Global Veterinary CRO Market By Service Type (Clinical Trials, Preclinical Studies, Toxicology Testing, Regulatory Consulting, Pharmacovigilance and Others), By Animal Type (Companion Animals, Livestock Animals and Others), By Application (Oncology, Infectious Diseases, Neurology, Cardiology, Orthopedics, Parasitology, Vaccine Development and Others), By End User (Pharmaceutical Companies, Biotechnology Companies, Veterinary Drug Manufacturers, Research and Academic Institutes and Others), Region and Companies – Industry Segment Outlook, Market Assessment, Competition Scenario, Trends and Forecast 2026-2035
- Published date: April 2026
- Report ID: 183693
- Number of Pages: 302
- Format:
-
keyboard_arrow_up
Quick Navigation
Report Overview
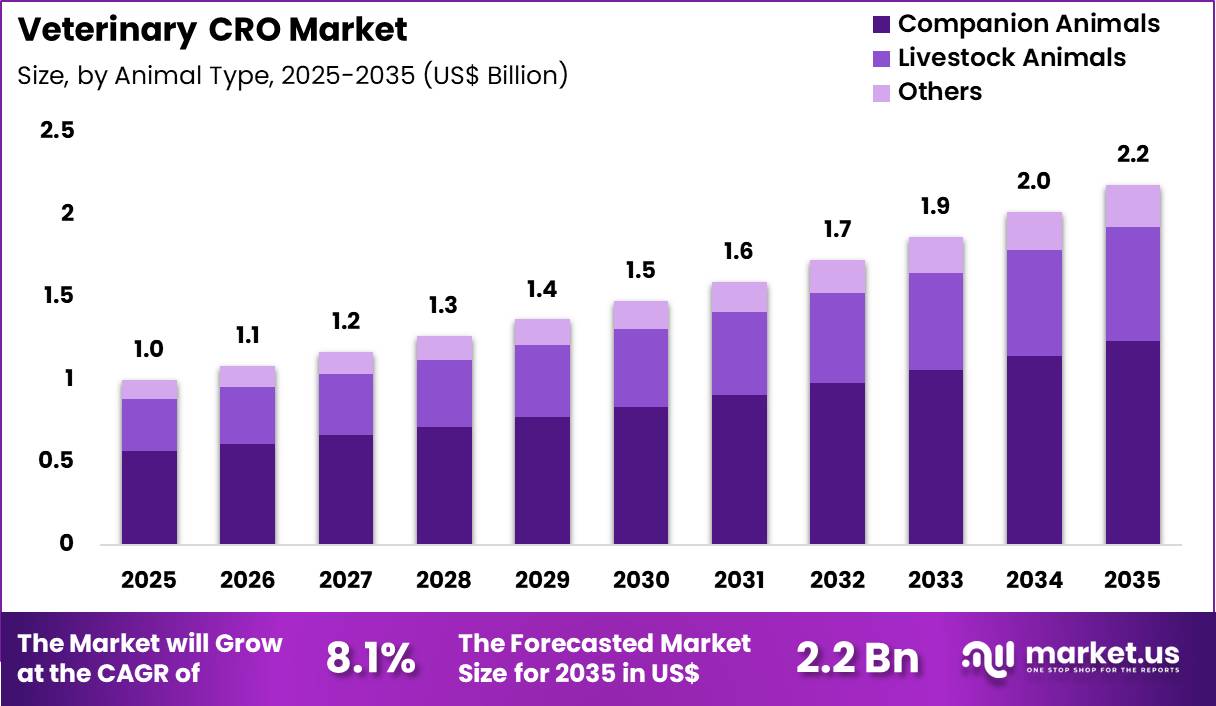
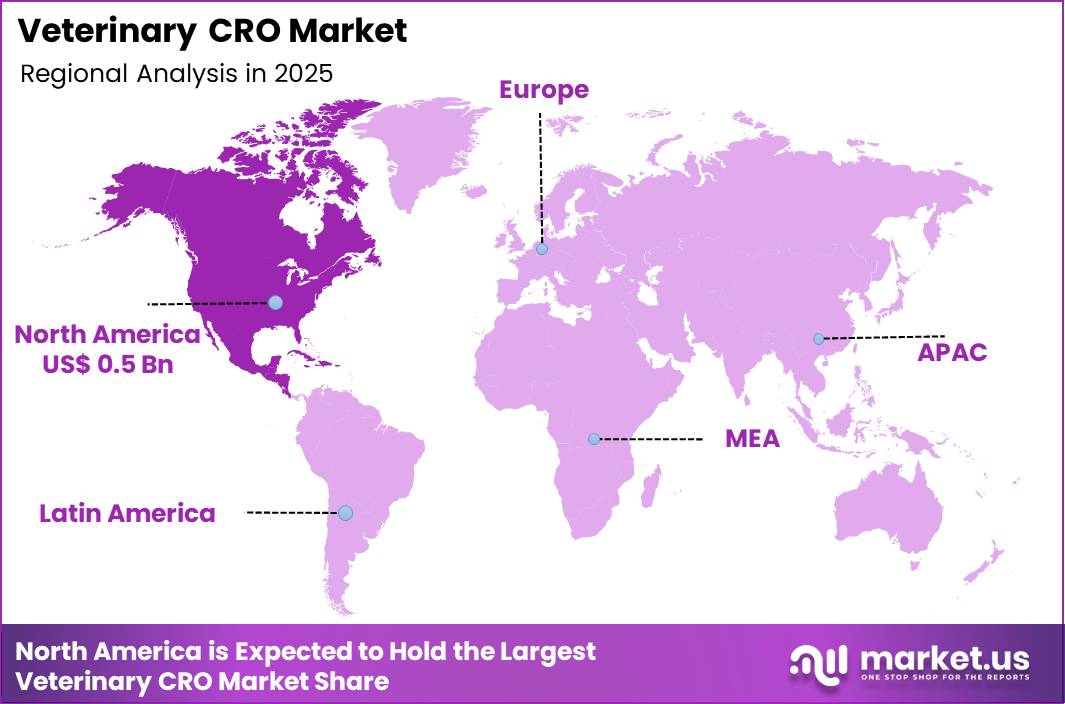
The Global Veterinary CRO Market size is expected to be worth around US$ 2.2 Billion by 2035 from US$ 1.0 Billion in 2025, growing at a CAGR of 8.1% during the forecast period 2026-2035. In 2025, North America led the market, achieving over 45.6% share with a revenue of US$ 0.5 Billion.
Increasing complexity of veterinary drug development and the need for specialized expertise accelerate the Veterinary CRO market as pharmaceutical companies and animal health firms outsource clinical and regulatory activities to organizations with deep domain knowledge.
Veterinary CROs increasingly conduct safety and efficacy studies for companion animal therapeutics, managing multi-center trials that evaluate new analgesics, anti-inflammatories, and parasiticides in dogs and cats under Good Clinical Practice standards.
These organizations support livestock product development by designing and executing field trials for vaccines, antibiotics, and growth promoters, ensuring compliance with regulatory requirements for food-producing animals.
CROs also facilitate bioequivalence studies for generic veterinary medicines, generating data required for approval of biosimilars and generic formulations across species. In addition, they provide regulatory consulting and dossier preparation services that streamline submissions to veterinary authorities for both new chemical entities and line extensions.
Contract research organizations pursue opportunities to integrate advanced data analytics and AI platforms that enhance study design, patient recruitment, and real-world evidence generation, expanding applications in precision animal health and personalized veterinary medicine. These capabilities support the development of targeted therapies for oncology and chronic diseases in companion animals, where biomarker-driven approaches can improve outcomes.
The collaboration between IQVIA and Boehringer Ingelheim announced in January 2026 exemplifies this direction. Under the agreement, Boehringer will utilize IQVIA’s DaaS+ platform to integrate and harmonize commercial and clinical datasets across 59 countries.
This initiative is expected to accelerate the use of AI-driven analytics in animal health research and development, improving decision-making and speeding up innovation cycles across global markets. Recent trends emphasize digital transformation, real-world data utilization, and strategic partnerships that combine clinical operations with advanced analytics, positioning veterinary CROs as essential partners in accelerating innovation while maintaining rigorous scientific and regulatory standards in animal health.
Key Takeaways
- In 2025, the market generated a revenue of US$ 1.0 Billion, with a CAGR of 8.1%, and is expected to reach US$ 2.2 Billion by the year 2035.
- The service type segment is divided into clinical trials, preclinical studies, toxicology testing, regulatory consulting, pharmacovigilance and others, with clinical trials taking the lead with a market share of 35.9%.
- Considering animal type, the market is divided into companion animals, livestock animals and others. Among these, companion animals held a significant share of 56.7%.
- Furthermore, concerning the application segment, the market is segregated into oncology, infectious diseases, neurology, cardiology, orthopedics, parasitology, vaccine development and others. The infectious diseases sector stands out as the dominant player, holding the largest revenue share of 28.4% in the market.
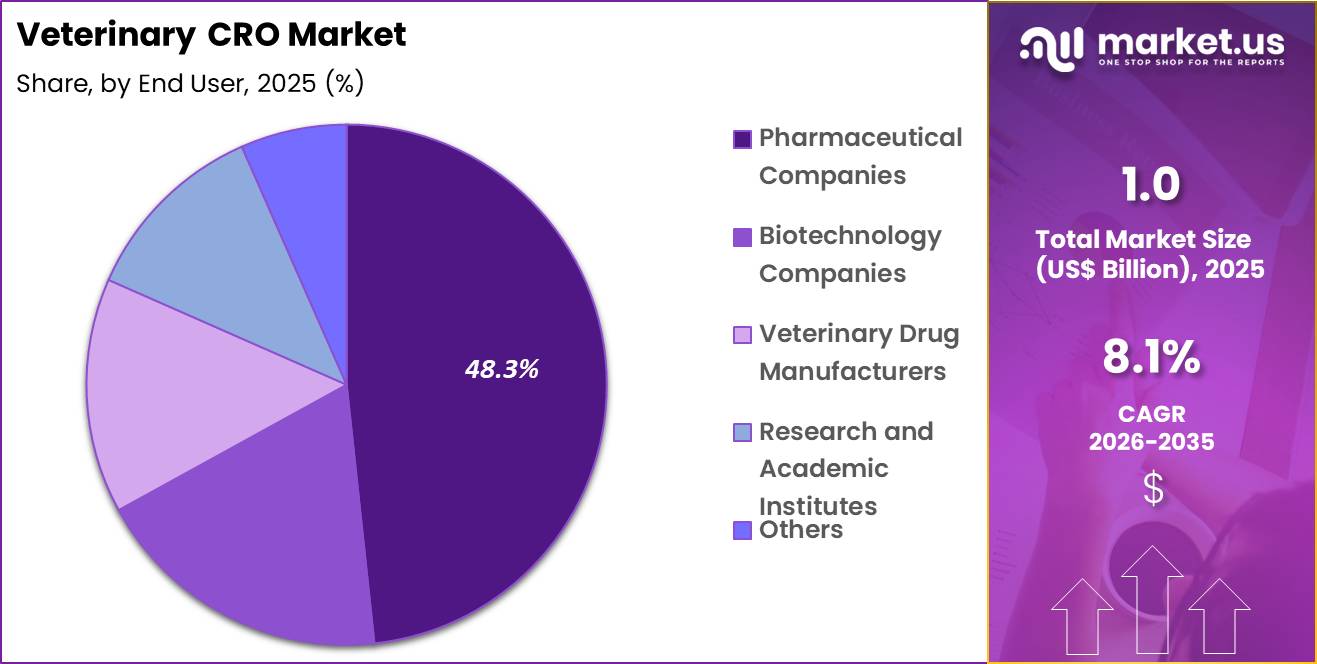
- The end user segment is segregated into pharmaceutical companies, biotechnology companies, veterinary drug manufacturers, research and academic institutes and others, with the pharmaceutical companies segment leading the market, holding a revenue share of 48.3%.
- North America led the market by securing a market share of 45.6%.
Service Type Analysis
Clinical trials accounted for 35.9% of growth within service type and dominate the veterinary CRO market due to the increasing need for validated safety and efficacy data before regulatory approval of animal health products. Veterinary drug development requires structured clinical evaluation across different animal species, which drives demand for CRO-led trial services.
Regulatory authorities mandate well-designed trials for approval, which strengthens the importance of this segment. Clinical trials are expected to expand as new therapeutics, vaccines, and biologics enter the development pipeline. The segment benefits from growing investment in animal health research and rising demand for companion animal treatments.
Pharmaceutical companies are likely to outsource trials to specialized CROs to reduce costs and improve efficiency. Increasing complexity of clinical study designs is projected to support further outsourcing. As veterinary drug innovation accelerates, clinical trials are estimated to remain the leading service segment in this market.
Animal Type Analysis
Companion animals accounted for 56.7% of growth within animal type and dominate the veterinary CRO market due to rising pet ownership and increased spending on pet healthcare. Dogs and cats represent a significant portion of clinical research focus, especially for chronic conditions and preventive treatments.
Veterinary studies indicate growing demand for advanced therapeutics in companion animals, which drives research activity. This segment is expected to grow as pet humanization trends continue to strengthen globally. Pet owners are likely to seek improved treatment options, which supports drug development efforts.
The segment benefits from increasing clinical trial activity focused on dermatology, oncology, and infectious diseases in pets. As the global pet population expands, companion animals are anticipated to maintain their dominant position in this market.
Application Analysis
Infectious diseases accounted for 28.4% of growth within application and dominate the veterinary CRO market due to the high prevalence of bacterial, viral, and parasitic infections in animals. Livestock and companion animals frequently face infectious disease risks, which drives continuous demand for new treatments and vaccines.
Veterinary health authorities emphasize disease control to protect animal health and food supply chains. This segment is expected to expand as zoonotic disease concerns increase globally. Research organizations are likely to focus on vaccine development and anti-infective therapies.
The segment benefits from rising awareness of disease prevention and biosecurity measures. Increasing outbreaks and evolving pathogens are projected to support research activity. As infectious disease management remains a priority, this application segment is estimated to retain its leading position in the veterinary CRO market.
End-User Analysis
Pharmaceutical companies accounted for 48.3% of growth within end user and dominate the veterinary CRO market due to their extensive involvement in developing animal health drugs and vaccines. These companies rely on CROs to conduct research, clinical trials, and regulatory processes efficiently.
Outsourcing allows pharmaceutical firms to reduce development timelines and access specialized expertise. The segment is expected to grow as investment in animal health R&D increases. Pharmaceutical companies are likely to expand their pipelines to address emerging diseases and improve treatment options.
The segment benefits from rising demand for innovative therapeutics and biologics. As competition in the animal health industry intensifies, pharmaceutical companies are anticipated to remain the dominant end-user segment in the veterinary CRO market.
Key Market Segments
By Service Type
- Clinical Trials
- Preclinical Studies
- Toxicology Testing
- Regulatory Consulting
- Pharmacovigilance
- Others
By Animal Type
- Companion Animals
- Livestock Animals
- Others
By Application
- Oncology
- Infectious Diseases
- Neurology
- Cardiology
- Orthopedics
- Parasitology
- Vaccine Development
- Others
By End User
- Pharmaceutical Companies
- Biotechnology Companies
- Veterinary Drug Manufacturers
- Research and Academic Institutes
- Others
Drivers
Rising companion animal populations and associated healthcare demands are driving the Veterinary CRO market.
The steady expansion of pet ownership worldwide has generated substantial requirements for outsourced research services in the development and validation of veterinary pharmaceuticals, biologics, and medical devices. According to the American Veterinary Medical Association’s 2025 Pet Ownership and Demographics Sourcebook, the United States recorded 87.3 million dogs and 76.3 million cats in 2025, reflecting continued growth from prior years.
This demographic shift corresponds with elevated spending on veterinary care, averaging $598 per dog-owning household and $529 per cat-owning household annually. Pet owners increasingly seek advanced therapeutic options, prompting sponsors to engage contract research organizations for efficient clinical trials and regulatory support.
Livestock sectors also contribute through demands for improved vaccines and feed additives amid intensified production systems. Outsourcing allows pharmaceutical entities to accelerate timelines while accessing specialized expertise in multi-species studies. Regulatory pathways for veterinary products necessitate comprehensive safety and efficacy data, which specialized providers deliver with established infrastructure.
The trend toward personalized animal health solutions further amplifies the volume of studies outsourced. These structural elements collectively underpin consistent utilization of contract research services. In summary, demographic and expenditure patterns establish a robust foundation for sustained market advancement over the 2022–2025 interval.
Restraints
Complex multi-jurisdictional regulatory compliance requirements are restraining the Veterinary CRO market.
Divergent approval standards across major regions impose significant operational challenges on contract research providers and their clients. The United States Food and Drug Administration Center for Veterinary Medicine, the European Medicines Agency under Regulation (EU) 2019/6, and corresponding authorities in Asia-Pacific territories each maintain distinct documentation, labeling, and good laboratory practice expectations.
Navigating these variations extends study timelines and elevates administrative expenditures for dossier preparation and submissions. Smaller contract research organizations often lack the global regulatory infrastructure necessary to support simultaneous multinational programs, thereby limiting competitive participation.
Animal welfare protocols add further layers of justification and oversight, particularly for in vivo investigations. These procedural intricacies increase overall project costs and risk delays in product commercialization.
Resource constraints in emerging markets compound difficulties in achieving harmonized compliance. Such systemic barriers reduce the agility of service delivery and moderate the pace of industry scaling. Consequently, regulatory fragmentation exerts a tangible limiting influence on broader market progression during the reviewed period.
Opportunities
Advancements in specialized outsourcing for biologics and personalized veterinary medicine are creating growth opportunities in the Veterinary CRO market.
The emergence of novel therapeutic modalities, including gene therapies and species-specific biologics, has opened avenues for contract research organizations to provide targeted development support. Sponsors benefit from access to advanced modeling capabilities and specialized facilities without incurring prohibitive internal capital investments.
Opportunities extend to integrated services encompassing preclinical testing, formulation optimization, and post-approval surveillance across companion and production animals. Collaborative frameworks enable smaller biotechnology firms to navigate complex development pathways efficiently.
Expansion into digital health integration and real-world evidence generation further diversifies service portfolios. Alignment with value-oriented animal health initiatives rewards demonstrated acceleration of safe product introductions.
Geographic diversification into high-growth regions supports scaled operations while addressing localized regulatory nuances. These dynamics foster recurring engagements and long-term partnerships between sponsors and service providers.
Enhanced capabilities in data management and analytics contribute to improved study outcomes and client retention. Overall, specialization in innovative segments positions the sector for diversified and resilient expansion.
Impact of Macroeconomic / Geopolitical Factors
Macroeconomic and geopolitical conditions are materially influencing operational models, outsourcing trends, and cost structures within the veterinary CRO market. Rising global expenditure on animal health and increasing outsourcing of research activities are strengthening demand for CRO services, as sponsors seek efficiency and specialized expertise to accelerate development timelines.
At the same time, inflationary pressure is increasing the cost of clinical trials, laboratory operations, and skilled workforce retention, which can compress margins for service providers. Currency fluctuations and trade uncertainties are also affecting cross-border project execution, particularly for studies that rely on multi-region regulatory approvals.
Geopolitical tensions are limiting international collaboration and slowing the movement of biological samples and research materials, which can delay project timelines. Current US tariffs on imported laboratory equipment, electronic systems, and certain research inputs are raising capital expenditure for CRO facilities, especially those dependent on advanced instrumentation.
These cost increases may discourage smaller firms from expanding capacity or entering high-complexity research segments. However, such trade measures are simultaneously encouraging regional infrastructure development and localized research capabilities, improving long-term resilience.
Overall, despite near-term financial and operational challenges, sustained demand for outsourced veterinary research and expanding therapeutic innovation are expected to support continued market progression.
Latest Trends
Increased integration of digital tools and data analytics platforms represents a recent trend in the Veterinary CRO market.
Throughout 2024 and 2025, contract research providers have progressively incorporated advanced digital solutions to streamline study design, monitoring, and reporting processes. This evolution facilitates real-time data capture, remote oversight, and predictive modeling that enhance operational precision while maintaining compliance standards.
Cloud-based platforms enable seamless collaboration across global teams and accelerate regulatory submissions through standardized electronic formats. The approach reduces reliance on traditional manual workflows, thereby improving efficiency in multi-site investigations. Emphasis on secure data handling aligns with evolving privacy expectations in veterinary research.
Industry implementations during this timeframe demonstrate measurable gains in study cycle times and resource optimization. Continued refinement of analytical capabilities supports more robust evidence generation for novel interventions.
This development reflects a broader transition toward technology-enabled service models that prioritize accuracy and scalability. Prominent adoption patterns observed in 2024–2025 indicate a maturing emphasis on intelligent systems within veterinary contract research activities.
Regional Analysis
North America is leading the Veterinary CRO Market
North America accounted for 45.6% of the veterinary CRO market in 2025, driven by the region’s mature animal health research ecosystem and strong presence of pharmaceutical and biotechnology companies engaged in veterinary drug development. Contract research organizations across the United States are increasingly supporting preclinical studies, clinical trials, and regulatory submissions for animal therapeutics, particularly in companion animal segments.
The U.S. Food and Drug Administration approved multiple new animal drug applications and conditional approvals in recent years, reflecting active development pipelines that rely heavily on outsourced research capabilities. Sponsors are outsourcing research activities to improve cost efficiency, accelerate timelines, and access specialized expertise in toxicology, pharmacokinetics, and clinical trial management.
Rising demand for innovative treatments targeting chronic conditions in pets has further expanded the scope of outsourced research services. CROs are also integrating digital tools and data analytics to enhance study design and regulatory compliance.
Academic institutions and research laboratories are collaborating with contract organizations to advance translational animal health research. Increased regulatory complexity has encouraged pharmaceutical companies to rely on CRO partners with strong compliance frameworks. These factors have collectively supported sustained expansion of outsourced veterinary research services across North America in 2025.
The Asia Pacific region is expected to experience the highest CAGR during the forecast period
Asia Pacific is expected to register notable growth over the forecast period as animal health research activity expands alongside pharmaceutical manufacturing and clinical development capabilities. Countries such as China, India, South Korea, and Australia are strengthening their position in global research outsourcing through cost advantages and a growing pool of skilled scientific professionals.
The World Organisation for Animal Health emphasizes the importance of veterinary research and disease control programs in improving livestock productivity and global food security, encouraging investment in animal health innovation across the region. Pharmaceutical companies are increasingly conducting clinical trials and preclinical studies in Asia to access diverse animal populations and accelerate development timelines.
Governments are supporting biotechnology and life sciences sectors through funding programs and regulatory reforms that facilitate research activities. CROs in the region are expanding service portfolios to include advanced analytics, biomarker studies, and regulatory consulting. Partnerships between global sponsors and regional research providers are becoming more frequent.
Academic and veterinary institutions are also contributing to clinical trial infrastructure and scientific expertise. These developments are expected to drive continued growth of outsourced veterinary research services across Asia Pacific in the coming years.
Key Regions and Countries
North America
- The US
- Canada
Europe
- Germany
- France
- The U.K.
- Italy
- Spain
- Russia & CIS
- Rest of Europe
Asia Pacific
- China
- India
- Japan
- South Korea
- ASEAN
- Australia & New Zealand
- Rest of Asia Pacific
Middle East & Africa
- GCC
- South Africa
- Rest of Middle East & Africa
Latin America
- Brazil
- Mexico
- Rest of Latin America
Key Players Analysis
Key participants in the Veterinary CRO Market expand growth by strengthening preclinical research capabilities, forming strategic partnerships with pharmaceutical and biotechnology companies, and offering end-to-end contract research services tailored for animal health studies. Companies invest in advanced laboratory infrastructure, toxicology testing, and regulatory consulting that support efficient veterinary drug development and approval processes.
They also focus on expanding global research networks and data management systems that improve study timelines and compliance standards. Charles River Laboratories represents a prominent participant in the Veterinary CRO Market and operates as a U.S.-based contract research organization that provides preclinical services, safety assessment, and research models for pharmaceutical and animal health companies.
The company emphasizes scientific expertise and integrated service offerings to support veterinary product development. Industry competitors continue to expand service portfolios, strengthen regulatory expertise, and invest in advanced research technologies to accelerate innovation and sustain long-term market growth.
Top Key Players
- Charles River Laboratories International, Inc.
- Labcorp Drug Development (Fortrea)
- IQVIA Holdings Inc.
- PPD, Inc. (Part of Thermo Fisher Scientific)
- Parexel International Corporation
- ICON plc
- Syneos Health, Inc.
- WuXi AppTec Co., Ltd.
- ORA, Inc.
- NAMSA
- VivoPath Sdn Bhd
Recent Developments
- In January 2026, Charles River Laboratories announced the acquisition of K.F. (Cambodia) Ltd. for $510 million to strengthen its in vivo research supply chain. At the same time, the company exercised its option to acquire PathoQuest SAS, indicating a strategic shift toward New Approach Methodologies (NAMs). This includes increased adoption of in vitro and next-generation sequencing technologies aimed at reducing reliance on traditional animal testing while enhancing research efficiency.
- In April 2025, Labcorp, through its drug development division Fortrea, introduced a focused initiative for veterinary vaccine development. The program is designed to optimize clinical trial processes for zoonotic diseases and livestock-related conditions. This aligns with the growing importance of infectious disease applications, as global biosecurity concerns continue to intensify and drive demand for faster and more effective vaccine development solutions.
Report Scope
Report Features Description Market Value (2025) US$ 1.0 Billion Forecast Revenue (2035) US$ 2.2 Billion CAGR (2026-2035) 8.1% Base Year for Estimation 2025 Historic Period 2020-2024 Forecast Period 2026-2035 Report Coverage Revenue Forecast, Market Dynamics, COVID-19 Impact, Competitive Landscape, Recent Developments Segments Covered By Service Type (Clinical Trials, Preclinical Studies, Toxicology Testing, Regulatory Consulting, Pharmacovigilance and Others), By Animal Type (Companion Animals, Livestock Animals and Others), By Application (Oncology, Infectious Diseases, Neurology, Cardiology, Orthopedics, Parasitology, Vaccine Development and Others), By End User (Pharmaceutical Companies, Biotechnology Companies, Veterinary Drug Manufacturers, Research and Academic Institutes and Others) Regional Analysis North America – The US, Canada; Europe – Germany, France, The U.K., Italy, Spain, Russia & CIS, Rest of Europe; Asia Pacific – China, India, Japan, South Korea, ASEAN, Australia & New Zealand, Rest of Asia Pacific; Middle East & Africa – GCC, South Africa, Rest of Middle East & Africa; Latin America – Brazil, Mexico, Rest of Latin America Competitive Landscape Charles River Laboratories International, Inc., Labcorp Drug Development, IQVIA Holdings Inc., PPD, Inc., Parexel International Corporation, ICON plc, Syneos Health, Inc., WuXi AppTec Co., Ltd., ORA, Inc., NAMSA, VivoPath Sdn Bhd. Customization Scope Customization for segments, region/country-level will be provided. Moreover, additional customization can be done based on the requirements. Purchase Options We have three licenses to opt for: Single User License, Multi-User License (Up to 5 Users), Corporate Use License (Unlimited User and Printable PDF) 
-
-
- Charles River Laboratories International, Inc.
- Labcorp Drug Development (Fortrea)
- IQVIA Holdings Inc.
- PPD, Inc. (Part of Thermo Fisher Scientific)
- Parexel International Corporation
- ICON plc
- Syneos Health, Inc.
- WuXi AppTec Co., Ltd.
- ORA, Inc.
- NAMSA
- VivoPath Sdn Bhd










