Global Life Sciences Translation Services Market By Services (Clinical Trial Documentation, Regulatory Submission & Compliance, Medical Device & Technical Documents, Pharmaceutical & Biotech Content, Patient Education & Healthcare, Scientific & Medical Research and Others), By Category (Manual and Technology/AI-Based), By End User (Healthcare Professionals (HCPs), Regulatory Authorities (e.g., FDA, EMA), Clinical Research Organizations (CROs) & Researchers, Marketing & Regulatory Teams and Others), Region and Companies – Industry Segment Outlook, Market Assessment, Competition Scenario, Trends and Forecast 2026-2035
- Published date: March 2026
- Report ID: 181583
- Number of Pages: 384
- Format:
-
keyboard_arrow_up
Quick Navigation
Report Overview
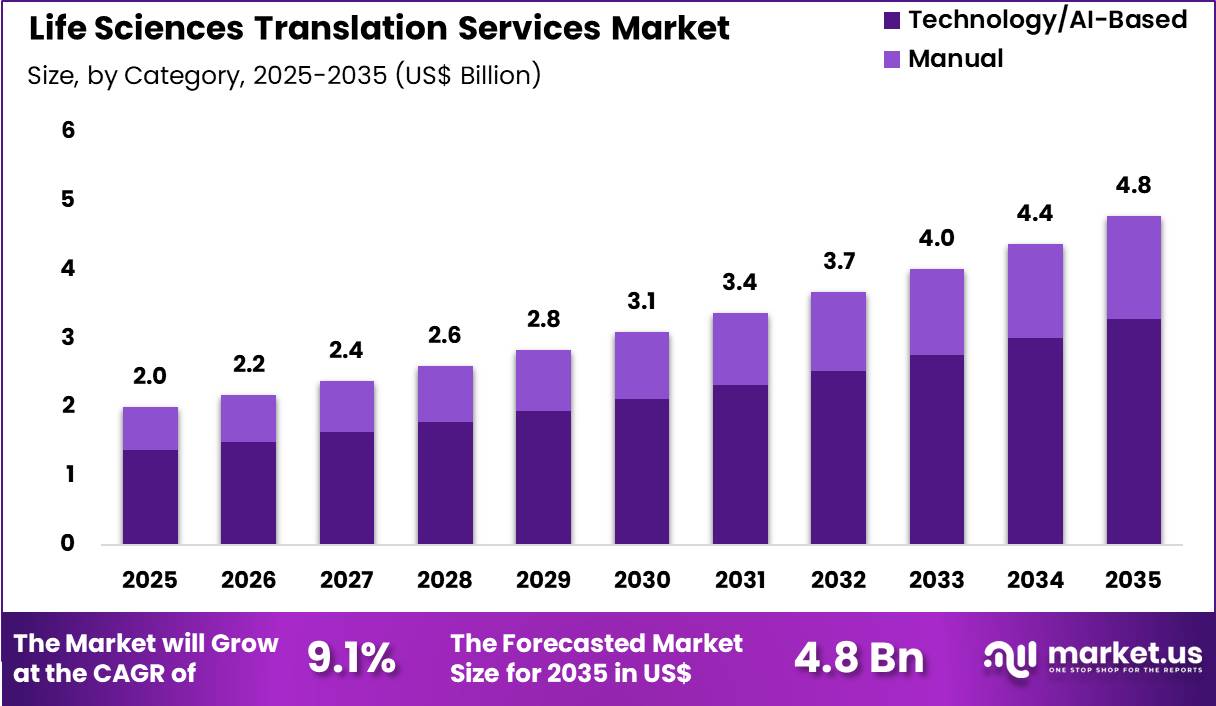
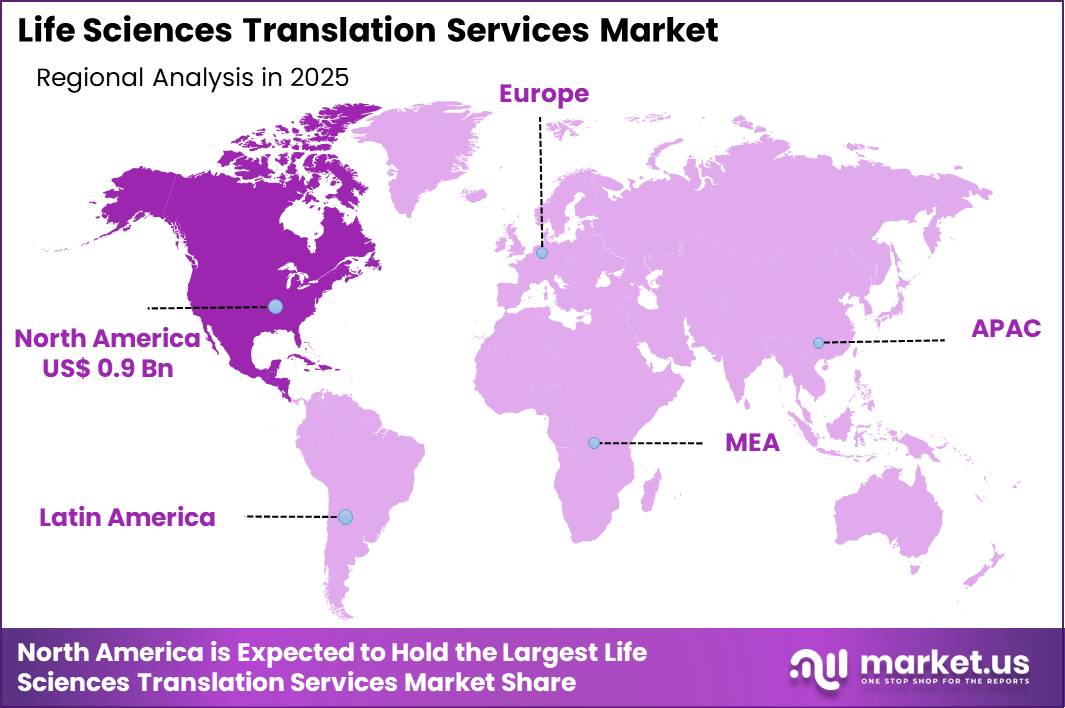
The Global Life Sciences Translation Services Market size is expected to be worth around US$ 4.8 Billion by 2035 from US$ 2.0 Billion in 2025, growing at a CAGR of 9.1% during the forecast period 2026-2035. In 2025, North America led the market, achieving over 46.2% share with a revenue of US$ 0.9 Billion.
Increasing globalization of clinical trials and pharmaceutical development propels the Life Sciences Translation Services market as companies require accurate, compliant multilingual content to support international regulatory submissions and global market access.
Sponsors increasingly rely on specialized translation providers to localize clinical trial protocols, informed consent forms, and patient-reported outcome measures, ensuring linguistic accuracy and cultural appropriateness for diverse participant populations across Phase I to Phase IV studies.
These services support regulatory documentation by translating investigator brochures, clinical study reports, and marketing authorization applications into multiple languages while maintaining scientific precision and adherence to ICH guidelines. Pharmaceutical manufacturers utilize translation expertise for product labeling, package inserts, and summaries of product characteristics, enabling compliant launches in multilingual markets with consistent safety and efficacy messaging.
Medical device companies apply these services to localize instructions for use, technical files, and risk management files, facilitating CE marking and global regulatory approvals. In addition, translation providers handle pharmacovigilance documentation, including adverse event reports and periodic safety update reports, ensuring timely and accurate communication to global health authorities.
Translation service providers pursue opportunities to integrate artificial intelligence and agent-based workflows that automate repetitive tasks, expanding applications in high-volume content localization for clinical trial amendments and post-marketing surveillance. These advancements support real-time collaboration platforms that streamline multilingual content review and approval across global teams.
Opportunities emerge in AI-assisted patent translation and IP portfolio management, accelerating research documentation and regulatory filings. In January 2026, Welocalize received industry recognition as a finalist at the Global Business Tech Awards for its OPAL Enable platform. The solution uses artificial intelligence to manage and optimize highly regulated multilingual content used in clinical and life sciences communications.
During 2026, localization providers began transitioning from traditional sequential translation workflows toward autonomous or agent-based systems. Companies such as Vistatec have implemented AI-driven agents that independently handle specialized stages of the localization process, improving turnaround times for multilingual content support.
In late 2025, Questel introduced new AI-based assistant tools designed to support patent management and intellectual property workflows. These tools help life sciences organizations improve productivity during research, patent drafting, and regulatory filing stages.
Recent trends emphasize AI-augmented translation, regulatory-focused automation, and seamless integration with content management systems, positioning the market for growth in efficient, compliant, and scalable multilingual support for life sciences innovation.
Key Takeaways
- In 2025, the market generated a revenue of US$ 2.0 Billion, with a CAGR of 9.1%, and is expected to reach US$ 4.8 Billion by the year 2035.
- The services segment is divided into clinical trial documentation, regulatory submission & compliance, medical device & technical documents, pharmaceutical & biotech content, patient education & healthcare, scientific & medical research and others, with medical device & technical documents taking the lead with a market share of 38.4%.
- Considering category, the market is divided into manual and technology/AI-based. Among these, technology/ai-based held a significant share of 68.7%.
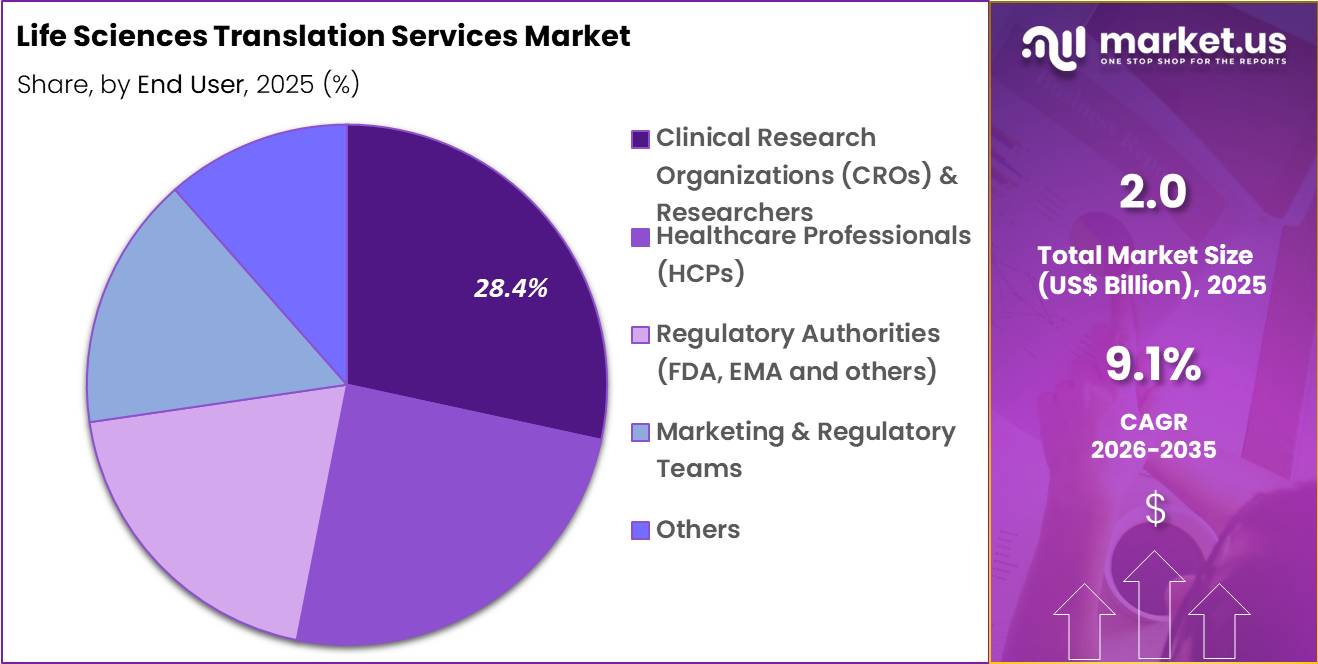
- Furthermore, concerning the end user segment, the market is segregated into healthcare professionals (HCPs), regulatory authorities, clinical research organizations (CROs) & researchers, marketing & regulatory teams and others. The clinical research organizations (cros) & researchers sector stands out as the dominant player, holding the largest revenue share of 28.4% in the market.
- North America led the market by securing a market share of 46.2%.
Services Analysis
Medical device and technical documents accounted for 38.4% of growth within services and dominate the life sciences translation services market due to the rising global trade of medical devices and increasing regulatory documentation requirements. Medical device manufacturers must translate instructions for use, safety documentation, labeling materials, and technical specifications into multiple languages to comply with international regulatory frameworks.
The global medical device sector continues to expand across emerging and developed markets, which increases the demand for multilingual technical documentation. Regulatory authorities require precise translations to ensure product safety and patient protection in local markets. Companies entering new geographic markets therefore rely heavily on specialized translation providers to meet regulatory standards.
The segment is expected to strengthen as global regulatory frameworks such as the European Union Medical Device Regulation increase documentation and localization requirements. Rising international device commercialization and stricter compliance standards are projected to further expand translation demand within this segment.
Category Analysis
Technology and AI-based translation accounted for 68.7% of growth within category and dominate the life sciences translation services market due to the increasing adoption of machine translation, language automation platforms, and AI-assisted localization tools. Life sciences organizations manage large volumes of documentation including clinical trial data, regulatory files, and research publications that require efficient multilingual processing.
AI-supported translation systems improve turnaround time and reduce operational costs while maintaining translation consistency across documents. Neural machine translation and automated terminology management tools help translation teams process highly technical medical content more efficiently.
Technology-driven workflows are expected to expand as language service providers integrate artificial intelligence into regulatory and clinical translation platforms. Growing digital transformation across pharmaceutical and biotechnology companies further strengthens the adoption of automated translation solutions in life sciences documentation.
End-User Analysis
Clinical research organizations and researchers accounted for 28.4% of growth within end users and dominate the life sciences translation services market due to the increasing globalization of clinical trials and multinational research collaborations. CROs manage clinical trials across multiple countries, which requires translation of patient consent forms, trial protocols, investigator brochures, and regulatory documentation.
The globalization of pharmaceutical research has significantly expanded the need for multilingual clinical trial materials. According to the U.S. National Library of Medicine clinical trial registry, hundreds of thousands of studies are conducted worldwide, many involving international research sites.
Researchers rely on translation services to ensure that study documentation complies with regulatory and ethical requirements in each participating country. The segment is anticipated to grow as pharmaceutical companies continue expanding global clinical trials and cross-border biomedical research initiatives.
Key Market Segments
By Services
- Clinical Trial Documentation
- Regulatory Submission & Compliance
- Medical Device & Technical Documents
- Pharmaceutical & Biotech Content
- Patient Education & Healthcare
- Scientific & Medical Research
- Others
By Category
- Manual
- Technology/AI-Based
By End User
- Healthcare Professionals (HCPs)
- Regulatory Authorities (e.g., FDA, EMA)
- Clinical Research Organizations (CROs) & Researchers
- Marketing & Regulatory Teams
- Others
Drivers
Rising volume of international clinical trials is driving the market.
The life sciences translation services market continues to expand due to sustained growth in multinational clinical research requiring accurate multilingual documentation. Sponsors increasingly conduct Phase II and Phase III studies across diverse geographic regions to access broader patient populations. Regulatory submissions demand certified translations of protocols, informed consent forms, and investigator brochures in local languages.
The driver aligns with globalization of pharmaceutical development pipelines targeting emerging markets. Contract research organizations report higher demand for linguistic validation of patient-reported outcome measures. The trend corresponds with increased filings in regions requiring native-language materials for ethics committee approval.
Translation providers support seamless integration of translated content into electronic data capture systems. The momentum reflects ongoing efforts to reduce enrollment delays through culturally appropriate communication. Enhanced quality assurance processes ensure compliance with Good Clinical Practice standards. This factor maintains consistent forward progression in specialized translation demand.
Restraints
Stringent regulatory requirements for linguistic validation are restraining the market.
Health authorities mandate rigorous linguistic validation processes for patient-facing materials used in global clinical trials. These requirements involve forward-backward translation, cognitive debriefing with target language speakers, and reconciliation steps to ensure conceptual equivalence. The restraint increases project timelines and associated costs for translation vendors.
Sponsors face challenges coordinating multiple rounds of review with clinical teams and linguistic experts. The factor limits flexibility in rapid-response scenarios for protocol amendments. Service providers must maintain extensive networks of qualified translators with domain-specific expertise.
The dynamic moderates scalability for smaller translation agencies lacking established validation infrastructure. The constraint contributes to concentrated market share among vendors with proven regulatory track records. This limitation persists in influencing pricing structures and project scoping. The requirement continues to temper accelerated growth in certain service segments.
Opportunities
Expansion of AI-supported translation memory systems is creating growth opportunities.
Advanced translation management platforms now incorporate neural machine translation engines fine-tuned on life sciences corpora alongside human post-editing workflows. These hybrid systems enable faster initial drafts of non-critical documents while preserving accuracy through specialized glossaries. Opportunities arise for cost optimization in high-volume translation projects such as pharmacovigilance reports.
The framework supports scalable handling of adverse event narratives across multiple languages. Developers gain capacity to reduce turnaround times for periodic safety update reports. The technology facilitates consistent terminology usage across large multinational programs. Such capabilities attract sponsors seeking efficiency gains without compromising quality.
The opportunity fosters differentiation through measurable productivity improvements. Stakeholders anticipate broader acceptance as validation data accumulate. This advancement positions participants for competitive advantage in high-throughput translation services.
Impact of Macroeconomic / Geopolitical Factors
Macroeconomic conditions influence the life sciences translation services market through pharmaceutical R&D spending, clinical trial activity, and regulatory compliance investments. Inflation increases costs for skilled linguistic professionals, AI-assisted translation platforms, and data security systems, which raises operating expenses for service providers.
Higher interest rates reduce funding flexibility for smaller biotech firms, which can slow international clinical trial expansion. Geopolitical tensions complicate cross-border research collaboration and increase regulatory scrutiny for multilingual documentation in global trials. Current US tariffs on imported IT infrastructure and language technology hardware increase operational costs for translation service firms.
These pressures may delay technology upgrades and compress margins for smaller service providers. At the same time, companies strengthen domestic technology partnerships and adopt AI-assisted translation tools to improve efficiency. Growing globalization of drug development and regulatory documentation continues to support steady and confident market growth.
Latest Trends
Increased demand for certified translations of regulatory dossiers is driving the market.
The U.S. Food and Drug Administration and European Medicines Agency continue to require certified translations of key sections within marketing authorization applications submitted in non-English languages. This requirement applies to summaries of product characteristics, labeling, and package inserts intended for multilingual markets.
The 2024-2025 period reflects ongoing expansion of centralized procedures necessitating translations into all official EU languages. Translation providers with ISO 17100 certification and sworn translator networks gain preference for high-stakes submissions. The development aligns with accelerated approval pathways requiring rapid turnaround of multilingual documentation.
Sponsors report improved submission readiness through dedicated regulatory translation teams. The trend supports investment in terminology management databases specific to therapeutic areas. The increased volume strengthens the strategic importance of specialized linguistic services.
Early implementations demonstrate reduced query-response cycles from regulatory authorities. Overall, this regulatory demand elevates the critical role of professional translation in global product approvals.
Regional Analysis
North America is leading the Life Sciences Translation Services Market
North America accounted for 38.8% of the life sciences translation services market in 2025 as pharmaceutical companies, biotechnology firms, and medical device manufacturers expanded global clinical research and regulatory submissions. Organizations across the United States and Canada increasingly require accurate translation of clinical trial documentation, regulatory dossiers, product labeling, and patient information materials to meet international compliance standards.
The U.S. Food and Drug Administration’s Center for Drug Evaluation and Research approved 55 novel drugs in 2023, reflecting strong pharmaceutical development activity that generates large volumes of multilingual clinical and regulatory documentation.
Growing globalization of clinical trials has encouraged pharmaceutical sponsors to conduct studies across multiple countries, increasing demand for specialized translation services that meet strict medical and regulatory requirements.
Life sciences companies rely on professional linguists with medical expertise to ensure scientific accuracy and regulatory consistency across global markets. Contract research organizations and regulatory consulting firms are also expanding partnerships with language service providers to support multinational trial operations.
Digital translation management systems and AI-assisted linguistic tools are improving efficiency while maintaining quality in complex regulatory content. Academic research institutions conducting international collaborations are also generating multilingual scientific publications and documentation. These factors collectively supported steady expansion of specialized life sciences translation services across North America in 2025.
The Asia Pacific region is expected to experience the highest CAGR during the forecast period
Asia Pacific is expected to experience strong growth during the forecast period as clinical research activity, pharmaceutical manufacturing, and regulatory documentation requirements expand across the region. Countries such as China, India, Japan, and South Korea are increasingly participating in multinational clinical trials and pharmaceutical research programs.
According to the World Health Organization International Clinical Trials Registry Platform, Asia accounts for a significant share of registered global clinical trials, reflecting growing research activity that requires multilingual documentation and regulatory translation. Pharmaceutical companies conducting trials across diverse linguistic regions rely on specialized translation services to ensure accurate communication with regulators, investigators, and patients.
Governments across the region are strengthening regulatory frameworks that require precise translation of product labeling, medical device instructions, and pharmacovigilance documentation. Biotechnology startups and contract research organizations are also expanding international collaborations that generate multilingual scientific reports and clinical data.
Universities and research institutes are increasing publication output in global scientific journals, which often requires professional language adaptation. Technology providers are introducing AI-assisted translation platforms that improve turnaround times for complex biomedical documents. These developments are expected to accelerate the demand for specialized life sciences translation services throughout Asia Pacific in the coming years.
Key Regions and Countries
North America
- The US
- Canada
Europe
- Germany
- France
- The U.K.
- Italy
- Spain
- Russia & CIS
- Rest of Europe
Asia Pacific
- China
- India
- Japan
- South Korea
- ASEAN
- Australia & New Zealand
- Rest of Asia Pacific
Middle East & Africa
- GCC
- South Africa
- Rest of Middle East & Africa
Latin America
- Brazil
- Mexico
- Rest of Latin America
Key Players Analysis
Key participants in the Life Sciences Translation Services Market expand their reach by strengthening specialized language capabilities, investing in AI-assisted translation tools, and building partnerships with pharmaceutical, biotechnology, and clinical research organizations. Companies focus on regulatory-compliant translation processes that support clinical trial documentation, product labeling, and medical device submissions across global markets.
They also establish dedicated life sciences language teams and quality review frameworks that ensure accuracy in highly regulated healthcare communications. IQVIA represents a prominent participant in the Life Sciences Translation Services Market and operates as a global healthcare services company headquartered in the United States that provides clinical research, data analytics, and regulatory support solutions for pharmaceutical and biotechnology companies.
The firm integrates language services with clinical trial operations and regulatory documentation to streamline global drug development programs. Industry competitors continue to expand multilingual capabilities, invest in automation technologies, and strengthen regulatory expertise to support the growing demand for accurate medical and scientific translation worldwide.
Top Key Players
- Vistatec
- Language Scientific
- marstranslation
- BayanTech.
- Lionbridge Technologies, LLC.
- Conversis
- Morningside, Inc.
- Crimson Interactive Inc.
- Welocalize Life Sciences
- ALM Translations Ltd
- Stepes
- Questel.
- BURG Translations
Recent Developments
- In September 2025, Conversis introduced SideKick, an artificial intelligence–enabled platform developed to streamline administrative and documentation tasks associated with clinical trial translation. The system focuses on improving efficiency and maintaining consistency across multilingual clinical research documents.
- In December 2025, Sesen launched TrialS, a regulatory validation solution designed for life sciences companies managing global labeling submissions. The platform helps organizations verify translation accuracy and maintain consistency across regulatory documents in real time during submission workflows.
- Throughout 2025 and into early 2026, Lionbridge expanded the use of its Aurora AI platform across core localization operations. The system incorporates large language model–supported post-editing processes that help reduce manual review requirements and improve translation workflow efficiency.
Report Scope
Report Features Description Market Value (2025) US$ 2.0 Billion Forecast Revenue (2035) US$ 4.8 Billion CAGR (2026-2035) 9.1% Base Year for Estimation 2025 Historic Period 2020-2024 Forecast Period 2026-2035 Report Coverage Revenue Forecast, Market Dynamics, COVID-19 Impact, Competitive Landscape, Recent Developments Segments Covered By Services (Clinical Trial Documentation, Regulatory Submission & Compliance, Medical Device & Technical Documents, Pharmaceutical & Biotech Content, Patient Education & Healthcare, Scientific & Medical Research and Others), By Category (Manual and Technology/AI-Based), By End User (Healthcare Professionals (HCPs), Regulatory Authorities (e.g., FDA, EMA), Clinical Research Organizations (CROs) & Researchers, Marketing & Regulatory Teams and Others) Regional Analysis North America – The US, Canada; Europe – Germany, France, The U.K., Italy, Spain, Russia & CIS, Rest of Europe; Asia Pacific – China, India, Japan, South Korea, ASEAN, Australia & New Zealand, Rest of Asia Pacific; Middle East & Africa – GCC, South Africa, Rest of Middle East & Africa; Latin America – Brazil, Mexico, Rest of Latin America Competitive Landscape Vistatec, Language Scientific, marstranslation, BayanTech., Lionbridge Technologies, LLC., Conversis, Morningside, Inc., Crimson Interactive Inc., Welocalize Life Sciences, ALM Translations Ltd, Stepes, Questel., BURG Translations. Customization Scope Customization for segments, region/country-level will be provided. Moreover, additional customization can be done based on the requirements. Purchase Options We have three licenses to opt for: Single User License, Multi-User License (Up to 5 Users), Corporate Use License (Unlimited User and Printable PDF)  Life Sciences Translation Services MarketPublished date: March 2026add_shopping_cartBuy Now get_appDownload Sample
Life Sciences Translation Services MarketPublished date: March 2026add_shopping_cartBuy Now get_appDownload Sample -
-
- Vistatec
- Language Scientific
- marstranslation
- BayanTech.
- Lionbridge Technologies, LLC.
- Conversis
- Morningside, Inc.
- Crimson Interactive Inc.
- Welocalize Life Sciences
- ALM Translations Ltd
- Stepes
- Questel.
- BURG Translations










