Global Plastic Injection Molding for Medical Device Market Dow, Inc., SABIC, Eastman Chemical Company, Covestro AG, Rutland Plastic, Evonik Industries AG, Sumitomo Chemical Co., Ltd., Arkema, INEOS Group, Other Key Players and others, By End User (Hospitals, clinics and R&D), By Region and Key Companies - Industry Segment Outlook, Market Assessment, Competition Scenario, Trends and Forecast 2024–2033
- Published date: March 2024
- Report ID: 30431
- Number of Pages: 289
- Format:
-
keyboard_arrow_up
Quick Navigation
Market Overview
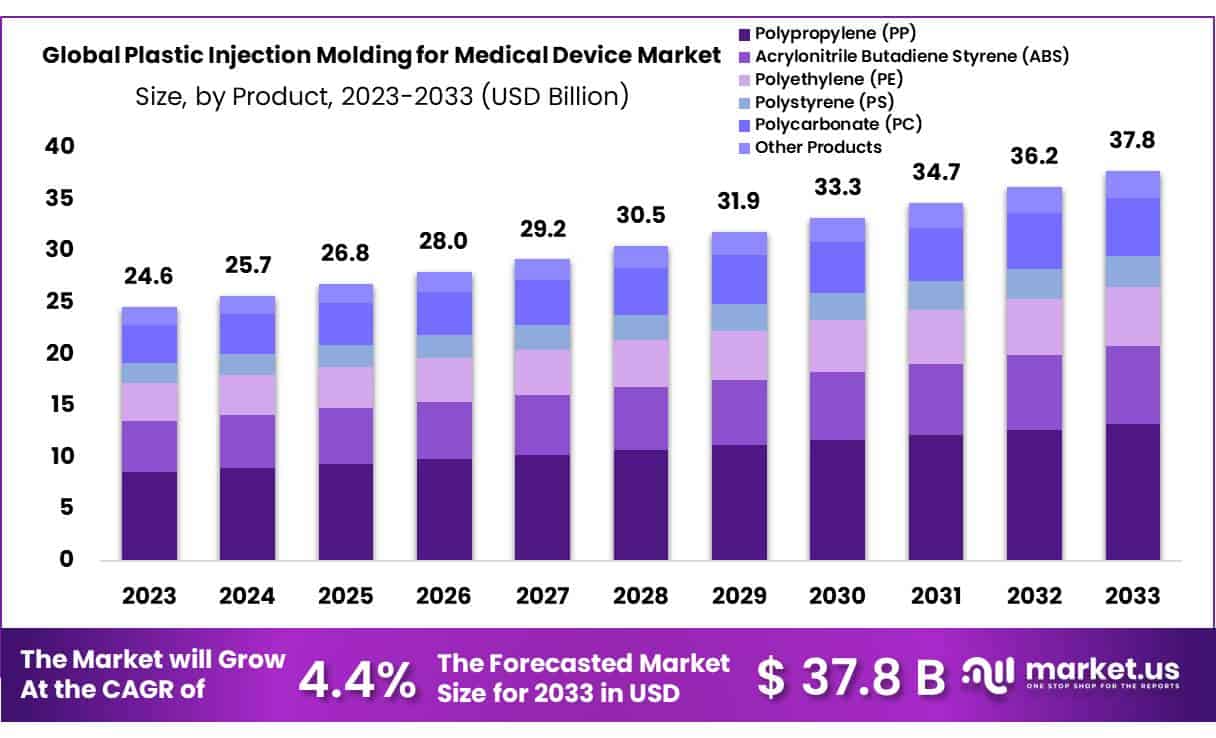
The Global Plastic Injection Molding for Medical Device Market size is expected to be worth around USD 37.8 Billion by 2033 from USD 24.6 Billion in 2023, growing at a CAGR of 4.4% during the forecast period from 2024 to 2033.
Plastic injection molding is the process of manufacturing products and parts by injecting molten materials into moulds. It can be performed with the help of several materials such as metals, glass, elastomers, and more commonly used thermoplastics and thermosetting polymers. Currently, plastic injection molding is considered one of the most versatile techniques of production, owing to several benefits offered by the method as compared to other techniques. Injection moulds are exposed to extremely high pressure, which facilitates production of complex and intricate designs which otherwise will be expensive to manufacture.
Plastic injection molding also provides enhanced strength to products and these can be customized as per customer requirements. Plastic injection provides the advantage of using different types of plastic simultaneously with the help of co-injection molding. Also, plastic injection molding is an automated process that requires minimal human intervention, thereby reducing labour cost and providing additional margins for manufacturers. The demand for the target market is expected to fuel by increasing healthcare spending and expenditure in the coming years
A rise in demand for medical devices is expected to propel their market over the forecasted timeline. According to the World Health Organization (WHO), currently, 2 million units of various types of medical devices exist globally and have been classified into more than 7000 generic device categories. Plastic injection molding has played an instrumental role in shaping the healthcare landscape, accommodating changing requirements while remaining relevant through versatile medical plastics used as injection moldable injection molding materials; applications span from disposable syringes and blood bags through innovative valve replacements and beyond.
Increasing healthcare spending and expenditure, in both developed and developing economies is one of the key factors expected to drive the growth of the global plastic injection molding for the medical device market. Benefits provided by plastic injection molding for medical devices as compared to 3D printing and other conventional manufacturing techniques, providing higher margins to manufacturers are also expected to increase the focus of medical device manufacturers towards this method.
Key Takeaways
- Market Size: Plastic Injection Molding for Medical Device Market size is expected to be worth around USD 37.8 Billion by 2033 from USD 24.6 Billion in 2023.
- Market Growth: The market growing at a CAGR of 4.4% during the forecast period from 2024 to 2033.
- Product Analysis: Polypropylene (PP) emerging as the dominant player, holding 35.1% market share and boasting versatility.
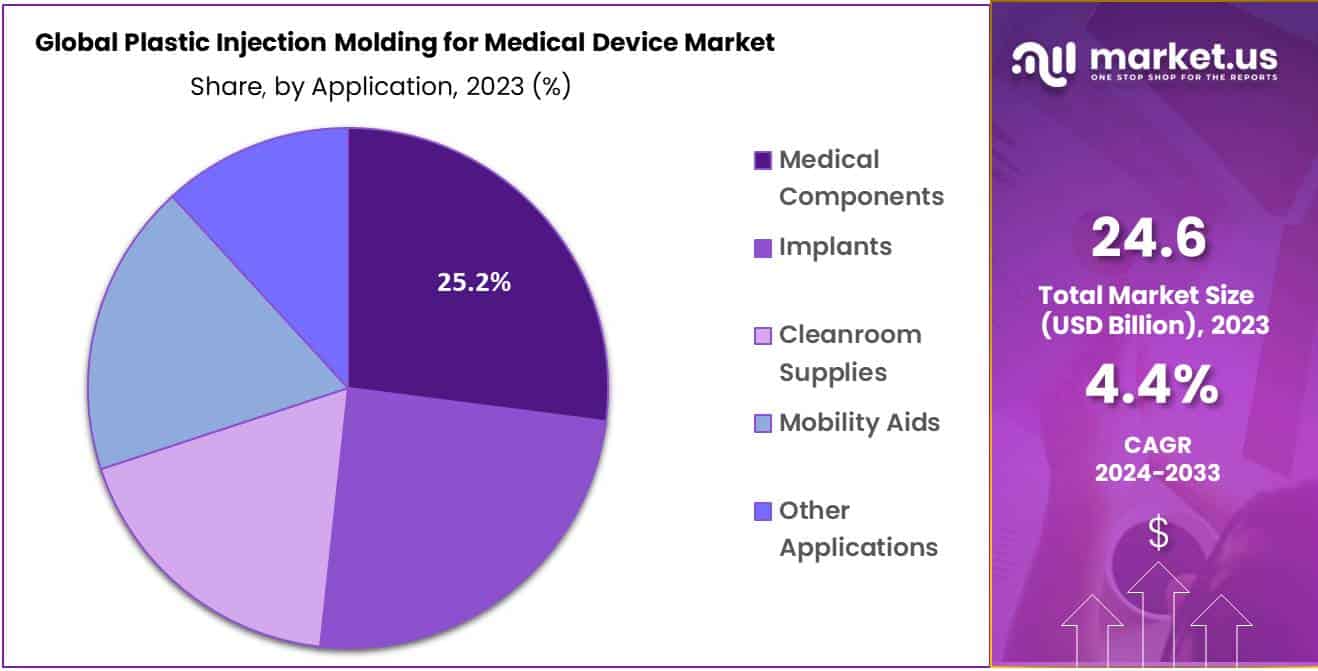
- Application Analysis: Plastic Injection Molding for Medical Device Market, medical components hold an overwhelming 25.2% market share.
- Regional Analysis: North America accounts for the majority share dominate 39.5% market share in 2023.
Product Analysis
Plastic Injection Molding for Medical Device Market is experiencing explosive growth, with Polypropylene (PP) emerging as the dominant player, holding 35.1% market share and boasting versatility, cost-effectiveness and compliance with medical device manufacturing requirements that contributes significantly to its popularity. Other key players in this market segment include Acrylonitrile Butadiene Styrene (ABS), Polyethylene (PE), Polystyrene (PS) and Polycarbonate (PC).
ABS plastic, known for its strength, impact resistance and heat tolerance is widely utilized in medical device manufacturing where durability and reliability are of utmost importance. PE with its flexibility and chemical resistance has applications in various medical devices including containers, tubing and prosthetics; PS is valued for its clarity and ease of processing making it popularly used in packaging and disposable medical devices; while PC stands out due to its exceptional strength and transparency.
As demand for high-quality medical devices continues to increase due to factors like rising healthcare expenditure and advancements in medical technology, the Plastic Injection Molding for Medical Device Market looks set for further expansion – offering exciting prospects for these multifaceted materials.
Application Analysis
Plastic Injection Molding for Medical Device Market, medical components hold an overwhelming 25.2% market share. These components play an essential role in various medical applications ranging from implants and cleanroom supplies to mobility aids and mobility aids. Implant manufacturing must meet stringent medical standards; plastic injection molding provides this precision manufacturing and ensures superior quality, biocompatibility, and durability of these implants.
Cleanroom supplies, essential for keeping medical facilities sterile environments, require durable plastic components to avoid contamination. Plastic injection molding provides precision and consistency when crafting cleanroom supplies such as trays, containers, packaging etc. Mobility aids like wheelchairs, walkers and prosthetic devices rely on plastic components for lightweight yet durable construction, with injection-molded parts providing optimal functionality, comfort and safety for users.
As healthcare continues to change in response to advances in medical technology and a rapidly aging population, demand for injection-molded medical components should rise accordingly. This trend highlights plastic injection molding’s crucial role in providing innovative solutions for medical device applications.
Key Market Segments
Product
- Polypropylene (PP)
- Acrylonitrile Butadiene Styrene (ABS)
- Polyethylene (PE)
- Polystyrene (PS)
- Polycarbonate (PC)
- Other Products
Application
- Medical Components
- Implants
- Cleanroom Supplies
- Mobility Aids
- Other Applications
Driver
Advancements in Medical Technologies and Rising Demand for Medical Devices
Plastic injection molding medical device manufacturing market is experiencing substantial expansion worldwide due to advances in medical technologies and increased consumer interest for these devices. Technological innovations have enabled the creation of advanced medical devices used for diagnosis, monitoring and therapeutic treatments. Plastic injection molding in medical applications has expanded with the integration of innovative polymers that boast increased strength, flexibility, and biocompatibility.
Industry reports predict a global medical device market expected to experience compound annual compound annual growth rate of approximately five percent over the coming decade, driven by factors including an aging population and chronic conditions that necessitate sophisticated interventions and devices for care.
Trend
Growing Adoption of Home Healthcare Services
One noteworthy trend in the plastic injection molding for medical device market is the rise of home healthcare services due to growing patient preference for person-centred care models. With more people opting for home healthcare solutions, demand has grown for portable, user-friendly medical devices that are cost effective – qualities which plastic injection molding excels in providing.
Plastic injection molding techniques offer tremendous design and production versatility that enables manufacturers to design lightweight medical devices suitable for home use that are durable yet user-friendly. Market analysis indicates that home healthcare segment is set for rapid expansion, with projected increases in market value over the coming years. This trend shows the importance of plastic injection molding manufacturers expanding product offerings to cater to this burgeoning sector of the market.
Restriction
Regulatory Challenges and Material Compatibility Issues
Plastic injection molding for medical devices faces stringent regulatory restrictions that can act as a hindrance. Regulation bodies such as the Food and Drug Administration (FDA) of the U.S. and Europe’s Medicines Agency (EMA) impose stringent standards on medical device production processes utilizing injection molding, including those produced through plastic injection molding technology. Compliance with these regulations often necessitates substantial investments in quality assurance and certification processes, potentially impeding market expansion.
Material compatibility issues associated with medical devices present unique obstacles. Ensuring materials don’t negatively impact human health or alter efficacy is no small task and must undergo exhaustive testing and validation, increasing both costs and complexity during production processes.
Opportunity
Sustainable Materials
An emerging opportunity within the plastic injection molding for medical device market lies in adopting eco-friendly and sustainable materials, driven by environmental considerations and regulatory pressures to use biodegradable or bio-based plastics. These materials not only decrease environmental impacts but also offer unique properties like enhanced biocompatibility and lower weight. Market participants have witnessed an upsurge in research initiatives and investments designed to produce eco-friendly polymers which could replace conventional plastics without impacting medical device performance or safety.
Sustainability represents an enormous opportunity for manufacturers, providing an opportunity for innovation and market differentiation. According to industry reports, demand for eco-friendly materials used in medical devices should experience a noticeable surge in 2017-18 – opening new growth paths for players in plastic injection molding sectors like plastic injection molding.
Regional Analysis
On the basis of region, the market is segmented into North America, Europe, Asia Pacific, South America and Middle East & Africa. North America accounts for the majority share dominate 39.5% market share in 2023 in the global plastic injection molding for the medical device market, followed by Europe. Emerging economies, such as China and India, are expected to register the highest growth rate over the forecast period. Countries and regions such as South America & Middle East & Africa are expected to register stable growth over the forecast period.

Key Regions and Countries
North America
- The US
- Canada
- Mexico
Latin America
- Brazil
- Colombia
- Chile
- Argentina
- Costa Rica
- Rest of Latin America
Eastern Europe
- Russia
- Poland
- The Czech Republic
- Greece
- Rest of Eastern Europe
Western Europe
- Germany
- France
- The UK
- Spain
- Italy
- Portugal
- Ireland
- Austria
- Switzerland
- Benelux
- Nordic
- Rest of Western Europe
APAC
- China
- Japan
- South Korea
- India
- Australia & New Zealand
- Indonesia
- Malaysia
- Philippines
- Singapore
- Thailand
- Vietnam
- Rest of APAC
Middle East & Africa
- Algeria
- Egypt
- Israel
- Kuwait
- Nigeria
- Saudi Arabia
- South Africa
- Turkey
- United Arab Emirates
- Rest of MEA
Market Player Analysis
The examination assists stakeholders in sculpting the competitive dynamics within various sectors, offering strategies to navigate the competitive milieu for maximized potential earnings. Moreover, it furnishes a straightforward model for assessing the business organization’s stance. The report’s architecture further emphasizes the competitive arena of the Global Plastic Injection Molding for Medical Device Market.
It meticulously delineates the market share, operational performance, product offerings, and operational status of leading entities, enabling industry participants to pinpoint principal adversaries and comprehend the market’s competitive framework in depth.
Market Key Players
- Dow, Inc.
- SABIC
- Eastman Chemical Company
- Covestro AG
- Rutland Plastic
- Evonik Industries AG
- Sumitomo Chemical Co., Ltd.
- Arkema
- INEOS Group
- Other Key Players
Recent Developments
- Dow: Focuses on biocompatible grades of polyethylene (PE) and polycarbonates (PC) for medical applications.
- SABIC: Introduced a new line of LNP CRX polycarbonate materials in 2020. These materials boast enhanced chemical resistance and mitigate stress cracking, a common issue in medical devices exposed to repeated cleaning.
- Eastman Chemical Company: Known for their Eastman Tritan copolyester, a popular choice for medical device housings due to its clarity and durability.
- Covestro AG: Launched a new line of materials specifically for medical device housings and hardware in December 2021. These materials prioritize biocompatibility, strength, and flame retardancy.
Report Scope
Report Features Description Market Value (2023) USD 24.6 Billion Forecast Revenue (2033) USD 37.8 Billion CAGR (2024-2033) 4.4% Base Year for Estimation 2023 Historic Period 2018-2022 Forecast Period 2024-2033 Report Coverage Revenue Forecast, Market Dynamics, Competitive Landscape, Recent Developments Segments Covered By Product (Polypropylene (PP), Acrylonitrile Butadiene Styrene (ABS), Polyethylene (PE), Polystyrene (PS), Polycarbonate (PC), Other Products) By Application (Medical Components, Implants, Cleanroom Supplies, Mobility Aids, Other Applications) Regional Analysis North America-US, Canada, Mexico;Europe-Germany, UK, France, Italy, Russia, Spain, Rest of Europe;APAC-China, Japan, South Korea, India, Rest of Asia-Pacific;South America-Brazil, Argentina, Rest of South America;MEA-GCC, South Africa, Israel, Rest of MEA Competitive Landscape Dow, Inc., SABIC, Eastman Chemical Company, Covestro AG, Rutland Plastic, Evonik Industries AG, Sumitomo Chemical Co., Ltd., Arkema, INEOS Group, Other Key Players Customization Scope Customization for segments, region/country-level will be provided. Moreover, additional customization can be done based on the requirements. Purchase Options We have three licenses to opt for: Single User License, Multi-User License (Up to 5 Users), Corporate Use License (Unlimited User and Printable PDF) Frequently Asked Questions (FAQ)
What is plastic injection molding?Plastic injection molding is a manufacturing process used to produce intricate and precise parts by injecting molten plastic material into a mold cavity. It is widely utilized in various industries, including medical device manufacturing, due to its ability to create complex geometries with high precision and efficiency.
How big is the Plastic Injection Molding for Medical Device Market?The global Plastic Injection Molding for Medical Device Market size was estimated at USD 24.6 Billion in 2023 and is expected to reach USD 37.8 Billion in 2033.
What is the Plastic Injection Molding for Medical Device Market growth?The global Plastic Injection Molding for Medical Device Market is expected to grow at a compound annual growth rate of 4.4%. From 2024 To 2033
Who are the key companies/players in the Plastic Injection Molding for Medical Device Market?Some of the key players in the Plastic Injection Molding for Medical Device Markets are Dow, Inc., SABIC, Eastman Chemical Company, Covestro AG, Rutland Plastic, Evonik Industries AG, Sumitomo Chemical Co., Ltd., Arkema, INEOS Group, Other Key Players.
Why is plastic injection molding important for the medical device market?Plastic injection molding plays a crucial role in the medical device market by enabling the production of components and parts essential for medical devices. These parts often require strict adherence to quality and regulatory standards, which plastic injection molding can achieve consistently.
 Plastic Injection Molding for Medical Device MarketPublished date: March 2024add_shopping_cartBuy Now get_appDownload Sample
Plastic Injection Molding for Medical Device MarketPublished date: March 2024add_shopping_cartBuy Now get_appDownload Sample -
-
- Dow, Inc.
- SABIC
- Eastman Chemical Company
- Covestro AG
- Rutland Plastic
- Evonik Industries AG
- Sumitomo Chemical Co., Ltd.
- Arkema
- INEOS Group
- Other Key Players










