Global Generic Sterile Injectable Market By Product Type (Monoclonal Antibodies, Antibiotics, Insulin, and Others), By Application (Cancer, Cardiovascular Disease, Diabetes, and Others), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies, and Others), Region and Companies – Industry Segment Outlook, Market Assessment, Competition Scenario, Trends and Forecast 2025-2034
- Published date: July 2025
- Report ID: 153936
- Number of Pages: 383
- Format:
-
keyboard_arrow_up
Quick Navigation
Report Overview
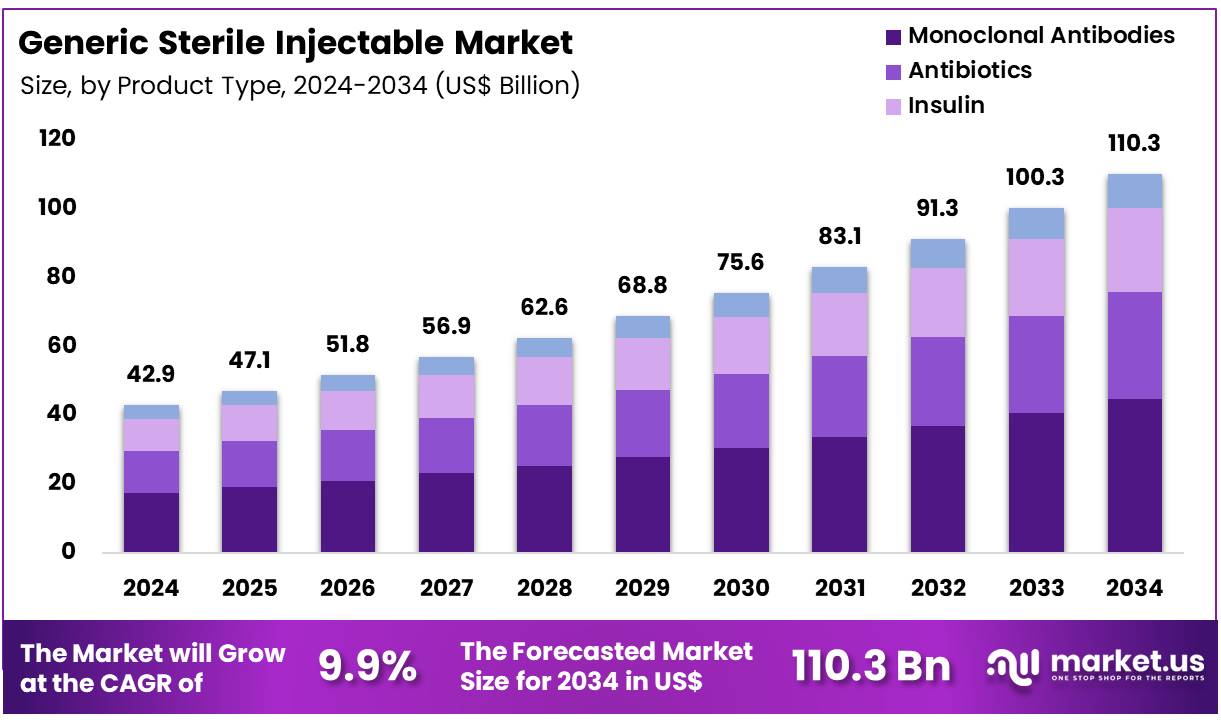
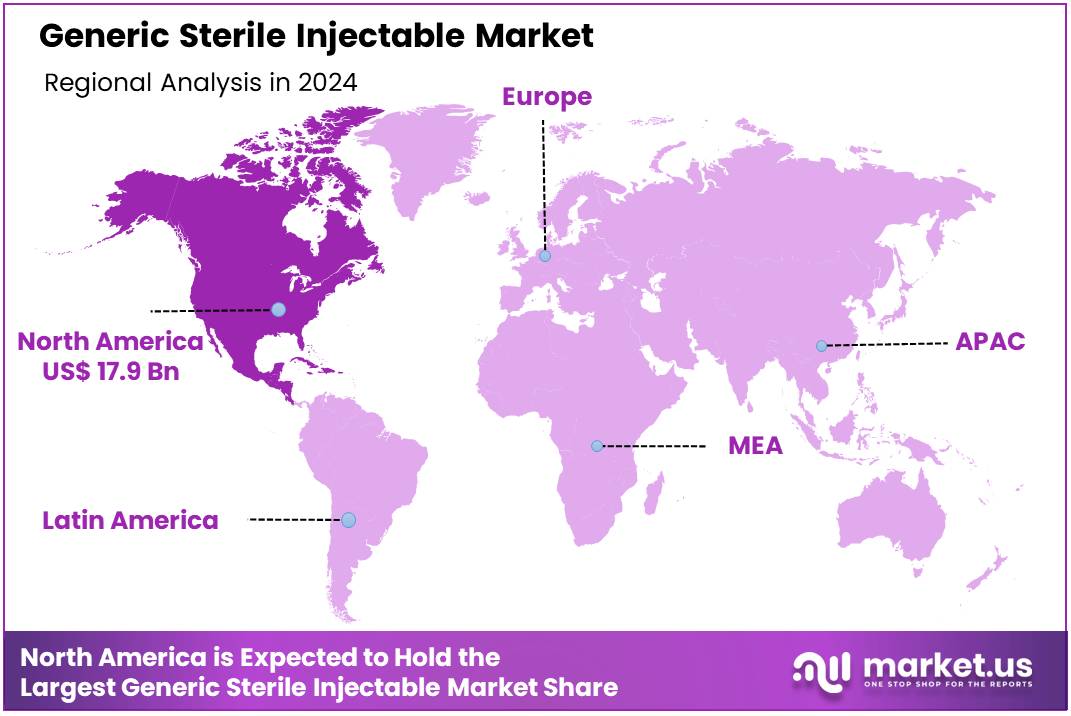
Global Generic Sterile Injectable Market size is expected to be worth around US$ 110.3 Billion by 2034 from US$ 42.9 Billion in 2024, growing at a CAGR of 9.9% during the forecast period 2025 to 2034. In 2024, North America led the market, achieving over 41.7% share with a revenue of US$ 17.9 Billion.
Increasing demand for cost-effective alternatives to branded pharmaceuticals and the rising prevalence of chronic diseases are driving the growth of the generic sterile injectable market. Sterile injectables are critical for the treatment of various conditions, including cancer, diabetes, infections, and autoimmune diseases, and they are commonly used in both hospital and outpatient settings. Generic sterile injectables offer an affordable option to patients while maintaining the same quality and efficacy as branded versions, contributing to the market’s expansion.
The market also benefits from the increasing focus on biologics and biosimilars, as many biologic drugs transition to generic forms once their patents expire, creating substantial opportunities for growth. Additionally, the rise in the number of patients requiring long-term treatments for conditions such as rheumatoid arthritis and multiple sclerosis further drives the demand for sterile injectable products. In May 2024, Cipla confirmed that it had received approval from the US Health Department to commercialize a generic version of a medication used to treat acromegaly and other conditions.
The Mumbai-based manufacturer announced it had received the final approval from the US Food and Drug Administration (USFDA) to offer Lanreotide injections in various dosages. This highlights a key trend in the market where increasing numbers of generic injectable versions of high-cost biologics are entering the market, offering patients more affordable treatment options. The market is also witnessing advances in the manufacturing processes of sterile injectables, improving efficiency and reducing production costs. As healthcare systems continue to focus on reducing pharmaceutical expenditure, the demand for generic sterile injectables is expected to grow, providing both economic and clinical benefits to healthcare providers and patients alike.
Key Takeaways
- In 2024, the market for generic sterile injectable generated a revenue of US$ 42.9 Billion, with a CAGR of 9.9%, and is expected to reach US$ 110.3 Billion by the year 2034.
- The product type segment is divided into monoclonal antibodies, antibiotics, insulin, and others, with monoclonal antibodies taking the lead in 2023 with a market share of 40.5%.
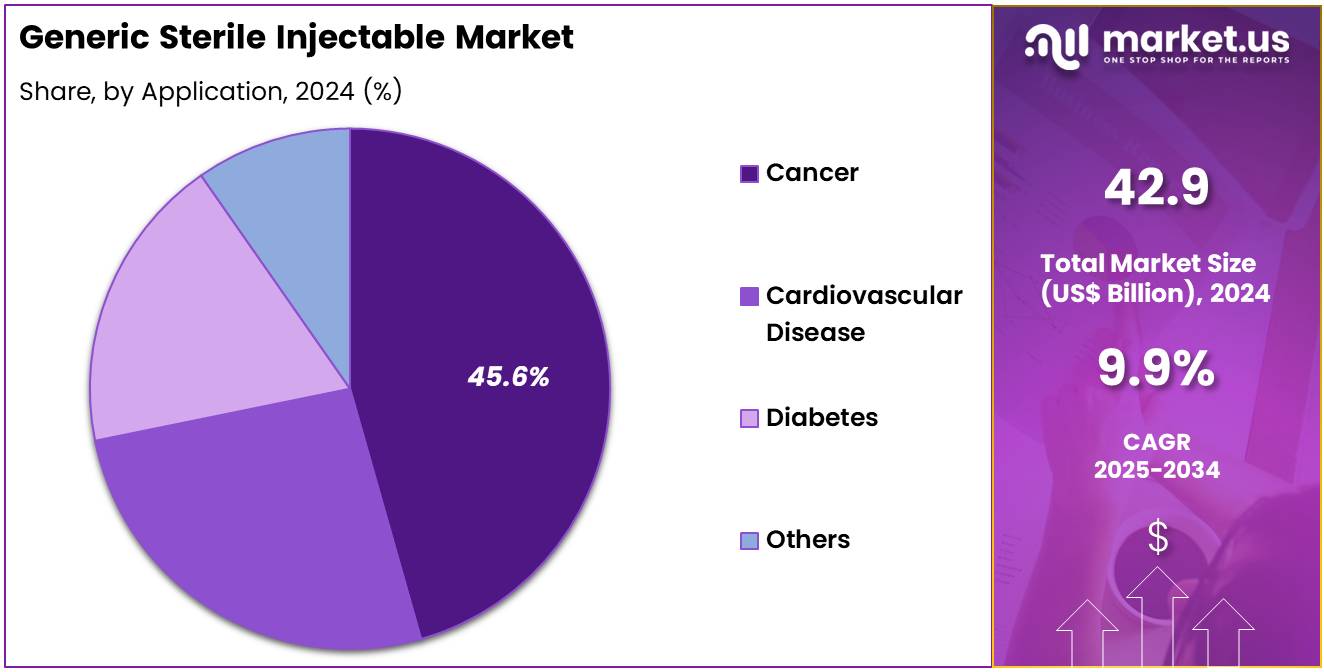
- Considering application, the market is divided into cancer, cardiovascular disease, diabetes, and others. Among these, cancer held a significant share of 45.6%.
- Furthermore, concerning the distribution channel segment, the market is segregated into hospital pharmacies, retail pharmacies, online pharmacies, and others. The hospital pharmacies sector stands out as the dominant player, holding the largest revenue share of 50.2% in the generic sterile injectable market.
- North America led the market by securing a market share of 41.7% in 2023.
Product Type Analysis
Monoclonal antibodies hold a dominant share of 40.5% in the generic sterile injectable market. This growth is expected to continue as monoclonal antibodies are increasingly used for the treatment of various conditions, particularly cancer, autoimmune diseases, and chronic conditions. Generic versions of monoclonal antibodies offer a cost-effective alternative to their branded counterparts, making them highly attractive to both healthcare providers and patients, especially in markets with high demand for biologic therapies.
The approval of generic versions of blockbuster monoclonal antibody drugs is projected to drive significant growth in this segment. As the patent expirations of leading biologics occur, more monoclonal antibody generics are entering the market, providing cost savings while maintaining efficacy. The rising adoption of biologic treatments in oncology and autoimmune diseases is anticipated to further support the growth of this segment. Furthermore, the increasing focus on targeted therapies and personalized medicine is likely to expand the use of monoclonal antibodies in clinical practice, further boosting their demand.
Application Analysis
Cancer holds the largest share of 45.6% in the application segment of the generic sterile injectable market. This growth is driven by the increasing incidence of cancer worldwide, with more patients requiring effective and affordable treatments. Generic sterile injectables, particularly monoclonal antibodies and chemotherapy drugs, are becoming more widely adopted due to their ability to provide cost-effective treatment options for cancer patients.
Advances in cancer research, along with the development of new therapies, are likely to increase the demand for injectable cancer treatments, including generics. The growing focus on immunotherapy, which includes the use of monoclonal antibodies to target cancer cells, is expected to drive further growth in this segment. The global rise in cancer prevalence, particularly in developing countries, is projected to increase the market for these treatments, as patients seek more affordable alternatives to branded cancer therapies. As healthcare systems globally prioritize cancer care and the availability of generics increases, the demand for cancer-related injectable drugs is expected to remain strong.
Distribution Channel Analysis
Hospital pharmacies are the leading distribution channel for generic sterile injectables, holding 50.2% of the market share. This growth is expected to continue as hospitals remain the primary setting for the administration of injectable therapies, especially for complex and critical conditions such as cancer and cardiovascular diseases. Hospital pharmacies are essential in ensuring the timely and accurate delivery of injectable medications to patients, which is critical for patient outcomes, particularly in oncology and emergency care.
The increasing complexity of healthcare treatments and the growing number of patients requiring injectable therapies in hospitals is likely to drive further demand for these medications. Additionally, the adoption of electronic systems for tracking drug usage and improving pharmacy operations in hospitals is expected to enhance the efficiency and safety of injectable drug administration. As the number of hospitalized patients and outpatient procedures continues to rise globally, the role of hospital pharmacies in distributing and managing injectable drugs will continue to be central, leading to continued growth in this segment.
Key Market Segments
By Product Type
- Monoclonal Antibodies
- Antibiotics
- Insulin
- Others
By Application
- Cancer
- Cardiovascular Disease
- Diabetes
- Others
By Distribution Channel
- Hospital Pharmacies
- Retail Pharmacies
- Online Pharmacies
- Others
Drivers
Growing Demand for Affordable Healthcare Solutions is Driving the Market
The escalating global demand for cost-effective healthcare solutions, driven by rising healthcare expenditures and increasing pressure on healthcare systems to manage budgets, is a significant driver propelling the generic sterile injectable market. Generic sterile injectables offer a more affordable alternative to their branded counterparts, providing access to essential medicines for a wider patient population, particularly in critical care, oncology, and infectious disease management.
As patent expirations on branded injectable drugs increase, generic versions quickly enter the market, offering substantial cost savings. The US Food and Drug Administration (FDA) plays a crucial role in enabling this access by approving Abbreviated New Drug Applications (ANDAs) for generics. In fiscal year 2022, the FDA’s Office of Generic Drugs (OGD) approved or tentatively approved 914 ANDAs, including 106 first generics, many of which are sterile injectables.
This consistent flow of generic approvals directly contributes to increased competition and lower drug prices, making treatments more accessible and reducing the financial burden on healthcare providers and patients alike. The imperative to provide high-quality, yet affordable, parenteral therapies underpins the continuous expansion and demand within this vital market segment.
Restraints
Stringent Regulatory Requirements and Manufacturing Complexities are Restraining the Market
The inherently stringent regulatory requirements and the significant manufacturing complexities associated with generic sterile injectables pose a considerable restraint on market growth. Producing sterile injectable drugs demands highly controlled environments (aseptic conditions), specialized equipment, and rigorous quality control measures to prevent contamination and ensure product safety. Meeting these Good Manufacturing Practice (GMP) standards requires substantial capital investment and ongoing compliance efforts.
Regulatory bodies like the US Food and Drug Administration (FDA) conduct frequent inspections and impose strict guidelines, and any deviations can lead to warning letters, import alerts, or even product recalls, disrupting supply. While specific figures for sterile injectable recalls due to contamination are often aggregated within broader medical product recalls, the FDA’s enforcement actions highlight persistent quality challenges.
For example, the FDA’s “2024 Medical Device Recalls” database, which often includes sterile components like pre-filled syringes and infusion pump administration sets, shows numerous recalls throughout 2024 due to manufacturing defects or potential particulate matter risks. These issues underscore the rigorous oversight and the high bar for compliance in sterile manufacturing. Furthermore, the specialized expertise required for aseptic processing and the lengthy approval processes for manufacturing sites deter new entrants and increase operational costs for existing players, thereby limiting market agility and overall supply.
Opportunities
Advancements in Manufacturing Technologies and Capacity Expansion are Creating Growth Opportunities
Significant advancements in pharmaceutical manufacturing technologies, coupled with strategic investments in expanding production capacities, are creating substantial growth opportunities in the generic sterile injectable market. Innovations such as continuous manufacturing, advanced aseptic filling lines, and digital quality control systems are enhancing efficiency, reducing production costs, and improving the quality and safety of these sensitive products. These technologies minimize human intervention, thereby reducing the risk of contamination and accelerating production cycles.
The US Food and Drug Administration (FDA) actively promotes the adoption of advanced manufacturing technologies (AMTs) within the pharmaceutical industry. The FDA’s “Advanced Manufacturing Technologies Designation Program,” for which final guidance was made available in December 2024, explicitly encourages the early adoption of such technologies to improve drug quality and supply chain resilience. Furthermore, several key generic drug manufacturers have announced investments in new or upgraded facilities.
For instance, in August 2023, the US Department of Health and Human Services (HHS) announced a US$50 million investment to expand domestic sterile injectable manufacturing capacity, specifically for critical medicines, through a partnership with Phlow Corporation and Civica Rx. These investments and technological leaps are crucial for meeting the rising demand, mitigating drug shortages, and ensuring a more robust and reliable supply of generic sterile injectables globally.
Impact of Macroeconomic / Geopolitical Factors
Global macroeconomic conditions, including inflation and the overall investment climate within the pharmaceutical industry, significantly influence the generic sterile injectable market by affecting manufacturing costs and capital availability. Inflation can increase the cost of raw materials, energy, labor, and transportation, directly impacting the profitability of generic drug manufacturers who often operate on thin margins. This can put pressure on companies to reduce output or delay investments in new facilities.
However, the essential nature of many generic sterile injectables for hospital and emergency care often ensures a baseline demand that is somewhat insulated from economic downturns. According to the American Hospital Association’s (AHA) “Costs of Caring” report from April 2025, US hospitals faced a 5.1% increase in total expenses in 2024, with pharmaceutical costs being a significant component. Despite these cost pressures, the ongoing need for affordable critical care medicines continues to drive investment.
Geopolitical stability is also crucial for maintaining robust global supply chains for active pharmaceutical ingredients (APIs) and excipients. The imperative to provide affordable, life-saving medicines ensures that the market for generic sterile injectables remains a priority for both public health initiatives and pharmaceutical companies, fostering ongoing development and supply.
Evolving US trade policies, including the potential for tariffs on imported pharmaceutical raw materials and finished drug products, are shaping the generic sterile injectable market by influencing manufacturing costs and driving supply chain restructuring. Many generic sterile injectables, or their key ingredients, are sourced from international suppliers, particularly from countries like India and China. While pharmaceuticals have historically enjoyed some exemptions, recent discussions around broader tariffs could impact this.
Al Jazeera reported on July 9, 2025, about discussions by US leadership to impose a 20% levy on pharmaceutical products, potentially rising to 200% in a year, which would significantly affect imported medicines. The US Bureau of Economic Analysis (BEA) reported in February 2025 that pharmaceutical preparations accounted for US$ 43.6 billion of increased capital goods imports into the US in 2024, highlighting the significant volume of these imports. Such tariffs could increase manufacturing costs for generic sterile injectables, potentially leading to higher drug prices or reduced profitability for manufacturers.
Conversely, these policies also encourage domestic manufacturing and diversification of supply chains, prompting investments in US-based production facilities to enhance resilience and reduce reliance on foreign sources. While tariffs present cost challenges, the strategic importance of a stable and affordable generic sterile injectable supply for national health security will likely drive continued efforts to optimize and secure the supply chain.
Latest Trends
Increasing Emergence of Biosimilars and Complex Generics is a Recent Trend
A prominent recent trend shaping the generic sterile injectable market in 2024 and continuing into 2025 is the increasing emergence and adoption of biosimilars and other complex generic injectable products. Biosimilars, which are highly similar to already approved biologic (often injectable) reference products, offer significant cost savings, analogous to traditional generics, but for more complex, often life-saving, protein-based therapies. As more biologic drugs lose patent exclusivity, the development and approval of biosimilars are accelerating.
The US Food and Drug Administration (FDA) has actively approved a growing number of biosimilars. As of July 15, 2025, the FDA’s “Biosimilar Product Information” database lists several recent approvals that directly impact the sterile injectable market. For instance, biosimilar versions of bevacizumab (Avastin) were approved in April 2025, and ustekinumab (Stelara) in May 2025, with denosumab (Xgeva/Prolia) biosimilars also receiving approvals in March and February 2025.
These approvals represent complex injectable products. The growth of this segment not only expands the overall generic sterile injectable market but also introduces heightened competition for high-value biologic drugs, increasing patient access to critical therapies for conditions like cancer, autoimmune diseases, and osteoporosis.
Regional Analysis
North America is leading the Generic Sterile Injectable Market
The generic sterile injectable market in North America, holding a significant 41.7% share, experienced robust growth in 2024. This expansion was primarily driven by the increasing demand for affordable pharmaceutical products, the rising prevalence of chronic and acute diseases requiring injectable treatments, and the strategic focus on cost containment within healthcare systems. Injectable drug products represented a substantial portion of all drug shortages, accounting for half of all drug shortages between 2018 and 2023, as reported by the US Department of Health and Human Services (HHS) in January 2025.
This highlights the persistent need for a reliable supply of these essential medications. In response, the US Food and Drug Administration (FDA) continues to prioritize the approval of generic injectable medicines to address these shortages and improve patient access. Leading pharmaceutical companies with strong generic portfolios have demonstrated positive performance in the region. Sandoz, a global leader in generics, reported net sales in North America of US$2.1 billion in 2023, driven by strong demand and new product launches.
Similarly, Teva Pharmaceutical Industries, another major player, announced global revenues of US$4.3 billion in the third quarter of 2024, reflecting a 15% increase in local currency terms compared to the third quarter of 2023, with its generics business contributing to growth across all geographies, including North America. This growth underscores the critical role that these products play in providing essential and cost-effective treatments.
The Asia Pacific region is expected to experience the highest CAGR during the forecast period
The generic sterile injectable market in Asia Pacific is expected to grow significantly during the forecast period. This anticipated expansion is primarily driven by the region’s large and aging population, increasing healthcare expenditure, and a growing emphasis on expanding access to affordable medicines. Governments across Asia Pacific are actively promoting domestic pharmaceutical manufacturing and the use of generic drugs to reduce healthcare costs and improve public health outcomes.
For instance, India’s pharmaceutical market for fiscal year 2023-2024 is valued at US$50 billion, with domestic consumption at US$23.5 billion and exports at US$26.5 billion, as reported by the Ministry of Chemicals and Fertilizers. This robust industry is a key supplier of generic medicines globally, including sterile injectables. China’s total expenditure on public health reached RMB 9,057,581 million (approximately US$1.25 trillion) in 2023, an increase from RMB 8,532,749 million in 2022, indicating substantial investment in healthcare infrastructure and pharmaceutical procurement.
Major pharmaceutical companies are actively expanding their presence and manufacturing capabilities in Asia Pacific to meet this rising demand. Fresenius Kabi, a prominent provider of injectable medicines, continues to invest in its global production network, including facilities in Asia, to ensure the supply of essential sterile injectables. This strategic focus by both governments and key industry players is expected to drive substantial growth in the market across the region.
Key Regions and Countries
North America
- US
- Canada
Europe
- Germany
- France
- The UK
- Spain
- Italy
- Russia
- Netherland
- Rest of Europe
Asia Pacific
- China
- Japan
- South Korea
- India
- Australia
- New Zealand
- Singapore
- Thailand
- Vietnam
- Rest of APAC
Latin America
- Brazil
- Mexico
- Rest of Latin America
Middle East & Africa
- South Africa
- Saudi Arabia
- UAE
- Rest of MEA
Key Players Analysis
Key players in the generic sterile injectable market implement several strategies to drive growth and enhance patient care. They focus on developing high-quality, cost-effective injectable medications to meet the increasing demand for affordable treatments. Companies are investing in advanced manufacturing technologies and expanding production capacities to improve efficiency and meet regulatory standards.
Strategic partnerships and collaborations with healthcare providers and research institutions enable these companies to innovate and broaden their product offerings. Additionally, they are enhancing their global presence by entering emerging markets, thereby increasing accessibility to their products. Emphasis on sustainability and supply chain resilience ensures a consistent and reliable supply of sterile injectables.
Hikma Pharmaceuticals, a leading player in the market, has been actively expanding its operations to strengthen its position. Founded in 1978 in Jordan, Hikma has grown to become a significant global provider of generic injectable medicines. The company has made substantial investments in the United States, including a US$1 billion commitment over five years to expand manufacturing and R&D operations in Ohio, aiming to increase domestic production and address drug price disparities.
In 2024, Hikma further bolstered its portfolio by acquiring a portfolio of products from Xellia Pharmaceuticals for up to US$185 million, which included eight approved products and eleven in development, along with a manufacturing facility in Cleveland and a research center in Zagreb. This acquisition is expected to contribute approximately US$75 million in annual revenue, enhancing Hikma’s capabilities in the generic sterile injectable market.
Top Key Players
- Teva Pharmaceuticals
- Sun Pharmaceutical Industries Ltd
- Pfizer
- Nichi-Iko
- Hikma
- AstraZeneca
- Alembic Pharmaceuticals Ltd
- 3M
Recent Developments
- In June 2024, Teva Pharmaceuticals, a US affiliate of Teva Pharmaceutical Industries Ltd., launched an approved generic version of Victoza (liraglutide injection 1.8 mg) in the US As of April 2024, Victoza generated annual revenue of $1.656 billion.
- In June 2024, Alembic Pharmaceuticals Ltd. announced that it had received final approval from the US health authorities for its generic Icatibant injectable, which is used to treat acute episodes of hereditary angioedema in adults.
Report Scope
Report Features Description Market Value (2024) US$ 42.9 Billion Forecast Revenue (2034) US$ 110.3 Billion CAGR (2025-2034) 9.9% Base Year for Estimation 2024 Historic Period 2020-2023 Forecast Period 2025-2034 Report Coverage Revenue Forecast, Market Dynamics, COVID-19 Impact, Competitive Landscape, Recent Developments Segments Covered By Product Type (Monoclonal Antibodies, Antibiotics, Insulin, and Others), By Application (Cancer, Cardiovascular Disease, Diabetes, and Others), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies, and Others) Regional Analysis North America – US, Canada; Europe – Germany, France, The UK, Spain, Italy, Russia, Netherlands, Rest of Europe; Asia Pacific – China, Japan, South Korea, India, Australia, New Zealand, Singapore, Thailand, Vietnam, Rest of APAC; Latin America – Brazil, Mexico, Rest of Latin America; Middle East & Africa – South Africa, Saudi Arabia, UAE, Rest of MEA Competitive Landscape Teva Pharmaceuticals, Sun Pharmaceutical Industries Ltd, Pfizer, Nichi-Iko, Hikma, AstraZeneca, Alembic Pharmaceuticals Ltd, 3M. Customization Scope Customization for segments, region/country-level will be provided. Moreover, additional customization can be done based on the requirements. Purchase Options We have three licenses to opt for: Single User License, Multi-User License (Up to 5 Users), Corporate Use License (Unlimited User and Printable PDF)  Generic Sterile Injectable MarketPublished date: July 2025add_shopping_cartBuy Now get_appDownload Sample
Generic Sterile Injectable MarketPublished date: July 2025add_shopping_cartBuy Now get_appDownload Sample -
-
- Teva Pharmaceuticals
- Sun Pharmaceutical Industries Ltd
- Pfizer
- Nichi-Iko
- Hikma
- AstraZeneca
- Alembic Pharmaceuticals Ltd
- 3M










