Global Malignant Mesothelioma Market By Drug Type (Pemetrexed & Combination, Gemcitabine & Combination, Cisplatin & Combination, Carboplatin & Combination, Vinorelbine & Combination and Others), By Route of Administration (Oral and Parenteral), By Distribution Channel (Hospital Pharmacies, Oncology Centers and Retail Pharmacies), Region and Companies – Industry Segment Outlook, Market Assessment, Competition Scenario, Trends and Forecast 2026-2035
- Published date: Feb 2026
- Report ID: 179751
- Number of Pages: 370
- Format:
-
keyboard_arrow_up
Quick Navigation
Report Overview
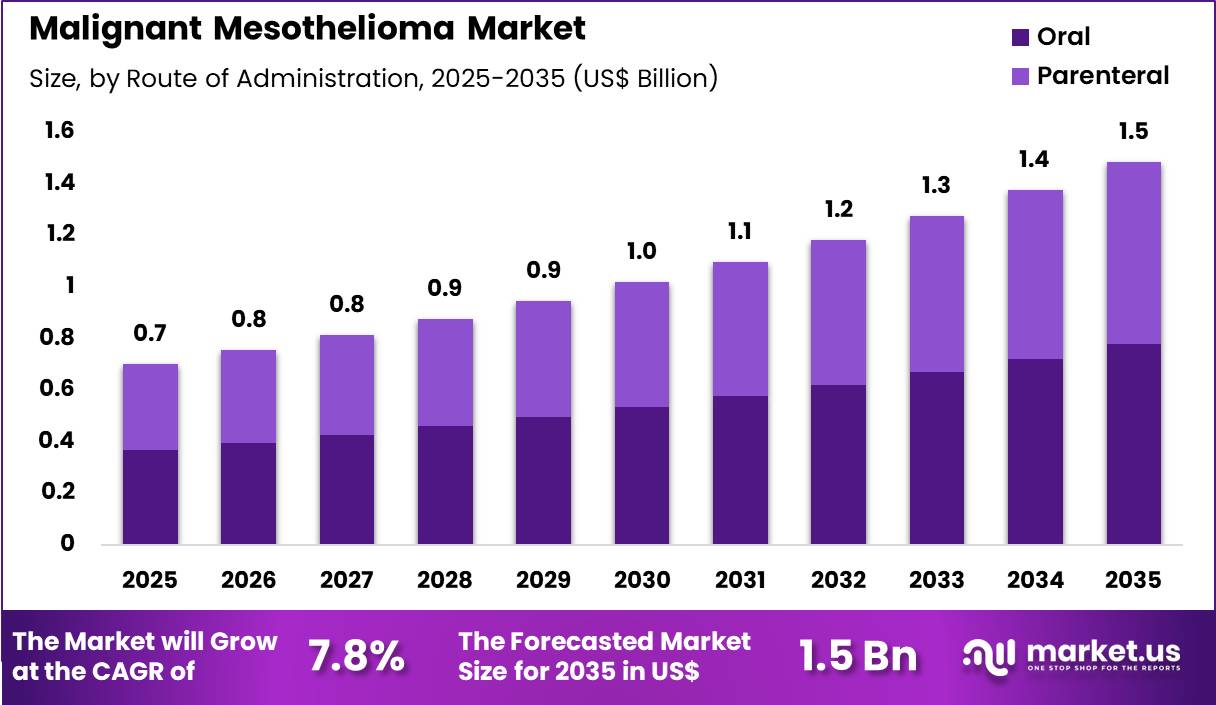
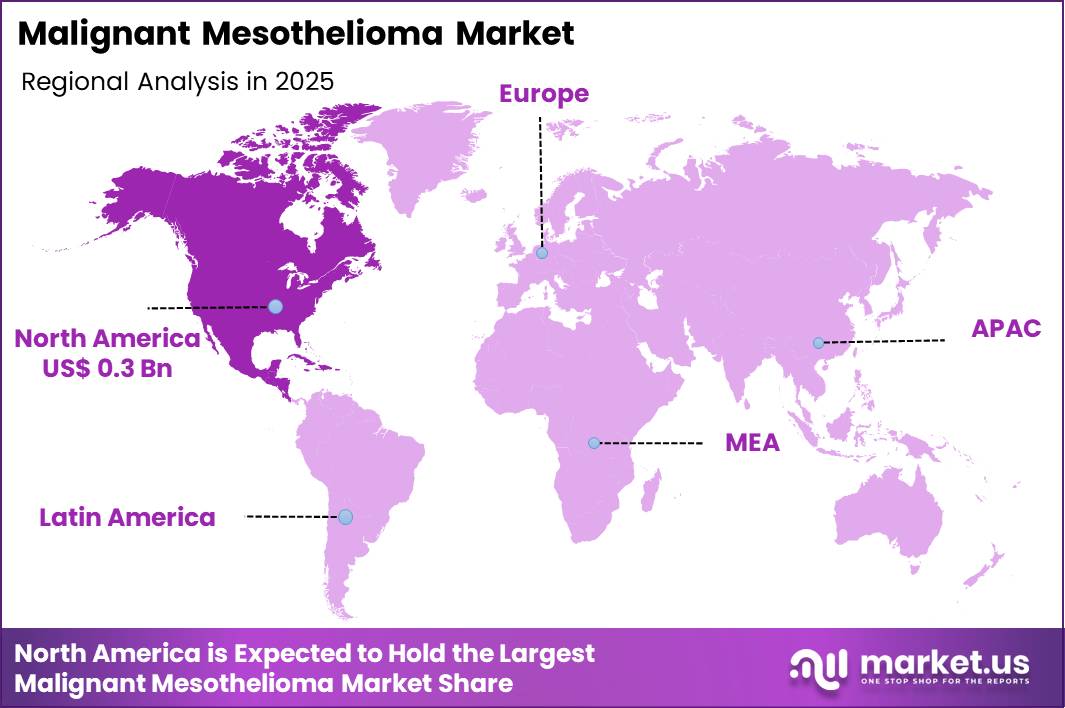
The Global Malignant Mesothelioma Market size is expected to be worth around US$ 1.5 Billion by 2035 from US$ 0.7 Billion in 2025, growing at a CAGR of 7.8% during the forecast period 2026-2035. In 2025, North America led the market, achieving over 42.3% share with a revenue of US$ 0.3 Billion.
Rising incidence of asbestos-related exposures and occupational hazards accelerates the malignant mesothelioma market as patients and healthcare providers demand innovative therapies that address this aggressive cancer’s poor prognosis.
Oncologists increasingly apply multimodal treatments combining surgery, chemotherapy, and radiation to manage pleural mesothelioma, where cytoreductive surgery removes visible tumors from the lung lining while pemetrexed-based chemotherapy targets residual disease to extend survival.
These approaches support applications in peritoneal mesothelioma management, where hyperthermic intraperitoneal chemotherapy delivers heated drugs directly to the abdominal cavity post-debulking, reducing recurrence and improving quality of life.
Immunotherapy with checkpoint inhibitors finds growing use in advanced cases, stimulating immune responses against mesothelioma cells expressing PD-L1 to achieve durable remissions in select patients. Palliative applications involve pleurodesis procedures that seal pleural spaces to alleviate dyspnea from effusions, while targeted therapies inhibit specific mutations in rare subtypes.
Gene therapy trials explore viral vectors to deliver tumor-suppressor genes, offering experimental options for refractory disease. Pharmaceutical companies pursue opportunities to develop bispecific antibodies that engage T-cells against mesothelioma antigens, expanding applications in combination regimens that enhance immunotherapy efficacy.
Developers advance nanoparticle drug delivery systems that improve chemotherapy penetration into tumor microenvironments, broadening utility in localized and metastatic settings. These innovations facilitate personalized medicine through biomarker-driven selections, optimizing outcomes for patients with BAP1 mutations or high tumor mutational burden.
Opportunities emerge in supportive care integrations that combine therapies with nutritional and psychosocial support, addressing holistic needs. Recent trends emphasize real-world evidence collection and collaborative trials, positioning the market for growth in value-based oncology care focused on survival extension and symptom control.
Key Takeaways
- In 2025, the market generated a revenue of US$ 0.7 Billion, with a CAGR of 7.8%, and is expected to reach US$ 1.5 Billion by the year 2035.
- The drug type segment is divided into pemetrexed & combination, gemcitabine & combination, cisplatin & combination, carboplatin & combination, vinorelbine & combination and others, with pemetrexed & combination taking the lead with a market share of 42.6%.
- Considering route of administration, the market is divided into oral and parenteral. Among these, oral held a significant share of 52.4%.
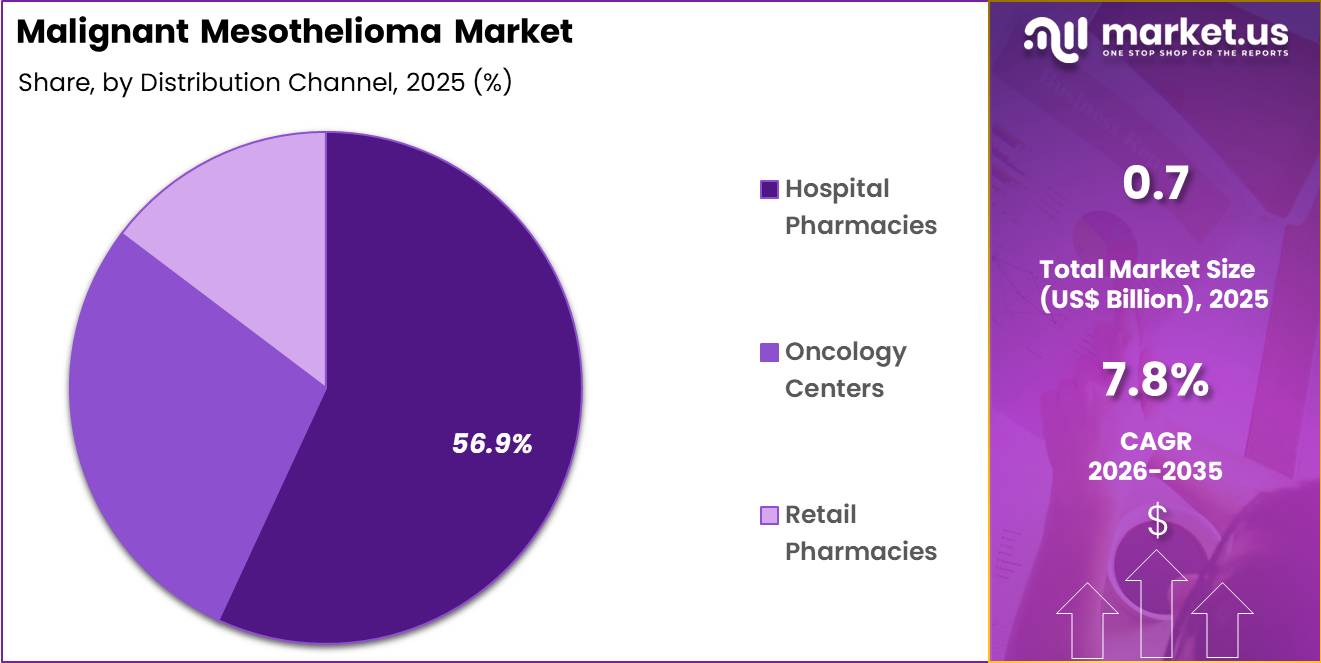
- Furthermore, concerning the distribution channel segment, the market is segregated into hospital pharmacies, oncology centers and retail pharmacies. The hospital pharmacies sector stands out as the dominant player, holding the largest revenue share of 56.9% in the market.
- North America led the market by securing a market share of 42.3%.
Drug Type Analysis
Pemetrexed & combination accounted for 42.6% of growth within drug type and dominate the malignant mesothelioma market due to its proven efficacy in improving overall survival and progression-free survival in first-line treatment. Physicians prefer pemetrexed combinations because they offer manageable toxicity profiles while enhancing patient outcomes.
Segment growth is projected to strengthen as clinical guidelines continue recommending pemetrexed-based regimens, particularly in combination with platinum-based chemotherapies. Pharmaceutical companies increasingly focus on formulation improvements and combination therapies to optimize dosing convenience.
Rising awareness among oncologists and early diagnosis initiatives drive higher utilization. Reimbursement policies and inclusion in hospital formularies further boost adoption. Expanding clinical trials for second-line and maintenance therapies support sustained growth.
Route of Administration Analysis
Oral administration accounted for 52.4% of growth within route of administration and dominates due to patient convenience, adherence benefits, and outpatient suitability. Segment growth is anticipated to continue as oral therapies reduce hospital visits, lower healthcare costs, and improve quality of life for patients undergoing long-term treatment.
Physicians prefer oral regimens for maintenance therapy, follow-ups, and combination protocols. Pharmaceutical development increasingly emphasizes oral formulations with stable bioavailability and minimal side effects. Patient-centric care models and remote monitoring programs strengthen adoption. Growth is likely in regions with robust outpatient oncology services and home-care support.
Distribution Channel Analysis
Hospital pharmacies accounted for 56.9% of growth within distribution channels and dominate due to centralized procurement, formulary inclusion, and direct access to oncology patients. Hospitals provide integrated care, combining chemotherapy administration, supportive care, and monitoring, ensuring optimal treatment adherence.
Segment growth is projected to continue as hospitals expand oncology departments and streamline pharmacy services. Strong relationships with pharmaceutical manufacturers and bulk purchasing agreements enhance availability.
Hospital pharmacies also support clinical trial enrollment and early access programs, further increasing utilization. Expansion of specialized cancer centers within hospitals strengthens this segment’s growth globally.
Key Market Segments
By Drug Type
- Pemetrexed & Combination
- Gemcitabine & Combination
- Cisplatin & Combination
- Carboplatin & Combination
- Vinorelbine & Combination
- Otherss
By Route of Administration
- Oral
- Parenteral
By Distribution Channel
- Hospital Pharmacies
- Oncology Centers
- Retail Pharmacies
Drivers
Increasing incidence of malignant mesothelioma is driving the market.
The gradual rise in diagnosed cases of malignant mesothelioma has significantly boosted the demand for diagnostic tools, treatments, and supportive care in affected populations. Enhanced surveillance and reporting systems have led to better identification of asbestos-related cancers, expanding the patient pool requiring medical intervention.
Healthcare providers are increasingly focusing on multidisciplinary approaches to manage this aggressive disease. The correlation between historical asbestos exposure and current incidence rates underscores the market’s growth for therapeutic options. Government occupational health programs continue to monitor exposed workers, contributing to case detection.
Malignant mesothelioma’s high mortality rate emphasizes the urgency for effective therapies and palliative care. National cancer registries document stable but persistent incidence, prompting investment in research infrastructure.
Key pharmaceutical companies are developing targeted treatments to address this clinical need. This driver fosters collaboration between regulators and industry for accelerated drug approvals. In 2022, there were 2,669 reported cases of malignant mesothelioma in the United States.
Restraints
Limited approved treatment options is restraining the market.
The scarcity of FDA-approved therapies specifically for malignant mesothelioma restricts market expansion and patient access to effective interventions. High failure rates in clinical trials for new agents contribute to slow progress in the therapeutic landscape. Healthcare providers often rely on off-label use of chemotherapy, leading to suboptimal outcomes.
The correlation between disease aggressiveness and limited response to existing treatments further constrains market growth. Government research funding, while present, has not yet yielded sufficient breakthroughs. Malignant mesothelioma’s rarity complicates large-scale trial recruitment and data generation.
National guidelines highlight the need for more options beyond pemetrexed-platinum combinations. Key stakeholders face challenges in justifying investments given the small patient population. This restraint impacts pharmaceutical innovation and market attractiveness. The limited approved treatment options remain a major market restraint.
Opportunities
Advancements in immunotherapy research is creating growth opportunities.
The progress in immunotherapy trials for malignant mesothelioma offers substantial potential for new treatment modalities to enter the market. Governmental funding for oncology research supports the development of checkpoint inhibitors and combination regimens. Increasing clinical evidence for improved survival with immuno-chemotherapy amplifies prospects for regulatory approvals.
Partnerships between biotech firms and academic centers facilitate biomarker-driven immunotherapy studies. The large unmet need in nonepithelioid subtypes magnifies opportunities for targeted immune therapies. Educational programs for oncologists promote inclusion of immunotherapy in treatment protocols. This opportunity enables expansion into combination therapies with existing standards.
Key companies are investing in phase III trials to validate immunotherapy efficacy. Overall, immunotherapy advancements align with efforts to improve long-term outcomes. Advancements in immunotherapy represent a key growth opportunity.
Impact of Macroeconomic / Geopolitical Factors
Macroeconomic trends shape the malignant mesothelioma market through healthcare spending, insurance coverage, and research funding allocation. Rising inflation and interest rates increase costs for drug development and clinical trials, which can slow pipeline progression and treatment accessibility. Geopolitical tensions disrupt global supply chains for key chemotherapeutics and biologics, raising operational risks for manufacturers.
Current US tariffs on imported active pharmaceutical ingredients and oncology equipment further elevate treatment costs and impact hospital budgets. These pressures may limit access in smaller healthcare centers and slow adoption of combination therapies.
On the positive side, tariffs stimulate local production, partnerships, and investment in domestic R&D facilities, enhancing supply security. Growing awareness, early diagnosis programs, and demand for targeted therapies sustain long-term market growth. With strategic collaborations, pricing models, and innovative therapies, the market remains on a steady growth trajectory.
Latest Trends
FDA approval of new combination therapies is a recent trend in the market.
In 2024, the authorization of novel immunotherapy combinations has transformed first-line treatment for unresectable pleural mesothelioma. These approvals integrate checkpoint inhibitors with chemotherapy for improved overall survival. Manufacturers have prioritized real-world evidence to support expanded indications. Clinical analyses emphasized benefits in nonepithelioid histology subgroups.
The FDA approved Keytruda in combination with chemotherapy in September 2024 for inoperable pleural mesothelioma. This trend focuses on dual immune checkpoint blockade for synergistic effects. Regulatory pathways have adapted to expedite reviews for mesothelioma therapies.
Industry synergies concentrate on biomarker selection for optimal patient response. These evolutions aim to elevate standard care while addressing resistance mechanisms. The approval of new combination therapies represents a pivotal trend in 2024.
Regional Analysis
North America is leading the Malignant Mesothelioma Market
North America held 42.3% of the malignant mesothelioma market in 2024 as ongoing clinical demand for advanced therapies and care pathways increased in response to persistent disease burden and rising diagnostic awareness. In the United States, 2 669 new cases of mesothelioma were reported in 2022, highlighting that despite declining asbestos use, significant numbers of diagnoses continue to drive demand for effective treatment regimens and supportive care infrastructure.
Historical asbestos exposure among aging industrial workers sustained a pool of high‑risk individuals whose disease manifestations often emerge decades after initial contact, prompting healthcare providers to invest in multidisciplinary oncology services. Healthcare systems expanded access to immunotherapies and multimodal treatment protocols, improving clinician confidence in managing this aggressive cancer type.
Research programs and institutional cancer centers in the U.S. and Canada accelerated clinical trials, creating pipelines for innovative agents that address unmet treatment needs. Enhanced diagnostic capabilities, including molecular profiling techniques, increased early detection rates in specialized centers.
Insurance coverage adjustments reduced financial barriers for patients to access newer therapies, boosting treatment uptake. Collaboration among oncologists, pulmonologists, and surgical teams optimized care pathways for complex mesothelioma cases. These cumulative factors supported steady regional growth in mesothelioma‑related clinical engagement throughout 2024.
The Asia Pacific region is expected to experience the highest CAGR during the forecast period
Healthcare capacity building across Asia Pacific is projected to strengthen over the forecast period as expanding cancer care networks and public health initiatives elevate treatment availability and disease monitoring capabilities. Australia recorded 684 diagnosed cases of mesothelioma in 2024, underscoring a continuing clinical presence of this rare cancer linked to historical asbestos exposure and drawing attention to persistent unmet medical needs.
Growing populations and increasing longevity in countries such as China, Japan, and South Korea are amplifying demand for oncology services capable of addressing rare malignancies alongside more prevalent cancers. Investments in comprehensive cancer centers and specialized training for thoracic oncology teams are improving diagnostic throughput and treatment delivery.
Governments and private stakeholders are enhancing surveillance systems and registries, ensuring more accurate capture of incidence and outcomes data, which supports targeted care planning. Pet owners and farming communities also represent emerging constituencies for environmental exposure awareness campaigns and early screening.
Healthcare insurers are broadening coverage for cancer treatments, including advanced targeted and immunotherapeutic options. Cross‑border research collaborations are aligning clinical expertise across the region, facilitating knowledge transfer and trials participation.
Urbanization trends and improved access to tertiary care facilities are shortening referral pathways for patients presenting with complex respiratory symptoms. As a result, demand for mesothelioma‑related care services and therapeutic solutions in Asia Pacific is expected to gain momentum through the forecast horizon.
Key Regions and Countries
North America
- US
- Canada
Europe
- Germany
- France
- The UK
- Spain
- Italy
- Russia
- Netherland
- Rest of Europe
Asia Pacific
- China
- Japan
- South Korea
- India
- Australia
- New Zealand
- Singapore
- Thailand
- Vietnam
- Rest of APAC
Latin America
- Brazil
- Mexico
- Rest of Latin America
Middle East & Africa
- South Africa
- Saudi Arabia
- UAE
- Rest of MEA
Key Players Analysis
Key competitors in the malignant mesothelioma market grow by advancing clinical pipelines with novel immunotherapies, targeted agents, and combination regimens that aim to extend survival and improve quality of life beyond current standards.
They also strengthen evidence generation through large‑scale, multicenter clinical programs that support broader regulatory submissions and help differentiate therapeutic profiles for oncologists and payers. Firms pursue strategic alliances with academic research networks and biotechnology innovators to access cutting‑edge science and accelerate early‑phase development while sharing risk for high‑innovation assets.
Companies expand commercial reach by engaging global key opinion leaders and building comprehensive medical education initiatives that reinforce guideline awareness and appropriate use in specialized oncology centers. Geographic expansion into North America, Europe, and select Asia Pacific markets diversifies revenue exposure and captures rising investments in rare cancer care infrastructure and early detection programs.
AstraZeneca plc exemplifies a major diversified biopharmaceutical company with a robust oncology portfolio and coordinated research strategy that includes mesothelioma‑focused immunotherapy combinations, global development expertise, and a commercialization model designed to align scientific innovation with unmet clinical needs in thoracic oncology.
The company advances its competitive agenda through disciplined R&D investment, targeted partnerships, and a customer‑centric approach that translates emerging science into tangible clinical and health‑economic value.
Top Key Players
- Novocure
- Bristol-Myers Squibb
- Merck & Co.
- F. Hoffmann-La Roche
- AstraZeneca
- Eli Lilly and Company
- Pfizer
- Boehringer Ingelheim
- Takeda Pharmaceutical Company
- Amgen
Recent Developments
- In June 2025, Ono Pharmaceutical and Bristol Myers Squibb’s Japanese subsidiary obtained supplemental approval in Japan for the Opdivo (nivolumab) and Yervoy (ipilimumab) combination therapy, expanding its indication to include patients with unresectable hepatocellular carcinoma and strengthening immunotherapy options in oncology treatment pathways.
- In September 2024, the U.S. Food and Drug Administration granted approval to Merck’s KEYTRUDA (pembrolizumab) in combination with pemetrexed and platinum-based chemotherapy as a first-line treatment for adults with unresectable advanced or metastatic malignant pleural mesothelioma, reinforcing the role of combination immunotherapy regimens in managing aggressive thoracic cancers.
Report Scope
Report Features Description Market Value (2025) US$ 0.7 Billion Forecast Revenue (2035) US$ 1.5 Billion CAGR (2026-2035) 7.8% Base Year for Estimation 2025 Historic Period 2020-2024 Forecast Period 2026-2035 Report Coverage Revenue Forecast, Market Dynamics, COVID-19 Impact, Competitive Landscape, Recent Developments Segments Covered By Drug Type (Pemetrexed & Combination, Gemcitabine & Combination, Cisplatin & Combination, Carboplatin & Combination, Vinorelbine & Combination and Others), By Route of Administration (Oral and Parenteral), By Distribution Channel (Hospital Pharmacies, Oncology Centers and Retail Pharmacies) Regional Analysis North America – US, Canada; Europe – Germany, France, The UK, Spain, Italy, Russia, Netherlands, Rest of Europe; Asia Pacific – China, Japan, South Korea, India, Australia, New Zealand, Singapore, Thailand, Vietnam, Rest of APAC; Latin America – Brazil, Mexico, Rest of Latin America; Middle East & Africa – South Africa, Saudi Arabia, UAE, Rest of MEA Competitive Landscape Novocure, Bristol-Myers Squibb, Merck & Co., F. Hoffmann-La Roche, AstraZeneca, Eli Lilly and Company, Pfizer, Boehringer Ingelheim, Takeda Pharmaceutical Company, Amgen Customization Scope Customization for segments, region/country-level will be provided. Moreover, additional customization can be done based on the requirements. Purchase Options We have three licenses to opt for: Single User License, Multi-User License (Up to 5 Users), Corporate Use License (Unlimited User and Printable PDF)  Malignant Mesothelioma MarketPublished date: Feb 2026add_shopping_cartBuy Now get_appDownload Sample
Malignant Mesothelioma MarketPublished date: Feb 2026add_shopping_cartBuy Now get_appDownload Sample -
-
- Novocure
- Bristol-Myers Squibb
- Merck & Co.
- F. Hoffmann-La Roche
- AstraZeneca
- Eli Lilly and Company
- Pfizer
- Boehringer Ingelheim
- Takeda Pharmaceutical Company
- Amgen










