobronchial Ultrasound (EBUS) Biopsy Device market is characterized by the presence of several established medical technology companies focusing on product innovation, minimally invasive diagnostic solutions, and strategic collaborations.
Key players such as Olympus Corporation, FUJIFILM Holdings Corporation, Boston Scientific Corporation, and Medtronic plc dominate the competitive landscape through extensive product portfolios and strong global distribution networks. These companies are actively investing in advanced ultrasound imaging technologies and needle biopsy systems to improve diagnostic accuracy in lung cancer and mediastinal disease detection.
Olympus Corporation maintains a leading position owing to its technologically advanced EBUS bronchoscopes and ultrasound processors widely adopted in hospitals and specialty clinics. FUJIFILM is strengthening its market presence through high-definition imaging systems and enhanced endoscopic visualization technologies. Meanwhile, Boston Scientific and Medtronic focus on expanding minimally invasive pulmonary diagnostic solutions through research and development activities and strategic acquisitions.
Emerging players are increasingly entering the market with cost-effective biopsy devices and disposable accessories, intensifying market competition. Furthermore, partnerships with healthcare institutions and rising investments in respiratory disease diagnostics are supporting product commercialization.
The competitive environment is expected to remain innovation-driven, with companies emphasizing precision diagnostics, workflow efficiency, and improved patient outcomes to strengthen their market share.
Market Key Players
- Olympus Corporation
- Boston Scientific Corporation
- Medtronic plc
- FUJIFILM Holdings Corporation
- Cook Medical, Inc.
- Pentax Medical (HOYA Group)
- CONMED Corporation
- Teleflex Incorporated
- Ambu A/S
- KARL STORZ SE & Co. KG
- Richard Wolf GmbH
- Medi-Globe GmbH
- Taewoong Medical Co., Ltd.
- Hobbs Medical, Inc.
- Micro-Tech Endoscopy USA, Inc.
- Others
Recent Developments
- Boston Scientific Corporation (March 2025): entered into a definitive agreement to acquire SoniVie Ltd., a medical device company specializing in ultrasound-based renal denervation technology. The acquisition expanded Boston Scientific’s ultrasound-enabled intervention portfolio and highlighted the company’s continued investment in advanced minimally invasive technologies.
- Olympus Corporation (September 2025): Introduced its latest BF-UCP190F EBUS bronchoscope across EMEA and Asia-Pacific markets. The new device was developed with a thinner outer diameter and enhanced maneuverability to improve access to deeper lung regions during EBUS-TBNA procedures. The launch is expected to support more precise lung cancer diagnosis and staging procedures.
- Medtronic plc (2025): continued strengthening its minimally invasive and pulmonary intervention business through ongoing investments in diagnostic technologies and surgical innovation platforms. The company also reported continued growth in its medical surgical segment during fiscal 2025, supported by rising demand for advanced procedural technologies.
- FUJIFILM Holdings Corporation (2025): Continued expanding its endoscopy and diagnostic imaging portfolio with strategic focus on advanced bronchoscopic visualization technologies and minimally invasive pulmonary diagnostic systems, supporting broader adoption of precision-guided respiratory procedures.
Report Scope
Report Features Description Market Value (2025) US$ 927.8 Million Forecast Revenue (2035) US$ 2,097.6 Million CAGR (2026-2035) 8.5% Base Year for Estimation 2025 Historic Period 2020-2024 Forecast Period 2026-2035 Report Coverage Revenue Forecast, Market Dynamics, Competitive Landscape, Recent Developments Segments Covered By Product Type (EBUS Bronchoscopes, EBUS Needles, EBUS Workstations and Processors, Others) By Usability (Reusable, Single-use) By Application (Lung Cancer Diagnosis and Staging, Infectious Disease Diagnosis, Mediastinal Lesion Biopsy, Others) By End User (Hospitals, Ambulatory Surgical Centers, Specialty Clinics) Regional Analysis North America – The US, Canada; Europe – Germany, France, U.K., Italy, Spain, Russia & CIS, Rest of Europe; Asia Pacific – China, India, Japan, South Korea, ASEAN, Australia & New Zealand, Rest of Asia Pacific; Middle East & Africa – GCC, South Africa, Rest of Middle East & Africa; Latin America – Brazil, Mexico, Rest of Latin America Competitive Landscape Olympus Corporation, Boston Scientific Corporation, Medtronic plc, FUJIFILM Holdings Corporation, Cook Medical, Inc., Pentax Medical (HOYA Group), CONMED Corporation, Teleflex Incorporated, Ambu A/S, KARL STORZ SE & Co. KG, Richard Wolf GmbH, Medi-Globe GmbH, Taewoong Medical Co., Ltd., Hobbs Medical, Inc., Micro-Tech Endoscopy USA, Inc., Others Customization Scope Customization for segments, region/country-level will be provided. Moreover, additional customization can be done based on the requirements. Purchase Options We have three licenses to opt for: Single User License, Multi-User License (Up to 5 Users), Corporate Use License (Unlimited User and Printable PDF)  Endobronchial Ultrasound Biopsy Device MarketPublished date: May 2026add_shopping_cartBuy Now get_appDownload Sample
Endobronchial Ultrasound Biopsy Device MarketPublished date: May 2026add_shopping_cartBuy Now get_appDownload Sample -
-
- Olympus Corporation
- Boston Scientific Corporation
- Medtronic plc
- FUJIFILM Holdings Corporation
- Cook Medical, Inc.
- Pentax Medical (HOYA Group)
- CONMED Corporation
- Teleflex Incorporated
- Ambu A/S
- KARL STORZ SE & Co. KG
- Richard Wolf GmbH
- Medi-Globe GmbH
- Taewoong Medical Co., Ltd.
- Hobbs Medical, Inc.
- Micro-Tech Endoscopy USA, Inc.
- Others










