Global 3D Printing Medical Implants Market By Material (Metals, Polymers, Ceramics), By Application (Dental, Orthopedic, Cranio-maxillofacial, Others), By Technology (Laser Beam Melting, Electronic Beam Melting, Droplet Deposition, Others), By End-User (Hospitals & Surgical Centers, Dental & Orthopedic Clinics, Academic Institutions & Research Laboratories, Pharma-Biotech & Medical Device Companies), Region and Companies – Industry Segment Outlook, Market Assessment, Competition Scenario, Trends and Forecast 2025-2034
- Published date: July 2025
- Report ID: 152159
- Number of Pages: 263
- Format:
-
keyboard_arrow_up
Quick Navigation
Report Overview
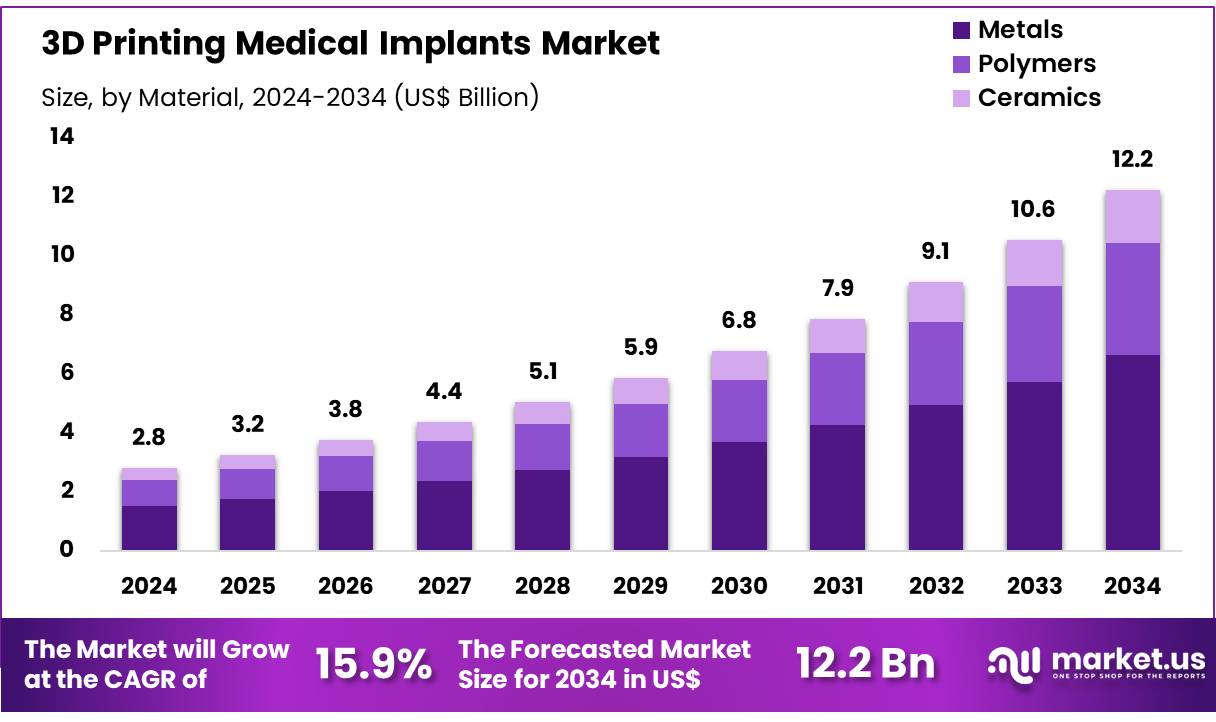
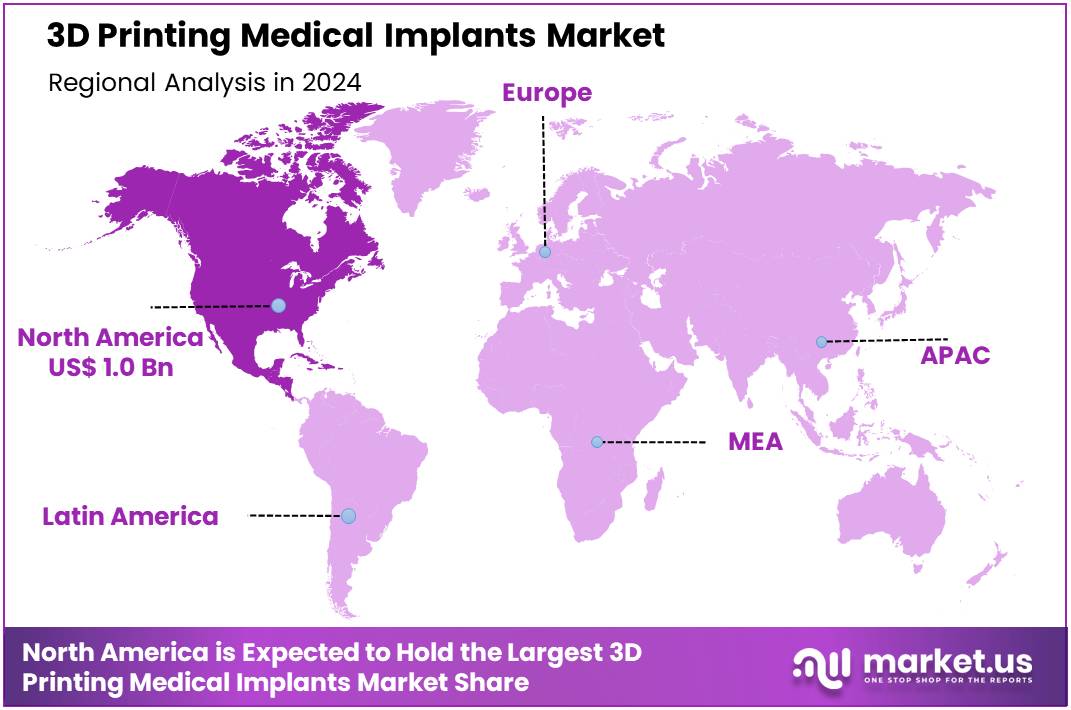
Global 3D Printing Medical Implants Market size is forecasted to be valued at US$ 12.2 Billion by 2034 from US$ 2.8 Billion in 2024, growing at a CAGR of 15.9% during the forecast period 2025 to 2034. In 2024, North America led the market, achieving over 36.6% share with a revenue of US$ 1.0 Billion.
The market is experiencing rapid growth, driven by technological advancements, increasing demand for personalized healthcare solutions, and the rising prevalence of chronic diseases. Technological innovations in 3D printing have significantly enhanced the precision and customization of medical implants.
Techniques such as Electron Beam Melting (EBM), Laser Beam Melting (LBM), and Stereolithography (SLA) enable the production of complex geometries tailored to individual patient anatomies. These advancements facilitate the creation of patient-specific implants, particularly in orthopedic, dental, and cranio-maxillofacial applications, leading to improved surgical outcomes and reduced recovery times.
This technology allows for the creation of custom-designed implants that are tailored to the specific anatomy of an individual, which has proven to be particularly beneficial in complex surgeries. The ability to print patient-specific implants ensures a perfect fit, reducing the chances of complications and improving recovery times. 3D printing, also known as additive manufacturing, enables the layer-by-layer construction of implants using a range of materials such as titanium, polymers, ceramics, and biodegradable materials, allowing for designs that were previously impossible to achieve using traditional manufacturing methods.
3D printing uses imaging technologies like CT scans and MRIs to create highly customized implants that conform precisely to the patient’s unique anatomical structure. This level of customization is especially important in fields such as orthopedics, where the fit of an implant can significantly impact the patient’s mobility and comfort.
Minimally invasive robot-assisted surgery currently achieves a success rate ranging from 94% to 100%. Between 2014 and 2020, the number of open and laparoscopic surgeries declined, dropping from 38.4% to 34.4% and from 57.4% to 50.8%, respectively. In contrast, robotic surgery cases saw a significant rise, increasing from 4.2% to 14.8%.
A major factor driving the adoption of 3D printing in the medical implant sector is the increasing demand for minimally invasive surgical procedures. With 3D printing, doctors can not only create implants but also print out models of the patient’s anatomy for pre-surgical planning.
Surgeons can study these models to practice the procedure in advance, leading to more precise operations with reduced operating times. Moreover, by using 3D-printed models, surgeons can better understand the intricacies of the patient’s condition and tailor the surgical approach accordingly. This capability can reduce the need for complex operations, shorten recovery periods, and improve patient outcomes.
Another significant benefit is the reduction in manufacturing time and costs. Traditional implant manufacturing often requires long lead times for production, which can delay surgeries. In contrast, 3D printing enables rapid prototyping and faster turnaround times. This ability to produce custom implants quickly has particular relevance in emergency surgeries or cases where time is of the essence. For example, when a patient suffers a traumatic injury that requires an implant, 3D printing can provide a solution much more quickly than traditional methods.
Despite these advancements, challenges remain, including the need for regulatory approval, material limitations, and cost factors. However, as research and technology continue to progress, it is expected that 3D printing will become an integral part of the healthcare system, transforming the way medical implants are designed, produced, and implemented. The future of 3D-printed medical implants looks promising, with continuous innovation expected to address current limitations and push the boundaries of what is possible in personalized healthcare.
Key Takeaways
- In 2024, the market for 3D printing medical implants generated a revenue of US$ 2.8 billion, with a CAGR of 15.9%, and is expected to reach US$ 12.2 billion by the year 2034.
- By material, metals segment dominated the market with 54.2% share in 2024.
- Among the application segment, dental held the largest revenue share of 36.6% in 2024.
- Laser Beam Melting technology captured the maximum share in 2024 of around 43.0%.
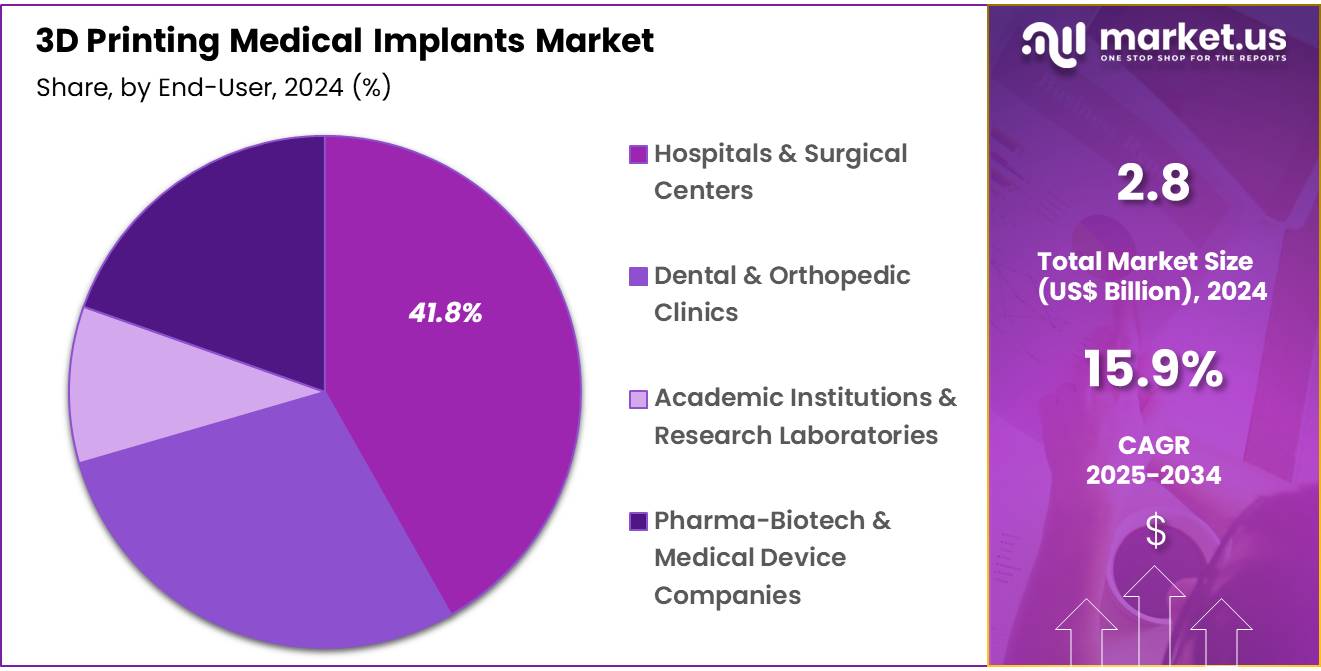
- By end-user, Hospitals & Surgical Centers was the dominant segment holding a market share of 41.8% in 2024.
- North America held the largest share of 36.6% in 2024 in the global market.
Material Analysis
The utilization of metal materials in 3D printing for medical implants has become a cornerstone of modern orthopedic and reconstructive surgery. This segment held 54.2% share in 2024. Metals such as titanium alloys, cobalt-chromium alloys, and stainless steel are favored for their exceptional mechanical properties, biocompatibility, and ability to be precisely tailored to individual patient anatomies.
Titanium and its alloys, particularly Ti-6Al-4V and Ti-6Al-7Nb, are extensively used in 3D-printed implants due to their high strength-to-weight ratio, corrosion resistance, and excellent osseointegration capabilities. These properties make titanium ideal for applications ranging from dental implants to complex spinal and joint replacements. The ability to design and manufacture patient-specific implants using 3D printing ensures a precise fit, which is crucial for the success of orthopedic surgeries.
For instance, the incorporation of bioactive glass into titanium implants has been shown to enhance bone ingrowth and accelerate healing times. Studies have demonstrated that bioactive glass coatings or doping in titanium implants can significantly improve bone-to-implant contact and bone density during the early stages of healing.
Furthermore, PEEK (polyether ether ketone) is an ideal material for producing medical devices due to its lightweight nature and high tensile strength, with properties similar to human bone. It maintains its mechanical and chemical stability even at elevated temperatures. When PEEK polymers are 3D printed, implants and instruments are produced more efficiently and with less waste compared to traditional milling methods.
Application Analysis
The dental segment dominated the 3D printing medical implants market with 36.6% share in 2024. The integration of dental in 3D printing technology has revolutionized the way dental professionals approach implantology, offering unprecedented levels of precision, customization, and efficiency. Unlike traditional methods that often rely on standardized, off-the-shelf solutions, 3D printing allows for the creation of patient-specific implants tailored to the unique anatomical structures of individuals.
A significant advancement in this field is the development of 3D-printed dental implants using materials such as zirconia and titanium. Zirconia, known for its biocompatibility and aesthetic qualities, is increasingly used in the production of crowns and bridges. Its tooth-like appearance and resistance to staining make it a preferred choice for many patients seeking natural-looking restorations. On the other hand, titanium remains a staple in dental implantology due to its strength, durability, and excellent osseointegration properties, ensuring long-term stability of the implant within the bone.
In March 2025, Lithoz introduced its new LithaBite alumina material for 3D-printed translucent orthodontic ceramic brackets, demonstrating how ceramic additive manufacturing is transforming digital dentistry.
Technology Analysis
Laser Beam Melting (LBM), a subset of Selective Laser Melting (SLM), has become the leading technology in the 3D printing of medical implants holding 43.0% share in 2024. This metal additive manufacturing process utilizes a high-powered laser to fully melt metal powders, layer by layer, to create fully dense, complex geometries that are often impossible to achieve with traditional manufacturing methods. The precision and versatility of LBM make it particularly well-suited for producing patient-specific implants in orthopedic, dental, and cranio-maxillofacial applications.
One of the primary advantages of LBM in medical implant production is its ability to create intricate internal structures, such as lattice frameworks, that enhance osseointegration the process by which bone grows into the implant. These porous structures can mimic the mechanical properties of natural bone, reducing stress shielding and promoting better integration with surrounding tissue.
For instance, studies have demonstrated that Ti6Al4V dental implants with controlled pore sizes fabricated via LBM exhibit improved cell proliferation and osteogenic activity, leading to better clinical outcomes. The capability to produce customized implants tailored to an individual patient’s anatomy is another significant benefit of LBM. By utilizing patient-specific imaging data, such as CT or MRI scans, LBM allows for the design and manufacture of implants that fit precisely within the patient’s unique anatomical structures.
End-User Analysis
Hospitals and surgical centers are the largest end-users in the 3D printing medical implants market accounting for 41.8% share in 2024, driven by the technology’s capacity to enhance surgical precision, reduce operational costs, and personalize patient care. The integration of 3D printing into clinical settings facilitates the production of patient-specific implants and surgical guides, thereby improving outcomes and expediting recovery times.
A notable example is the Bristol 3D Medical Centre in the UK, part of the North Bristol NHS Trust. This facility employs 3D printing to create custom prosthetic body parts, such as noses, ears, eyes, and fingers, for patients with facial injuries or congenital deformities. Similarly, AIIMS Bhopal in India is incorporating 3D printing technology into kidney surgeries to improve the precision and safety of complex stone removal procedures.
The Department of Urology has secured a research grant of INR 9 lakh (US$ 10,000) to develop patient-specific kidney models and customized puncture guides using 3D printing. This initiative aims to reduce complications, shorten operation times, and enhance postoperative outcomes by enabling surgeons to visualize stone locations and tailor surgical tools to each patient’s unique anatomy. In addition, Materialise has opened a metal 3D printing center in Plymouth, Michigan, specializing in personalized titanium cranio-maxillofacial implants.
Key Market Segments
By Material
- Metals
- Polymers
- Ceramics
By Application
- Dental
- Orthopedic
- Cranio-maxillofacial
- Others
By Technology
- Laser Beam Melting
- Electronic Beam Melting
- Droplet Deposition
- Others
By End-User
- Hospitals & Surgical Centers
- Dental & Orthopedic Clinics
- Academic Institutions & Research Laboratories
- Pharma-Biotech & Medical Device Companies
Drivers
Technological Advancements in 3D Printing Technologies
Technological advancements in 3D printing have significantly transformed the medical implant industry, enabling the creation of highly personalized, complex, and efficient solutions that were previously unattainable. These innovations have not only improved patient outcomes but also streamlined surgical procedures and reduced recovery times.
A notable advancement is the development of 3D-printed titanium spinal implants, which have been instrumental in treating complex spinal conditions. These implants are designed using patient-specific data obtained from CT scans, allowing for a precise fit that enhances the integration with the patient’s bone structure. The use of titanium, known for its strength and biocompatibility, ensures the durability and safety of the implants.
Furthermore, the intricate lattice structures achievable through 3D printing promote bone in-growth, leading to better long-term outcomes. In 2022, a study found that 3D-printed heart models cut surgery times by up to 25%, which improved patient results and decreased surgical risks.
By setting up 3D printing facilities on-site, hospitals can make medical tools and implants right away. This allows surgeons to get tailored solutions in hours. This change saves the wait times and improves care for each patient. Advancements in materials science have also played a crucial role in the evolution of 3D-printed medical implants. Researchers are exploring the use of diamond-based materials for implants due to their exceptional hardness, biocompatibility, and resistance to bacterial colonization.
While the fabrication of diamond materials presents challenges, ongoing developments in additive manufacturing techniques are making it increasingly feasible to produce diamond-based implants that can promote bone growth and integration. Combining advanced 3D printing, AI-driven design, and digital health solutions, restor3d, for example, leads personalized musculoskeletal reconstruction with efficient, scalable care. Restor3d operates on a data first approach to enhance implant architecture with expertise in osseointegrative biomaterials.
Restraints
Regulatory Challenges
Regulatory challenges remain a significant barrier to the widespread adoption and commercialization of 3D-printed medical implants. The innovative nature of additive manufacturing introduces complexities that existing regulatory frameworks are often ill-equipped to address, leading to delays, increased costs, and uncertainty for manufacturers.
One of the primary issues is the classification of 3D-printed medical devices. Many 3D-printed implants, especially those that are patient-specific, do not have direct counterparts in traditional manufacturing, complicating the determination of their appropriate classification. This lack of clear classification can lead to delays in the approval process and uncertainty for manufacturers regarding the regulatory requirements they must meet. Additionally, the customization inherent in 3D printing poses challenges for regulatory bodies. Each implant is often tailored to an individual patient’s anatomy, making it difficult to apply standardized testing and validation procedures.
The introduction of point-of-care manufacturing complicates the regulatory process, as healthcare facilities could become de facto manufacturers. Beitler et al. pointed out that the regulatory challenges arise from the overlap between healthcare providers and device manufacturers. Point-of-care (PoC) 3D printing facilities are urged to comply with quality system regulations such as ISO 13485 and FDA Title 21 standards to maintain the safety and efficacy of their devices.
These standards call for comprehensive quality management systems, validated internal processes, and thorough documentation, including mechanical testing and risk assessments. While the FDA’s current regulations apply to 3D-printed devices, the agency is collaborating with stakeholders to create specific guidelines for PoC 3D printing.
Opportunities
Growth in CAD/CAM technology
The integration of Computer-Aided Design (CAD) and Computer-Aided Manufacturing (CAM) technologies has significantly advanced the field of 3D printing for medical implants, offering unprecedented precision and customization. By leveraging patient-specific imaging data, CAD software enables the design of implants that closely match an individual’s unique anatomical structures. This level of personalization is particularly beneficial in complex surgeries, where the fit and functionality of the implant are critical to the patient’s recovery and quality of life.
Once the implant design is finalized using CAD, CAM technology facilitates the actual manufacturing process. CAM software translates the digital design into machine instructions, guiding 3D printers or CNC machines to produce the implant with high accuracy. This seamless transition from design to manufacturing ensures that the final product aligns precisely with the intended specifications, reducing the risk of errors and the need for adjustments during surgery.
The adoption of CAD/CAM technologies in medical implant manufacturing has led to several notable advancements. For instance, the development of patient-specific cranial implants has been made possible through the use of CAD/CAM systems. These implants are designed based on detailed imaging data, allowing for a perfect fit and improved surgical outcomes. Similarly, in dental applications, CAD/CAM technologies have revolutionized the production of crowns, bridges, and dentures, enabling faster turnaround times and enhanced patient satisfaction.
PMMA (Polymethyl Methacrylate) is a durable, biocompatible material commonly used for temporary restorations, offering the strength necessary to withstand daily oral functions. The digital CAD/CAM workflow optimizes the production of PMMA Implant Provisionals, reducing the period patients are without teeth or depend on temporary solutions. 3D-printed CAD/CAM PMMA Provisionals enable patients to assess the appearance and functionality of their future implant-supported teeth, ensuring comfort and satisfaction before proceeding with the final restoration.
Impact of Macroeconomic / Geopolitical Factors
Macroeconomic and geopolitical factors significantly influence the 3D printing medical implants market, affecting production costs, supply chain dynamics, and market accessibility. For instance, inflationary pressures and rising material costs have impacted the manufacturing sector globally, including 3D printing of medical implants. As the price of raw materials like titanium and cobalt-chromium alloys rises, manufacturers face challenges in maintaining cost-effective production while ensuring the quality and safety of implants. This can lead to higher prices for 3D-printed medical implants, potentially limiting access in price-sensitive regions.
The ongoing geopolitical tensions, such as the Iran-Israel conflict, also have a profound impact on the 3D printing market. Disruptions in the Middle East, particularly in the wake of ongoing hostilities, can destabilize supply chains and affect the availability of essential raw materials. For example, the conflict may interfere with global supply lines for key components or materials, causing delays in manufacturing processes or leading to scarcity in essential metals used in 3D-printed medical implants. Additionally, the uncertainty surrounding the region’s stability can discourage foreign investment in countries directly affected by the conflict.
Latest Trends
Integration with Digital Health Technologies
The integration of 3D printing with digital health technologies is revolutionizing the medical implant sector, ushering in an era of personalized and data-driven healthcare. This convergence enables the creation of patient-specific implants and facilitates real-time monitoring of recovery, thereby enhancing surgical outcomes and patient satisfaction.
A notable example is the development of the TrabeculeX Continuum by Xenco Medical, which combines a regenerative biomaterial implant with a remote therapeutic monitoring app. This system not only promotes bone growth through its innovative design but also tracks patient recovery via virtual physical therapy sessions and asynchronous communication, creating a seamless continuum from surgery to rehabilitation.
Furthermore, advancements in augmented reality (AR) are enhancing the precision of surgical procedures. For instance, a study demonstrated that using AR guidance during CT-guided interventions reduced the number of needle passes by 54% and decreased radiation exposure by 41%, highlighting the potential of AR in improving surgical efficiency and safety.
The integration of artificial intelligence (AI) with 3D printing technologies has further enhanced the design and manufacturing processes of medical implants. AI algorithms can analyze vast amounts of patient data to optimize implant designs, ensuring better compatibility and performance. At Fraunhofer IGD, a research scientist is advancing the 3D printing of eye implants. Using AI to design implants from optical coherence tomography (OCT) images, researchers can produce custom prosthetic eyes that precisely match the patient’s anatomy.
Regional Analysis
North America is leading the 3D Printing Medical Implants Market
North America held the largest share of the 3D printing medical implants market, largely due to its advanced healthcare infrastructure, significant investment in research and development, and early adoption of cutting-edge medical technologies. The region, particularly the United States, continues to dominate the market with its focus on personalized medicine and the increasing demand for customized healthcare solutions.
In the United States, surgeons perform around 700,000 total knee replacements annually, as reported by the American Academy of Orthopaedic Surgeons (AAOS). In 2023, Materialise produced over 7,000 patient-specific implants and surgical guides in the U.S. and opened a new metal 3D printing facility in Michigan. Materialise also noted a significant adoption of its FDA-cleared cranial maxillofacial patient-specific implants in 2023 for patients with cancer and trauma.
The Asia Pacific region is expected to experience the highest CAGR during the forecast period
Asia-Pacific region is experiencing the fastest growth in the 3D printing medical implants market. The rapid expansion of healthcare infrastructure, coupled with a large and aging population, is driving the demand for affordable and customized medical solutions. Countries like China, India, and Japan are at the forefront of this growth, where increasing healthcare investments and government initiatives are making 3D printing technologies more accessible.
In China, for example, the government’s push for technological innovation has facilitated advancements in medical 3D printing, positioning the country as a leader in the field. Similarly, India’s rising demand for affordable healthcare options and the growing adoption of 3D printing in medical applications have positioned it as one of the fastest-growing markets in the region. Spinal fusion surgery in India has a success rate of 85% to 95%, with significant pain relief and improved mobility for patients. Additionally, India has become a leading destination for orthognathic surgery, with a success rate exceeding 98% for jaw deformity corrections.
Key Regions and Countries
North America
- US
- Canada
Europe
- Germany
- France
- The UK
- Spain
- Italy
- Russia
- Netherland
- Rest of Europe
Asia Pacific
- China
- Japan
- South Korea
- India
- Australia
- New Zealand
- Singapore
- Thailand
- Vietnam
- Rest of APAC
Latin America
- Brazil
- Mexico
- Rest of Latin America
Middle East & Africa
- South Africa
- Saudi Arabia
- UAE
- Rest of MEA
Key Players Analysis
The 3D printing medical implants market is characterized by a dynamic and evolving competitive landscape, with a diverse range of players from established medical device companies to specialized 3D printing firms and emerging startups. This broad competition drives rapid technological advancements and fosters the development of personalized medical solutions. Specialized 3D printing companies have carved out significant niches. Companies like 3D Systems and Stratasys have been pivotal in providing 3D printing technologies that cater to the medical sector.
These companies have developed specialized 3D printers capable of producing medical devices such as customized implants and surgical guides, helping medical professionals meet the needs of patients in a more precise manner. Additionally, Materialise, a company known for its expertise in medical applications of 3D printing, offers software and services that allow healthcare providers to design and produce patient-specific medical implants with high precision, facilitating the adoption of 3D printing in clinical settings.
Top Key Players
- 3D Bioprinting Solutions
- BioBots
- ANDREAS STIHL AG & Co. KG
- Aspect Biosystems
- Formlabs
- Medprin
- Stratasys Ltd.
- Organovo Holdings Inc.
- Rokit
- Materialise N.V.
- Cyfuse Biomedical K.K.
- EnvisionTEC
- Renishaw PLC
- Arcam AB
- BICO Group
Recent Developments
- In March 2025, PHC Corporation and Cyfuse Biomedical K.K. announced a new production technology for regenerative medicine and cell therapy. Their collaboration enables real-time monitoring of the quality of 3D cell constructs containing living cells in three-dimensional structures, addressing a challenge in production and ensuring more stable production of high-quality cell products.
- In February 2025, Stratasys Ltd. revealed that its Stratasys Direct manufacturing facility in Tucson, Arizona, received ISO 13485 certification, a globally recognized standard for quality management systems in medical device manufacturing. The company plans to extend this certification to its facilities in Texas and Minnesota.
- In April 2024, 3D Systems announced that its VSP® PEEK Cranial Implant, a patient-specific solution, received 510(k) clearance from the FDA. The VSP PEEK Cranial Implant includes a complete FDA-cleared workflow, which incorporates segmentation and 3D modeling software, the 3D Systems EXT 220 MED 3D printer, Evonik VESTAKEEP® i4 3DF PEEK (polyetheretherketone), and a pre-defined production process.
Report Scope
Report Features Description Market Value (2024) US$ 2.8 Billion Forecast Revenue (2034) US$ 12.2 Billion CAGR (2025-2034) 15.9% Base Year for Estimation 2024 Historic Period 2020-2023 Forecast Period 2025-2034 Report Coverage Revenue Forecast, Market Dynamics, COVID-19 Impact, Competitive Landscape, Recent Developments Segments Covered By Material (Metals, Polymers, Ceramics), By Application (Dental, Orthopedic, Cranio-maxillofacial, Others), By Technology (Laser Beam Melting, Electronic Beam Melting, Droplet Deposition, Others), By End-User (Hospitals & Surgical Centers, Dental & Orthopedic Clinics, Academic Institutions & Research Laboratories, Pharma-Biotech & Medical Device Companies) Regional Analysis North America – US, Canada; Europe – Germany, France, The UK, Spain, Italy, Russia, Netherlands, Rest of Europe; Asia Pacific – China, Japan, South Korea, India, Australia, New Zealand, Singapore, Thailand, Vietnam, Rest of APAC; Latin America – Brazil, Mexico, Rest of Latin America; Middle East & Africa – South Africa, Saudi Arabia, UAE, Rest of MEA Competitive Landscape 3D Bioprinting Solutions, BioBots, ANDREAS STIHL AG & Co. KG, Aspect Biosystems, Formlabs, Medprin, Stratasys Ltd., Organovo Holdings Inc., Rokit, Materialise N.V., Cyfuse Biomedical K.K., EnvisionTEC, Renishaw PLC, Arcam AB, BICO Group Customization Scope Customization for segments, region/country-level will be provided. Moreover, additional customization can be done based on the requirements. Purchase Options We have three licenses to opt for: Single User License, Multi-User License (Up to 5 Users), Corporate Use License (Unlimited User and Printable PDF)  3D Printing Medical Implants MarketPublished date: July 2025add_shopping_cartBuy Now get_appDownload Sample
3D Printing Medical Implants MarketPublished date: July 2025add_shopping_cartBuy Now get_appDownload Sample -
-
- 3D Bioprinting Solutions
- BioBots
- ANDREAS STIHL AG & Co. KG
- Aspect Biosystems
- Formlabs
- Medprin
- Stratasys Ltd.
- Organovo Holdings Inc.
- Rokit
- Materialise N.V.
- Cyfuse Biomedical K.K.
- EnvisionTEC
- Renishaw PLC
- Arcam AB
- BICO Group










