Global Postpartum Hemorrhage Management Devices MarketBy Product Type (Intrauterine Balloon Tamponade, Uterine Compression Devices, Resuscitators, Vacuum-Induced Hemorrhage Control Systems, Intrauterine Vacuum Devices, Others) By Application (Vaginal Delivery, Cesarean Section, Termination of Pregnancy, Others) By End-User (Hospitals, Ambulatory Surgical Centers, Maternity and Fertility Clinics, Others), Region and Companies – Industry Segment Outlook, Market Assessment, Competition Scenario, Trends and Forecast 2026-2035
- Published date: May 2026
- Report ID: 185854
- Number of Pages: 255
- Format:
-
keyboard_arrow_up
Quick Navigation
Report Overview
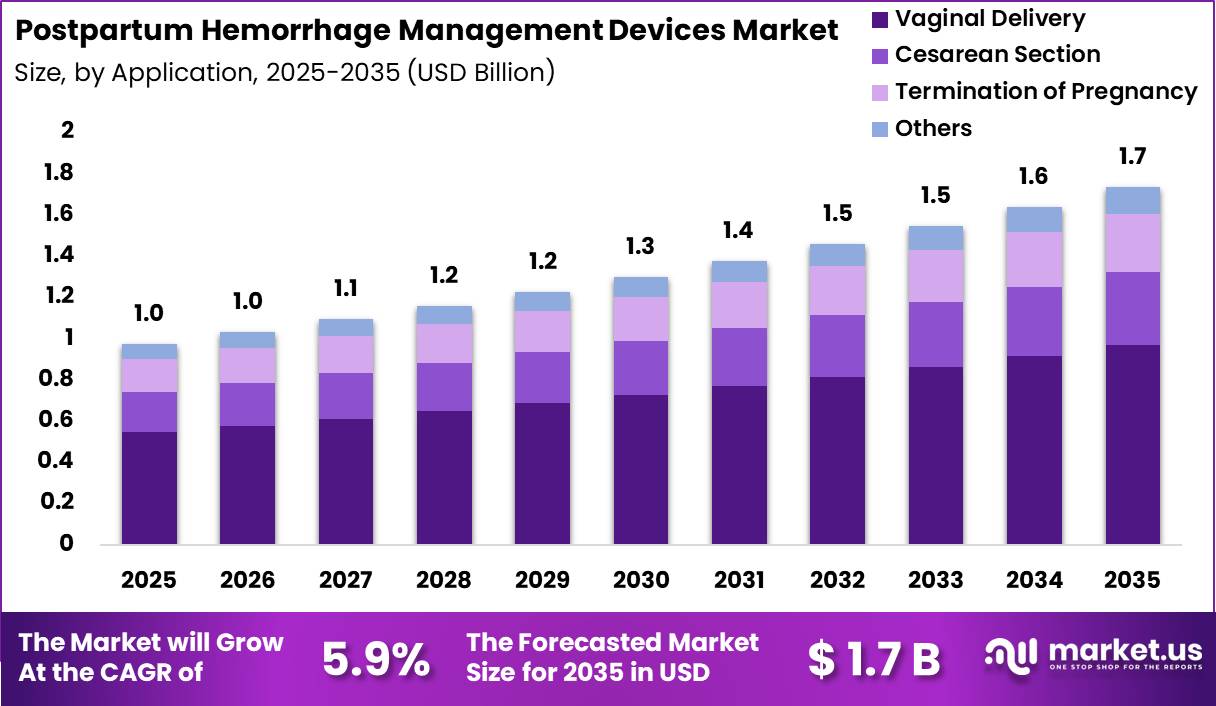
Global Postpartum Hemorrhage Management Devices Market size is expected to be worth around US$ 1.7 Billion by 2035 from US$ 1.0 Billion in 2025, growing at a CAGR of 5.9% during the forecast period from 2026 to 2035. In 2025, North America led the market, achieving over 34.4% share with a revenue of US$ 0.3 Billion.
The global Postpartum Hemorrhage Management Devices Market is witnessing significant growth due to the increasing burden of postpartum hemorrhage (PPH), which remains one of the leading causes of maternal mortality worldwide.
Postpartum hemorrhage, commonly defined as blood loss of 500 mL or more within 24 hours after childbirth, has become a major public health concern, particularly in low- and middle-income countries where access to emergency obstetric care remains limited. According to the World Health Organization (WHO), nearly 14 million women experience PPH annually, resulting in approximately 70,000 maternal deaths each year globally.
The growing incidence of childbirth-related complications such as uterine atony, retained placenta, anemia, and prolonged labor has increased the demand for advanced hemorrhage control technologies. Healthcare facilities are increasingly adopting devices such as uterine balloon tamponades, non-pneumatic anti-shock garments, suction-based hemorrhage control systems, and minimally invasive surgical instruments to improve maternal outcomes and reduce emergency hysterectomy procedures.
In addition, the rising number of institutional deliveries and expanding access to maternal healthcare services are contributing to the increased deployment of PPH management devices across hospitals and maternity centers.
Government agencies and international healthcare organizations are also strengthening efforts to reduce maternal mortality through awareness campaigns, funding initiatives, and clinical guidelines. In 2023, the World Health Organization (WHO) launched the “Roadmap to Combat Postpartum Hemorrhage between 2023 and 2030,” aimed at improving prevention, diagnosis, and treatment strategies globally. Furthermore, WHO estimates indicate that obstetric hemorrhage accounts for nearly 27% of maternal deaths worldwide, highlighting the urgent need for effective management solutions.
Technological advancements, including AI-enabled predictive monitoring systems, portable emergency devices, and connected maternal care platforms, are further supporting market expansion. Increasing investments in maternal healthcare infrastructure, particularly in Asia-Pacific and Africa, are expected to create substantial growth opportunities for manufacturers operating in the postpartum hemorrhage management devices industry over the coming years.
Key Takeaways
- Market size: Global Postpartum Hemorrhage Management Devices Market size is expected to be worth around US$ 1.7 Billion by 2035 from US$ 1.0 Billion in 2025.
- Market Share: The market growing at a CAGR of 5.9% during the forecast period from 2026 to 2035.
- Product Type Analysis: Intrauterine Balloon Tamponade emerged as the leading segment, accounting for 48.5% of the global market share in 2025.
- Application Analysis: The Vaginal Delivery dominated the market with a 55.9% share in 2025.
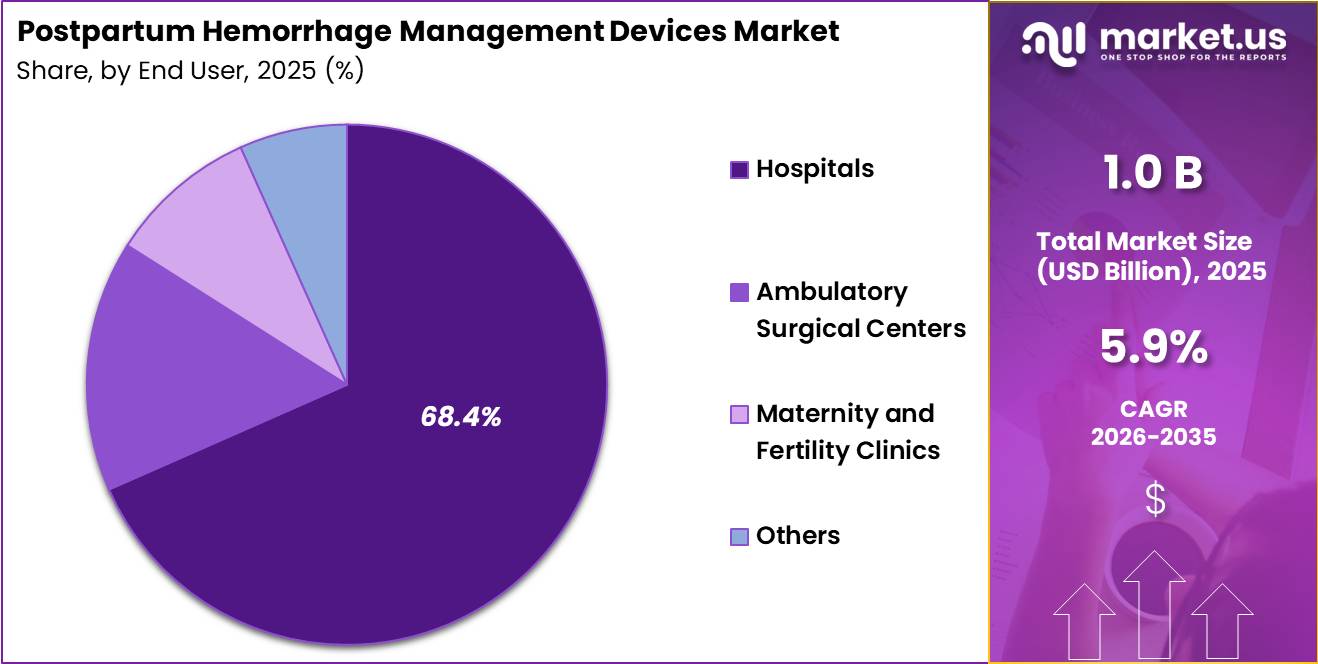
- End-User Analysis: The Hospitals accounted for the largest market share of 52.8% in 2025.
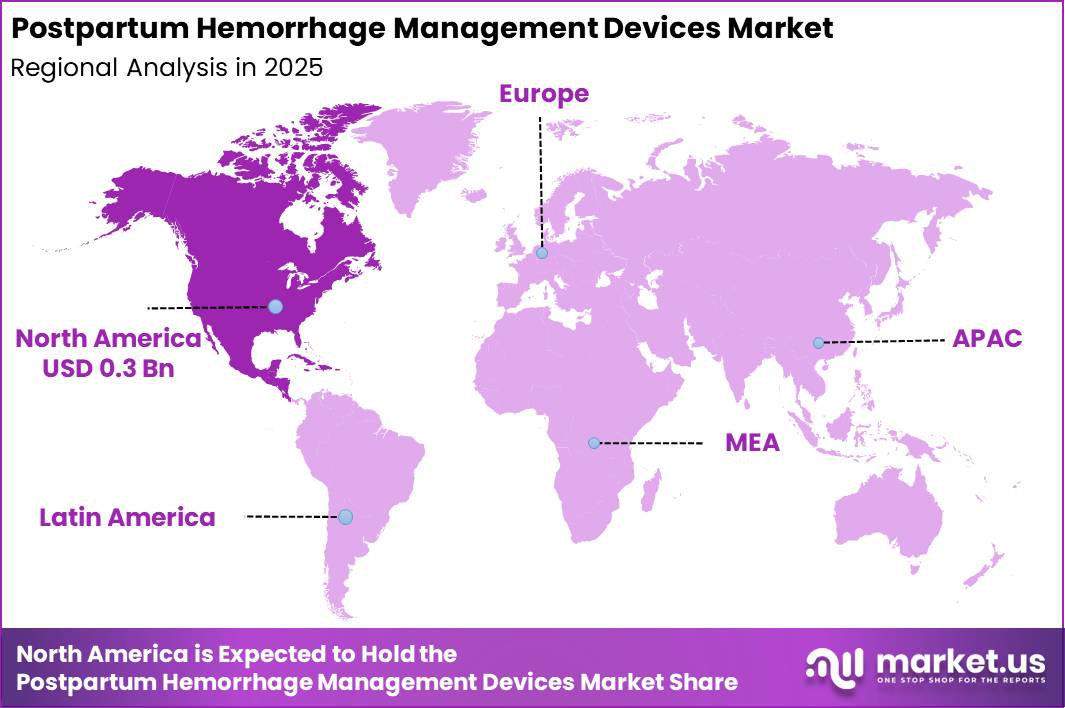
- Regional Analysis : North America dominated the global Postpartum Hemorrhage Management Devices Market in 2025, accounting for over 34.4% of the total market share and generating revenue of approximately US$ 0.3 billion.
Product Type Analysis
The product type segment of the Postpartum Hemorrhage Management Devices Market is categorized into Intrauterine Balloon Tamponade, Uterine Compression Devices, Resuscitators, Vacuum-Induced Hemorrhage Control Systems, Intrauterine Vacuum Devices, and Others. Intrauterine Balloon Tamponade emerged as the leading segment, accounting for 48.5% of the global market share in 2025.
The dominance of this segment is attributed to the increasing adoption of minimally invasive hemorrhage control techniques, high clinical success rates, and widespread use in emergency obstetric care. These devices are extensively utilized in hospitals and maternity centers owing to their rapid bleeding control capabilities and reduced requirement for surgical intervention.
Uterine Compression Devices are witnessing steady adoption due to their effectiveness in managing severe uterine atony and controlling excessive bleeding during delivery procedures. Resuscitators also hold a significant share as they are essential in emergency maternal care for stabilizing patients experiencing critical blood loss.
Vacuum-Induced Hemorrhage Control Systems and Intrauterine Vacuum Devices are gaining traction owing to technological advancements and increasing preference for innovative hemorrhage management approaches. The Others segment includes surgical accessories and adjunctive hemorrhage management tools used in specialized obstetric procedures.
Application Analysis
Based on application, the Postpartum Hemorrhage Management Devices Market is segmented into Vaginal Delivery, Cesarean Section, Termination of Pregnancy, and Others. Vaginal Delivery dominated the market with a 55.9% share in 2025.
The large share of this segment is primarily driven by the high global volume of vaginal births and the elevated incidence of postpartum hemorrhage associated with prolonged labor and uterine atony. Increasing awareness regarding maternal healthcare safety and rising implementation of preventive hemorrhage management protocols in delivery units further support segment growth.
The Cesarean Section segment represents a substantial portion of the market due to the growing number of surgical deliveries worldwide and the increased risk of excessive blood loss during cesarean procedures. Healthcare providers are increasingly utilizing advanced hemorrhage control devices during cesarean deliveries to reduce maternal complications and improve surgical outcomes.
The Termination of Pregnancy segment is also experiencing moderate growth owing to the requirement for effective bleeding management during gynecological procedures. Additionally, the Others segment includes postpartum complications and trauma-related obstetric interventions where hemorrhage management devices are routinely utilized to ensure maternal stabilization and recovery.
End-User Analysis
Based on end user, the Postpartum Hemorrhage Management Devices Market is segmented into Hospitals, Ambulatory Surgical Centers, Maternity and Fertility Clinics, and Others. Hospitals accounted for the largest market share of 52.8% in 2025.
The dominance of hospitals is attributed to the high patient inflow for childbirth procedures, availability of advanced obstetric care infrastructure, and presence of skilled healthcare professionals capable of managing severe postpartum complications. Hospitals also serve as primary centers for emergency maternal care, contributing significantly to the demand for postpartum hemorrhage management devices.
Ambulatory Surgical Centers are gaining adoption due to the increasing preference for cost-effective outpatient gynecological and obstetric procedures. These facilities are increasingly integrating advanced hemorrhage control technologies to enhance procedural safety and reduce postoperative complications.
Maternity and Fertility Clinics also represent a growing segment owing to rising fertility treatments, increasing institutional deliveries, and expanding maternal healthcare services across developing economies. The Others segment includes community healthcare centers and specialty women’s care facilities that utilize hemorrhage management devices for emergency obstetric interventions and maternal stabilization procedures.
Key Market Segments
By Product Type
- Intrauterine Balloon Tamponade
- Uterine Compression Devices
- Resuscitators
- Vacuum-Induced Hemorrhage Control Systems
- Intrauterine Vacuum Devices
- Others
By Application
- Vaginal Delivery
- Cesarean Section
- Termination of Pregnancy
- Others
By End-User
- Hospitals
- Ambulatory Surgical Centers
- Maternity and Fertility Clinics
- Others
Driving Factors
The Postpartum Hemorrhage Management Devices market is primarily driven by the alarming global prevalence of PPH, which affects approximately 14 million women annually and accounts for 70,000 maternal deaths worldwide. According to WHO, PPH represents over 20% of all maternal deaths globally.
In India, the Ministry of Health and Family Welfare reports that PPH accounts for 38% of maternal deaths, exacerbated by widespread anemia among pregnant women. The pregnancy-related mortality ratio for hemorrhage in the United States stands at 1.94 per 100,000 live births, with postpartum hemorrhage representing 21.2% of hemorrhage-related deaths.
India’s Maternal Mortality Ratio has declined from 130 per 100,000 live births in 2014-16 to 97 per 100,000 live births in 2018-20. Government initiatives under National Health Mission’s RMNCAH+N strategy and increasing institutional deliveries from 79% in 2015-16 to 89% in 2019-21 are accelerating device adoption. The CDC reports hemorrhage causes 8.7% of overall pregnancy-related deaths.
Trending Factors
Technological innovation is transforming the Postpartum Hemorrhage Management Devices market through integration of digital monitoring systems and advanced tamponade technologies. The WHO recommends intrauterine balloon tamponade devices for PPH treatment when uterotonics fail, supporting widespread clinical adoption.
India’s National Health Mission developed comprehensive PPH management protocols incorporating uterine tamponade devices, non-pneumatic anti-shock garments, and advanced resuscitation equipment. The E-MOTIVE intervention trial demonstrated that early detection bundled with treatment reduced PPH exceeding 500 ml from 16.7% to 8.5%, and severe PPH exceeding 1000 ml from 4.3% to 1.6%. WHO’s 2023-2030 Roadmap to Combat Postpartum Haemorrhage emphasizes prevention, detection, and treatment standardization.
The PMSMA Portal developed by India’s Ministry of Health monitors antenatal care services, high-risk pregnancy tracking, and emergency preparedness. Training programs for healthcare providers on device deployment and WHO’s certification courses on bleeding management are expanding accessibility to advanced PPH interventions across healthcare facilities globally.
Restraining Factors
Limited healthcare infrastructure and trained personnel availability significantly restrain the Postpartum Hemorrhage Management Devices market expansion, particularly in low-resource settings. The Ministry of Health and Family Welfare India identifies that 46% of counties in certain regions lack practicing obstetrician-gynecologists, creating critical workforce gaps.
Training requirements for advanced hemorrhage devices span 8 to 12 hours of hands-on instruction plus periodic competency audits, which rural practitioners struggle to accommodate. India’s PPH guidance notes emphasize that inadequate human resources, logistical supply chain issues, and lack of on-the-job training limit proper implementation of Active Management of Third Stage of Labour protocols.
WHO reports that most PPH-associated deaths occur in low and middle-income countries where 25 to 43% of maternal deaths result from PPH due to poor access to modern medical devices. The Indian government highlights delays in emergency obstetric care access due to poor communications, referral system failures, and shortages of necessary drugs and equipment. Visual estimation methods underestimate blood loss by 33 to 50% compared to gold-standard photo-spectrometry.
OpportunityOpportunity
Expanding healthcare infrastructure in emerging markets presents substantial opportunities for Postpartum Hemorrhage Management Devices adoption. India’s National Health Mission supports states through annual Programme Implementation Plans to reduce maternal mortality, with institutional births reaching 100% in Kerala, Goa, Lakshadweep, Puducherry, and Tamil Nadu.
The proportion of pregnant women receiving antenatal care in the first trimester increased from 59% in 2015-16 to 70% in 2019-21, while women receiving four or more ANC visits rose from 51% to 59% nationally. WHO’s global action plan emphasizes uterine balloon tamponade device deployment at primary health center levels with trained medical officers present. India’s Extended PMSMA strategy launched in January 2022 provides financial incentivization for high-risk pregnancy tracking until safe delivery.
The government recommendation for blood storage units within maternity wings of high-volume tertiary care facilities creates infrastructure demand. Cost-effective uterine balloon tamponade alternatives like condom catheters offer affordable solutions for resource-limited settings. India’s commitment to UN Sustainable Development Goal targets for MMR at 70 per 100,000 live births by 2030 drives investment.
Regional Analysis
North America dominated the global Postpartum Hemorrhage Management Devices Market in 2025, accounting for over 34.4% of the total market share and generating revenue of approximately US$ 0.3 billion. The regional market growth is primarily driven by the increasing prevalence of postpartum hemorrhage cases, rising maternal healthcare expenditure, and the strong presence of advanced healthcare infrastructure across the United States and Canada.
The adoption of technologically advanced hemorrhage management devices, including uterine balloon tamponade systems and non-pneumatic anti-shock garments, has increased significantly due to growing awareness regarding maternal mortality prevention and emergency obstetric care.
Furthermore, supportive government initiatives and favorable reimbursement frameworks have contributed to the widespread utilization of postpartum hemorrhage management devices in hospitals and maternity care centers. The presence of major market players, coupled with continuous investments in research and development activities, has further accelerated product innovation and market penetration in the region.
In addition, the increasing number of institutional deliveries and the availability of skilled healthcare professionals are supporting market expansion. Rising emphasis on early diagnosis and rapid intervention for obstetric complications is also expected to strengthen demand for postpartum hemorrhage management devices across North America during the forecast period.
Key Regions and Countries
North America
- The US
- Canada
Europe
- Germany
- France
- The U.K.
- Italy
- Spain
- Russia & CIS
- Rest of Europe
Asia Pacific
- China
- India
- Japan
- South Korea
- ASEAN
- Australia & New Zealand
- Rest of Asia Pacific
Middle East & Africa
- GCC
- South Africa
- Rest of Middle East & Africa
Latin America
- Brazil
- Mexico
- Rest of Latin America
Key Players Analysis
The Postpartum Hemorrhage Management Devices Market is characterized by intense competition among global medical device manufacturers focusing on product innovation, strategic collaborations, and geographic expansion.
Major companies operating in the market include Becton, Dickinson and Company, Cook Medical, Teleflex Incorporated, Utah Medical Products, Inc., and Organon & Co.. These players are actively investing in advanced postpartum hemorrhage management technologies, particularly uterine balloon tamponade systems and vacuum-induced hemorrhage control devices, to strengthen their market presence.
Becton, Dickinson and Company holds a leading market position due to its extensive women’s health portfolio, strong global distribution network, and continuous investments in research and development. The company has strengthened its market position through acquisitions and innovative product launches targeting rapid hemorrhage control solutions.
Cook Medical is recognized for its focus on emergency obstetric care products and customized hemorrhage management kits designed for hospital settings. Meanwhile, Teleflex Incorporated emphasizes minimally invasive and clinically efficient solutions to improve maternal healthcare outcomes.
In addition, emerging companies and regional manufacturers are increasingly entering the market with cost-effective products, particularly for low-resource healthcare settings. Strategic partnerships with healthcare organizations, government maternal health programs, and expanding presence in emerging economies are expected to remain key competitive strategies among market participants during the forecast period.
Market Key Players
- Medtronic plc
- Becton, Dickinson and Company
- Teleflex Incorporated
- CooperSurgical, Inc.
- Cook Medical, LLC
- Utah Medical Products, Inc.
- Clinical Innovations, LLC
- Integra LifeSciences Holdings Corporation
- Inpress Technologies, Inc.
- Dornier MedTech GmbH
- Olympus Corporation
- Karl Storz SE & Co. KG
- Ethicon, Inc. (Johnson & Johnson)
- Zinpro Corporation
- Cryo-Cell International, Inc.
- Others
Recent Developments
- Utah Medical Products, Inc. (February 2025): Utah Medical Products reported expanded adoption of its Bakri postpartum balloon in high-risk obstetric centers as updated PPH protocols in North America and Europe elevated uterine tamponade balloons to front-line therapy, reinforcing the company’s positioning in guideline-driven PPH management pathways.
- Becton, Dickinson and Company (BD) (March 2025): BD strengthened its role in postpartum hemorrhage treatment by emphasizing uterine balloon tamponade and related surgical instruments within its broader women’s health portfolio, supported by rising global PPH incidence and growing hospital demand for standardized device kits that integrate seamlessly into emergency obstetric care workflows.
- Medtronic plc (April 2025): Medtronic leveraged its maternal health footprint by aligning advanced monitoring and surgical technologies with postpartum hemorrhage management bundles, as hospitals prioritized comprehensive obstetric solutions that combine rapid detection, minimally invasive intervention, and post-procedure monitoring to reduce maternal morbidity.
- Cook Medical LLC (May 2025): Cook Medical continued to expand access to its obstetric and gynecologic surgical instruments used in refractory PPH cases, with hospitals in developed markets consolidating purchasing contracts around vendors that can support both conservative and procedural interventions within a single postpartum hemorrhage device portfolio.
Report Scope
Report Features Description Market Value (2025) US$ 1.0 Billion Forecast Revenue (2035) US$ 1.7 Billion CAGR (2026-2035) 5.9% Base Year for Estimation 2025 Historic Period 2020-2024 Forecast Period 2026-2035 Report Coverage Revenue Forecast, Market Dynamics, Competitive Landscape, Recent Developments Segments Covered By Product Type (Intrauterine Balloon Tamponade, Uterine Compression Devices, Resuscitators, Vacuum-Induced Hemorrhage Control Systems, Intrauterine Vacuum Devices, Others) By Application (Vaginal Delivery, Cesarean Section, Termination of Pregnancy, Others) By End-User (Hospitals, Ambulatory Surgical Centers, Maternity and Fertility Clinics, Others) Regional Analysis North America – The US, Canada; Europe – Germany, France, U.K., Italy, Spain, Russia & CIS, Rest of Europe; Asia Pacific – China, India, Japan, South Korea, ASEAN, Australia & New Zealand, Rest of Asia Pacific; Middle East & Africa – GCC, South Africa, Rest of Middle East & Africa; Latin America – Brazil, Mexico, Rest of Latin America Competitive Landscape Medtronic plc, Becton, Dickinson and Company, Teleflex Incorporated, CooperSurgical, Inc., Cook Medical, LLC, Utah Medical Products, Inc., Clinical Innovations, LLC, Integra LifeSciences Holdings Corporation, Inpress Technologies, Inc., Dornier MedTech GmbH, Olympus Corporation, Karl Storz SE & Co. KG, Ethicon, Inc. (Johnson & Johnson), Zinpro Corporation, Cryo-Cell International, Inc., Others, Customization Scope Customization for segments, region/country-level will be provided. Moreover, additional customization can be done based on the requirements. Purchase Options We have three licenses to opt for: Single User License, Multi-User License (Up to 5 Users), Corporate Use License (Unlimited User and Printable PDF)  Postpartum Hemorrhage Management Devices MarketPublished date: May 2026add_shopping_cartBuy Now get_appDownload Sample
Postpartum Hemorrhage Management Devices MarketPublished date: May 2026add_shopping_cartBuy Now get_appDownload Sample -
-
- Medtronic plc
- Becton, Dickinson and Company
- Teleflex Incorporated
- CooperSurgical, Inc.
- Cook Medical, LLC
- Utah Medical Products, Inc.
- Clinical Innovations, LLC
- Integra LifeSciences Holdings Corporation
- Inpress Technologies, Inc.
- Dornier MedTech GmbH
- Olympus Corporation
- Karl Storz SE & Co. KG
- Ethicon, Inc. (Johnson & Johnson)
- Zinpro Corporation
- Cryo-Cell International, Inc.
- Others










