Global Antifungal Nail Drugs Market By Drug Class (Allylamines, Azoles, Echinocandins, Polyenes, Others) By Medication Type (Terbinafine, Itraconazole, Fluconazole, Ciclopirox, Efinaconazole, Tavaborole, Others) By Application (Onychomycosis, Tinea Unguium, Candidal Onychomycosis, Others) By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies, Dermatology Clinics), Region and Companies – Industry Segment Outlook, Market Assessment, Competition Scenario, Trends and Forecast 2026-2035
- Published date: May 2026
- Report ID: 185690
- Number of Pages: 277
- Format:
-
keyboard_arrow_up
Quick Navigation
Report Overview
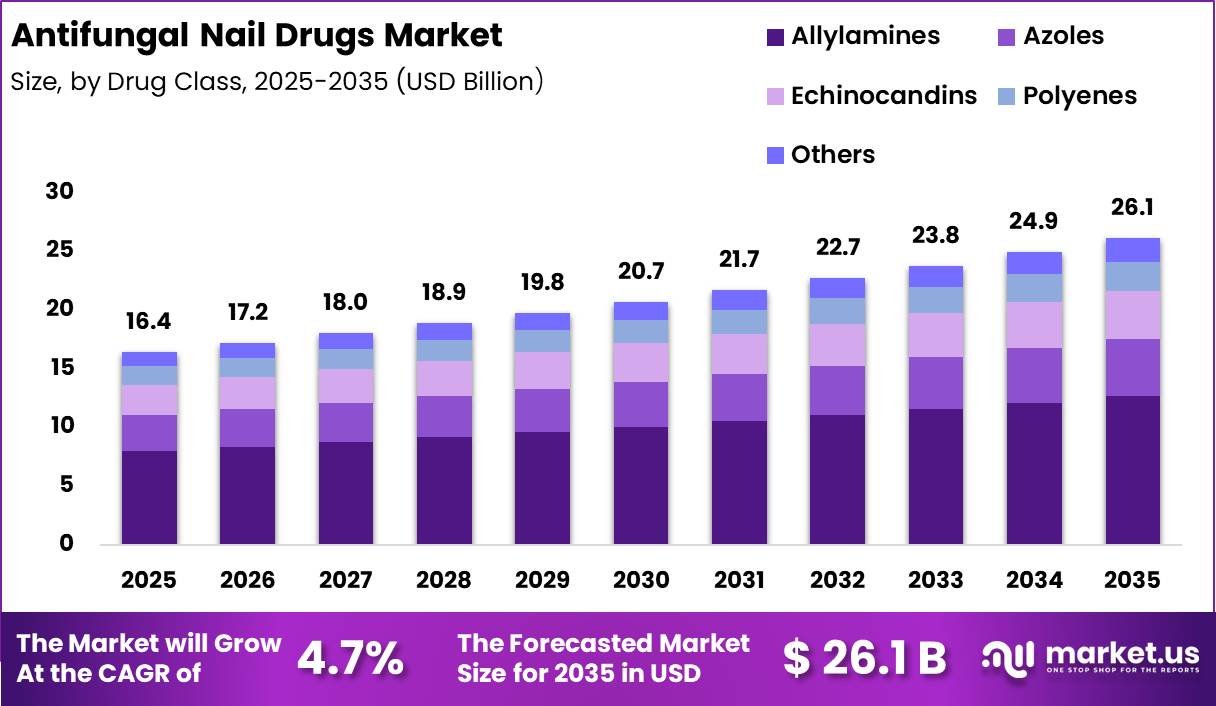
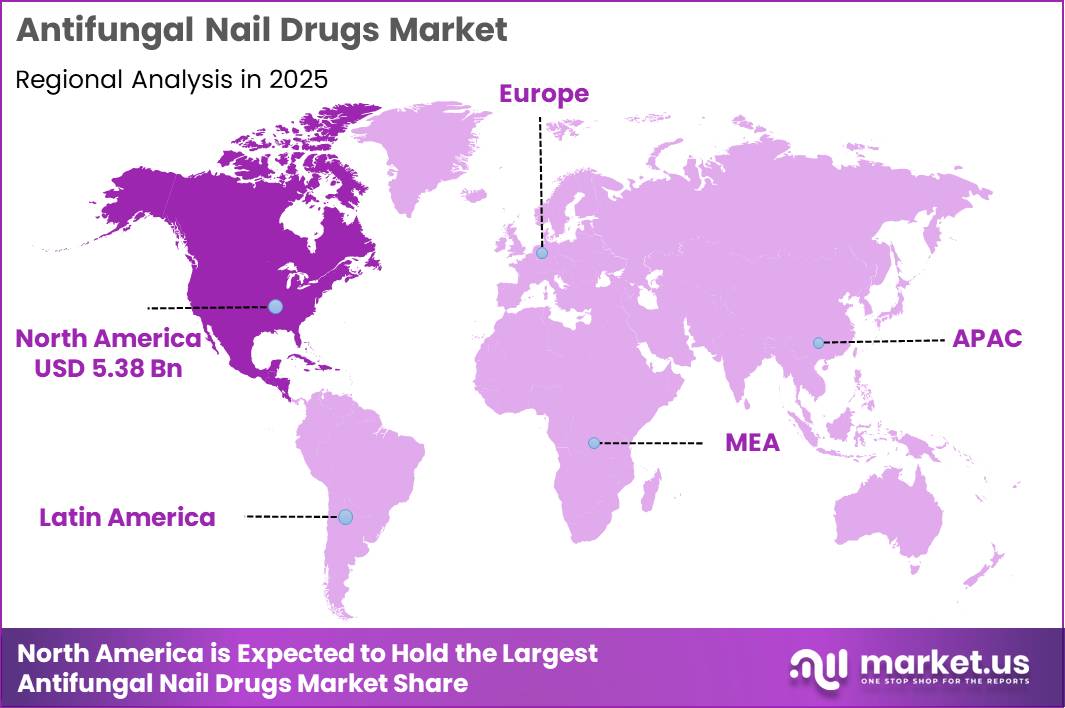
Global Antifungal Nail Drugs Market size is expected to be worth around US$ 26.1 Billion by 2035 from US$ 16.4 Billion in 2025, growing at a CAGR of 4.9% during the forecast period from 2026 to 2035. In 2025, North America led the market, achieving over 39.50% share with a revenue of US$ 5.38 Billion.
The global market for antifungal nail drugs is undergoing a structural transformation, driven by increased clinical awareness and a shift toward therapeutic eradication rather than cosmetic management.Onychomycosis, a primary fungal nail infection, is estimated to affect approximately 10–14% of the global population, with prevalence rising to nearly 20% among individuals above 60 years of age, as reported by World Health Organization and supported by dermatological studies referenced by the Centers for Disease Control and Prevention. This growing patient pool is contributing to sustained demand for effective antifungal interventions.
Investment in research and development is increasing, particularly in advanced formulations such as transungual delivery systems, which improve drug penetration through the dense keratin structure of the nail.
Clinical success rates for newer topical agents have improved, with some therapies demonstrating efficacy rates exceeding 50–60% in mild-to-moderate cases, compared to less than 30% for traditional lacquers. Oral antifungal therapies, although highly effective with cure rates approaching 70–80%, continue to face limitations due to systemic side effects, encouraging innovation in safer topical alternatives.
Behavioral and lifestyle factors are significantly influencing market expansion. Increased use of occlusive footwear, rising participation in fitness activities, and exposure to communal environments are elevating infection risks.
Additionally, diabetic populations estimated at over 530 million globally represent a high-risk segment, where antifungal prophylaxis plays a critical role in preventing complications such as foot ulcers and secondary infections.
Emerging economies are expected to witness accelerated growth, supported by improving healthcare infrastructure and rising disposable incomes. Government-led hygiene awareness programs and faster regulatory pathways for dermatological treatments are further strengthening market penetration.
As a result, pharmaceutical companies are prioritizing high-efficacy prescription therapies, positioning the antifungal nail drugs market for steady expansion with a focus on innovation, safety, and improved patient outcomes.
Key Takeaways
- Market Size: Global Antifungal Nail Drugs Market size is expected to be worth around US$ 26.1 Billion by 2035 from US$ 16.4 Billion in 2025.
- Market Share: The market growing at a CAGR of 4.9% during the forecast period from 2026 to 2035.
- Drug Class Analysis: Allylamines are projected to dominate the market with a 48.5% share.
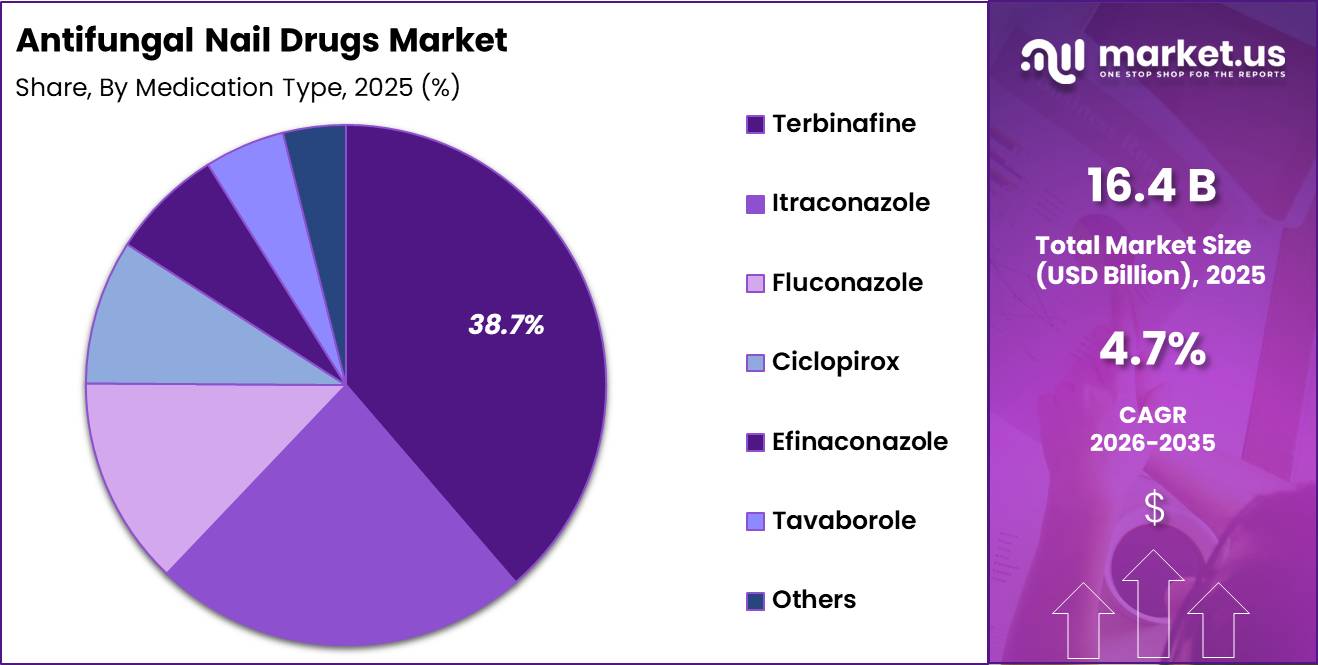
- Medication Type Analysis: Terbinafine is anticipated to lead the segment with a 38.7% market share in 2025.
- Application Analysis: Onychomycosis dominate the antifungal nail drugs market, accounting for an estimated 68.4% share in 2025.
- Distribution Channel Analysis: retail pharmacies projected to account for 44.6% of the market share in 2025
- Regional Analysis: In 2025, North America led the market, achieving over 39.50% share with a revenue of US$ 5.38 Billion.
Drug Class Analysis
The drug class segmentation of the antifungal nail drugs market demonstrates a structured distribution across multiple therapeutic categories, with allylamines maintaining a leading position. Allylamines are projected to dominate the market with a 48.5% share in 2025, driven by their fungicidal mechanism and strong efficacy against dermatophytes, which are the primary causative agents of nail infections.
The widespread clinical adoption of terbinafine-based formulations has further strengthened this segment’s growth trajectory. Azoles represent the second-largest segment, supported by their broad-spectrum antifungal activity and availability in both oral and topical formulations. Their effectiveness in treating mixed infections contributes to sustained demand.
Echinocandins, although limited in topical use, are gaining attention in systemic fungal infection management, indirectly supporting market expansion. Polyenes hold a smaller share due to limited nail penetration but continue to be used in specific clinical scenarios.
The “others” category, including newer and combination therapies, is expected to witness gradual growth due to ongoing research and development efforts. Overall, increasing prevalence of onychomycosis and rising awareness regarding early treatment are key factors supporting segmental expansion.
Medication Type Analysis
Based on medication type, the antifungal nail drugs market is characterized by a mix of established oral therapies and emerging topical solutions. Terbinafine is anticipated to lead the segment with a 38.7% market share in 2025, attributed to its high cure rates, shorter treatment duration, and strong clinical recommendation as a first-line therapy.
Its systemic action ensures deeper nail bed penetration, enhancing treatment outcomes. Itraconazole follows as a significant segment, widely used for patients intolerant to terbinafine and for broader antifungal coverage. Fluconazole, though less commonly prescribed for nail infections, maintains a niche presence due to its favorable safety profile.
Topical agents such as ciclopirox and efinaconazole are gaining traction, particularly among patients preferring non-systemic therapies or those with mild to moderate infections. Tavaborole has emerged as an innovative treatment option, offering improved nail penetration and patient compliance.
The “others” category includes combination therapies and pipeline drugs, reflecting ongoing innovation. Market growth across segments is supported by increasing demand for effective, convenient, and low-side-effect treatment options.
Application Analysis
Application-based segmentation highlights the dominance of onychomycosis in the antifungal nail drugs market, accounting for an estimated 68.4% share in 2025. This dominance is primarily attributed to the high global prevalence of fungal nail infections, particularly among aging populations and individuals with comorbidities such as diabetes.
The chronic nature of onychomycosis and its impact on quality of life have led to increased diagnosis and treatment rates. Tinea unguium represents a closely related segment, often used interchangeably with dermatophyte nail infections, and continues to contribute significantly to market demand.
Candidal onychomycosis, while less prevalent, is gaining attention due to rising cases associated with immunocompromised patients and frequent exposure to moist environments. The “others” segment includes rare fungal infections and mixed pathogen cases, which require specialized treatment approaches.
Growth across all application segments is being driven by advancements in diagnostic techniques, improved treatment accessibility, and rising awareness regarding the importance of early intervention to prevent complications and recurrence.
Distribution Channel Analysis
The distribution channel landscape of the antifungal nail drugs market is led by retail pharmacies, which are projected to account for 44.6% of the market share in 2025. Their dominance is supported by widespread accessibility, availability of over-the-counter topical treatments, and strong consumer preference for convenient purchasing options.
Hospital pharmacies constitute a significant segment, particularly for prescription-based oral antifungal therapies and severe infection cases requiring clinical supervision. Online pharmacies are experiencing rapid growth, driven by increasing digital adoption, home delivery convenience, and competitive pricing strategies. This channel has gained further momentum following shifts in consumer behavior toward e-commerce platforms.
Dermatology clinics represent a specialized distribution segment, where targeted treatment regimens and professional consultation enhance patient outcomes. These clinics often serve as key points for diagnosis and prescription of advanced therapies. The “others” category includes independent drug stores and specialty clinics.
Overall, the expansion of distribution channels is being facilitated by improved healthcare infrastructure and growing emphasis on patient-centric access to antifungal treatments.
Key Market Segments
By Drug Class
- Allylamines
- Azoles
- Echinocandins
- Polyenes
- Others
By Medication Type
- Terbinafine
- Itraconazole
- Fluconazole
- Ciclopirox
- Efinaconazole
- Tavaborole
- Others
By Application
- Onychomycosis
- Tinea Unguium
- Candidal Onychomycosis
- Others
By Distribution Channel
- Hospital Pharmacies
- Retail Pharmacies
- Online Pharmacies
- Dermatology Clinics
Driving Factors
Rising Prevalence of Fungal Nail Infections
The growth of the antifungal nail drugs market is primarily driven by the increasing global burden of onychomycosis (fungal nail infection). Epidemiological studies indicate that approximately 10% of the global adult population is affected, with prevalence rising to 20% among individuals above 60 years and up to 50% in those over 70 years.Furthermore, fungal nail infections account for nearly 50% of all nail disorders, demonstrating a substantial disease burden. The condition is also linked with comorbidities such as diabetes, where up to one-third of diabetic patients are affected.
Public health bodies and clinical literature highlight that aging populations, increased urbanization, and lifestyle factors such as occlusive footwear contribute to higher incidence rates. The chronic nature of the disease, combined with discomfort and risk of secondary infections, necessitates medical intervention. As a result, sustained demand for topical and oral antifungal therapies is being observed, supporting consistent market expansion.
Trending Factors
Shift Toward Advanced and Topical Therapies
A significant trend observed in the antifungal nail drugs market is the transition toward advanced topical therapies and improved drug formulations. Clinical evidence shows that traditional oral antifungal agents such as terbinafine achieve mycological cure rates of approximately 76%, significantly higher than itraconazole at 38%.However, concerns regarding systemic side effects, including liver toxicity, have accelerated the adoption of topical alternatives. Newer topical agents such as efinaconazole have demonstrated improved outcomes, with cure rates two to three times higher than older treatments like ciclopirox.
Healthcare guidelines emphasize long-duration topical therapies (often up to 12 months) to achieve effective results. Additionally, innovations such as medicated nail lacquers and low systemic absorption formulations are gaining traction due to enhanced safety profiles.
This trend is aligned with patient preference for non-invasive treatments and increasing regulatory focus on safer therapies, thereby shaping product development pipelines and influencing prescribing patterns globally.
Restraining Factors
Low Treatment Success Rates and High Recurrence
Despite growing demand, the antifungal nail drugs market faces notable restraints due to suboptimal treatment outcomes and high recurrence rates. Clinical studies indicate that even after successful therapy, recurrence occurs in approximately 20–25% of cases within a few years.Moreover, complete cure rates remain relatively low across treatment categories, particularly for topical drugs, where effectiveness can be as low as 6–9% for certain formulations such as ciclopirox. The long duration of therapy, often exceeding 6–12 months, reduces patient adherence and overall treatment success.
Additionally, oral antifungal medications are associated with adverse effects, limiting their use in certain patient populations. Healthcare sources also indicate that the disease is difficult to eradicate due to slow nail growth and fungal persistence in nail beds. These factors collectively reduce treatment efficiency and create barriers to widespread adoption, thereby constraining market growth despite high disease prevalence.
Opportunity
Growing Geriatric Population and Untapped Patient Pool
Significant opportunities exist in the antifungal nail drugs market due to demographic shifts and underdiagnosed patient populations. The global elderly population is expanding rapidly, and prevalence of onychomycosis increases substantially with age, reaching up to 50% in individuals above 70 years.
Additionally, global estimates suggest that fungal nail infections affect between 5% and 25% of the population, translating to hundreds of millions of cases worldwide. Despite this, a considerable proportion of patients remain untreated due to lack of awareness or perception of the condition as cosmetic.
Healthcare institutions emphasize the importance of early diagnosis to prevent complications such as pain, mobility impairment, and secondary infections. Emerging markets, particularly in Asia and Latin America, present untapped opportunities due to improving healthcare access and rising awareness.
Furthermore, ongoing research into novel drug delivery systems and combination therapies offers potential to improve cure rates, thereby expanding the addressable market and supporting long-term growth.
Regional Analysis
The regional landscape of the antifungal nail drugs market demonstrates a concentrated dominance in North America, which accounted for over 39.50% of the global market share in 2025, generating revenue of approximately US$ 5.38 billion. This leadership position can be attributed to a well-established healthcare infrastructure, high awareness regarding fungal infections, and increased access to advanced treatment options.
The prevalence of onychomycosis has been observed to be relatively high in this region, further supporting sustained demand for antifungal therapeutics. Additionally, strong presence of key pharmaceutical manufacturers and ongoing investments in research and development have accelerated product innovation and availability.
Europe represents the second-largest regional market, supported by favorable reimbursement policies and growing geriatric population, which is more susceptible to fungal nail infections. Meanwhile, the Asia-Pacific region is expected to witness the fastest growth rate over the forecast period, driven by expanding healthcare access, rising disposable incomes, and increasing awareness of personal hygiene and dermatological conditions.
Emerging markets in Latin America and the Middle East & Africa are also experiencing gradual growth, supported by improving healthcare infrastructure and increased penetration of over-the-counter antifungal products. However, limited awareness and affordability constraints may continue to restrict rapid market expansion in these regions.
Key Regions and Countries
North America
- The US
- Canada
Europe
- Germany
- France
- The U.K.
- Italy
- Spain
- Russia & CIS
- Rest of Europe
Asia Pacific
- China
- India
- Japan
- South Korea
- ASEAN
- Australia & New Zealand
- Rest of Asia Pacific
Middle East & Africa
- GCC
- South Africa
- Rest of Middle East & Africa
Latin America
- Brazil
- Mexico
- Rest of Latin America
Key Players Analysis
Key players in the antifungal nail drugs market are increasingly prioritizing differentiation strategies to address the saturation of generic azole-based treatments. Innovation in drug delivery technologies has become a central focus, with investments directed toward advanced systems such as transfersomes and polymeric lacquers, which are designed to improve nail plate penetration and enhance therapeutic outcomes. These advancements are expected to improve patient adherence by minimizing the need for invasive procedures.
A notable strategic approach involves the transition of prescription products to over-the-counter availability, enabling companies to expand their consumer base and extend the commercial lifecycle of established formulations. This trend aligns with the growing demand for self-care solutions in dermatology.
Collaborations with diagnostic companies are also being leveraged to develop integrated “test-and-treat” offerings, which enhance treatment precision and strengthen relationships with healthcare professionals. Additionally, sustainability initiatives, including the adoption of biodegradable packaging, are gaining importance in brand positioning.
Market consolidation remains prominent, with larger firms acquiring smaller biotechnology companies possessing late-stage pipeline assets, thereby reinforcing product portfolios and mitigating risks associated with patent expirations.
Market Key Players
- Pfizer Inc.
- Novartis AG
- GlaxoSmithKline plc
- Bayer AG
- Johnson & Johnson
- Merck & Co., Inc.
- Abbott Laboratories
- Bausch Health Companies Inc.
- Mylan N.V.
- Teva Pharmaceutical Industries Ltd.
- Cipla Inc.
- Sun Pharmaceutical Industries Ltd.
- Dr. Reddy’s Laboratories Ltd.
- Galderma S.A.
- Valeant Pharmaceuticals International Inc.
- Others
Recent Developments
- Novartis AG (Feb, 2025): Novartis expanded its oral antifungal offering for onychomycosis across selected European markets, improving access to systemic nail fungus therapies and strengthening its dermatology portfolio focus on high-need fungal indications.
- Pfizer Inc (Apr, 2025 ): Pfizer continued to leverage its recently granted patent on novel antifungal pyridine derivatives to build a broader late‑stage antifungal pipeline, positioning the company to refresh its systemic antifungal portfolio used in difficult nail and skin mycoses in the coming years.
- Bayer AG (Jun, 2025): Bayer intensified investment in antifungal research and over‑the‑counter dermatology lines, using new collaborations and R&D programs highlighted in 2025–2026 dermal mycosis market updates to expand future options for topical nail fungus therapies under its consumer health brands.
- Moberg Pharma (Jun, 2025): Terclara, a high‑penetration topical terbinafine lacquer for nail fungus, became the market leader in Norway only months after its OTC launch, underlining how rapid uptake of effective topicals can shift share away from legacy oral regimens marketed by large pharma players such as Novartis, Johnson & Johnson, and Merck.
Report Scope
Report Features Description Market Value (2025) US$ 16.4 Billion Forecast Revenue (2035) US$ 26.1 Billion CAGR (2026-2035) 4.9% Base Year for Estimation 2025 Historic Period 2020-2024 Forecast Period 2026-2035 Report Coverage Revenue Forecast, Market Dynamics, Competitive Landscape, Recent Developments Segments Covered By Drug Class (Allylamines, Azoles, Echinocandins, Polyenes, Others) By Medication Type (Terbinafine, Itraconazole, Fluconazole, Ciclopirox, Efinaconazole, Tavaborole, Others) By Application (Onychomycosis, Tinea Unguium, Candidal Onychomycosis, Others) By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies, Dermatology Clinics) Regional Analysis North America – The US, Canada; Europe – Germany, France, U.K., Italy, Spain, Russia & CIS, Rest of Europe; Asia Pacific – China, India, Japan, South Korea, ASEAN, Australia & New Zealand, Rest of Asia Pacific; Middle East & Africa – GCC, South Africa, Rest of Middle East & Africa; Latin America – Brazil, Mexico, Rest of Latin America Competitive Landscape Pfizer Inc., Novartis AG, GlaxoSmithKline plc, Bayer AG, Johnson & Johnson, Merck & Co., Inc., Abbott Laboratories, Bausch Health Companies Inc., Mylan N.V., Teva Pharmaceutical Industries Ltd., Cipla Inc., Sun Pharmaceutical Industries Ltd., Dr. Reddy’s Laboratories Ltd., Galderma S.A., Valeant Pharmaceuticals International Inc., Others, Customization Scope Customization for segments, region/country-level will be provided. Moreover, additional customization can be done based on the requirements. Purchase Options We have three licenses to opt for: Single User License, Multi-User License (Up to 5 Users), Corporate Use License (Unlimited User and Printable PDF)  Antifungal Nail Drugs MarketPublished date: May 2026add_shopping_cartBuy Now get_appDownload Sample
Antifungal Nail Drugs MarketPublished date: May 2026add_shopping_cartBuy Now get_appDownload Sample -
-
- Pfizer Inc.
- Novartis AG
- GlaxoSmithKline plc
- Bayer AG
- Johnson & Johnson
- Merck & Co., Inc.
- Abbott Laboratories
- Bausch Health Companies Inc.
- Mylan N.V.
- Teva Pharmaceutical Industries Ltd.
- Cipla Inc.
- Sun Pharmaceutical Industries Ltd.
- Dr. Reddy's Laboratories Ltd.
- Galderma S.A.
- Valeant Pharmaceuticals International Inc.
- Others










