Global Lyophilization Equipment and Services Market By Product Type (Lyophilizers, Vacuum Pumps, Loading and Unloading Systems and Accessories and Consumables), By Application (Pharmaceutical and Biotechnology, Food Processing, Diagnostic Testing, Environmental Testing, Cosmetics and Nutraceuticals and Others), By End User (Pharmaceutical and Biotechnology Companies, CROs and CMOs/CDMOs, Academic and Research Institutes, Food and Beverage Companies, Diagnostic Laboratories and Others), Region and Companies – Industry Segment Outlook, Market Assessment, Competition Scenario, Trends and Forecast 2026-2035
- Published date: April 2026
- Report ID: 184720
- Number of Pages: 212
- Format:
-
keyboard_arrow_up
Quick Navigation
Report Overview
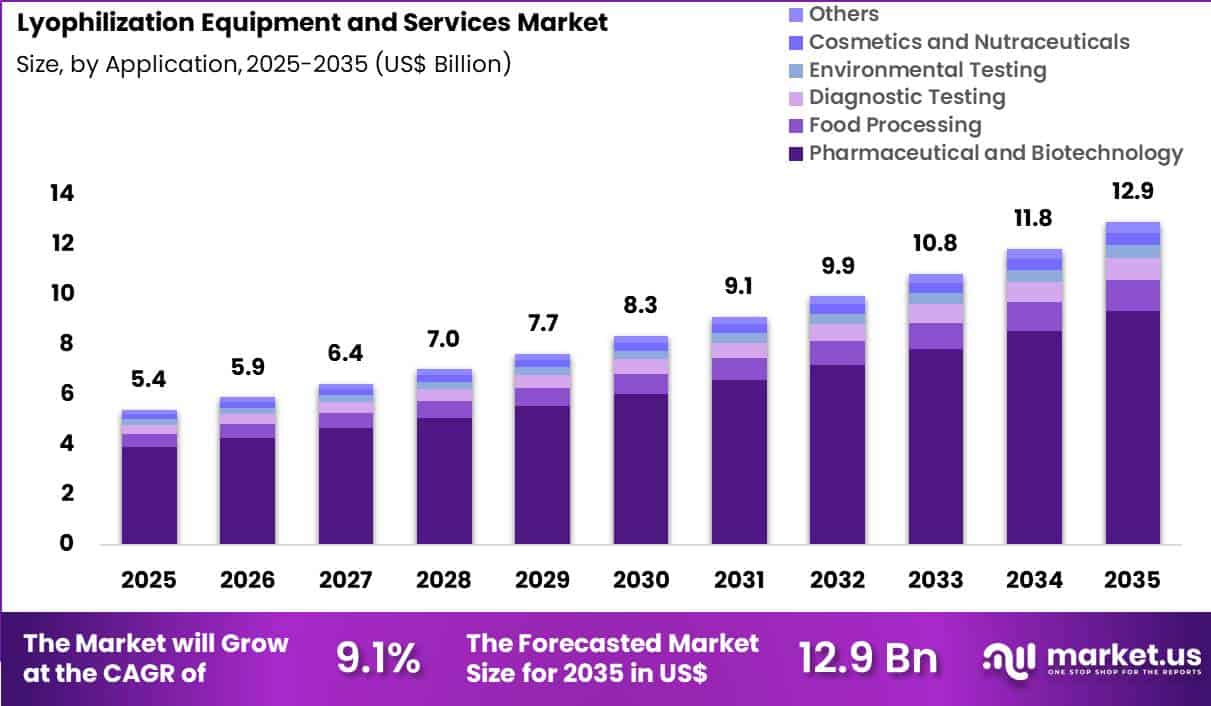
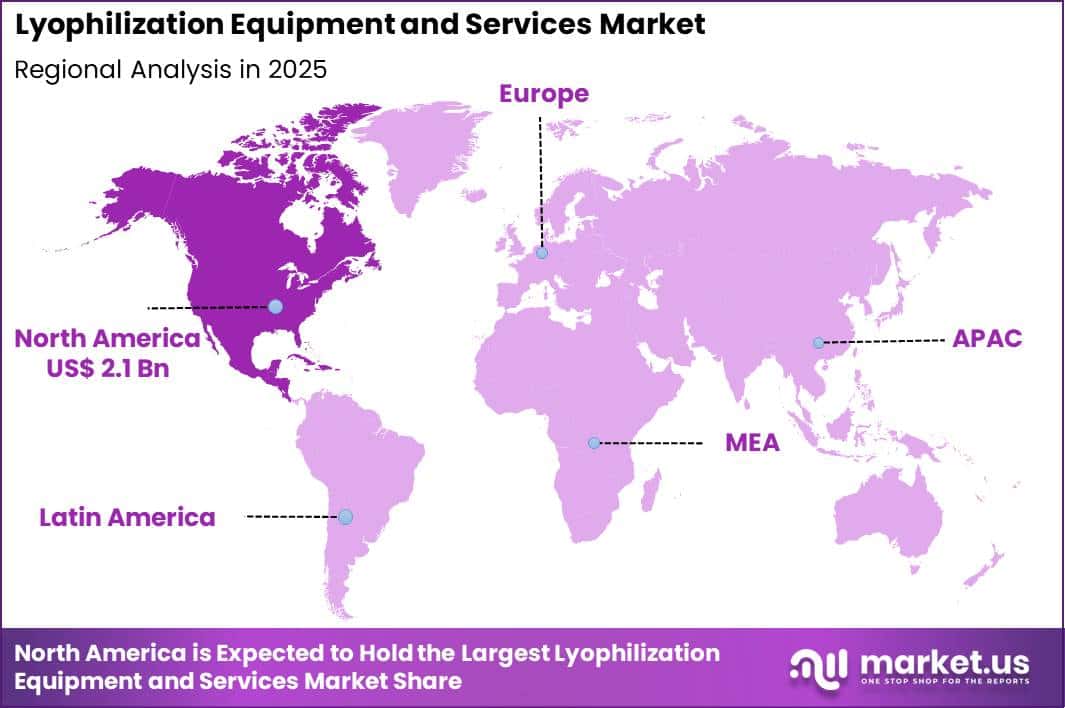
The Global Lyophilization Equipment and Services Market size is expected to be worth around US$ 12.9 Billion by 2035 from US$ 5.4 Billion in 2025, growing at a CAGR of 9.1% during the forecast period 2026-2035. In 2025, North America led the market, achieving over 39.4% share with a revenue of US$ 2.1 Billion.
Increasing complexity of biologic drug formulations and stringent requirements for product stability accelerate the Lyophilization Equipment and Services market as pharmaceutical manufacturers seek advanced solutions that preserve the integrity, potency, and shelf life of sensitive injectable and biological products.
Biopharmaceutical companies increasingly utilize lyophilization equipment to convert liquid formulations of vaccines, monoclonal antibodies, and recombinant proteins into stable dry powders, eliminating the need for cold-chain storage and extending usability in clinical and commercial settings.
These systems support the development of highly potent oncology drugs and orphan medicines, where precise control of temperature, vacuum, and pressure during primary and secondary drying ensures uniform cake structure and minimal degradation.
Contract development and manufacturing organizations rely on lyophilization services to scale production of sterile injectables, offering cycle optimization, formulation development, and fill-finish capabilities that reduce time-to-market for emerging therapies.
In addition, lyophilization equipment plays a critical role in diagnostic reagent production, preserving enzyme activity and antigen stability for long-term storage and consistent test performance.
Manufacturers pursue opportunities to implement continuous lyophilization platforms that replace traditional batch processes with real-time monitoring and improved process efficiency. These advancements support Process Analytical Technology initiatives by integrating sensors and data analytics that optimize critical quality attributes throughout the drying cycle.
In May 2025, SP Scientific, a part of ATS Corporation, introduced the LyoConstellation system with advanced continuous freeze-drying capabilities. This innovation aligns with updated Process Analytical Technology guidelines issued by the U.S. Food and Drug Administration, encouraging a transition from traditional batch processing to continuous, real-time monitored manufacturing.
The shift is expected to improve process efficiency, product consistency, and scalability across pharmaceutical production environments in 2026. Recent trends emphasize automation, energy-efficient designs, and hybrid systems that combine lyophilization with fill-finish operations, positioning the market for growth in scalable, compliant, and cost-effective production of high-value biologics and sterile pharmaceuticals.
Key Takeaways
- In 2025, the market generated a revenue of US$ 5.4 Billion, with a CAGR of 9.1%, and is expected to reach US$ 12.9 Billion by the year 2035.
- The product type segment is divided into lyophilizers, vacuum pumps, loading and unloading systems and accessories and consumables, with lyophilizers taking the lead with a market share of 58.1%.
- Considering application, the market is divided into pharmaceutical and biotechnology, food processing, diagnostic testing, environmental testing, cosmetics and nutraceuticals and others. Among these, pharmaceutical and biotechnology held a significant share of 72.4%.
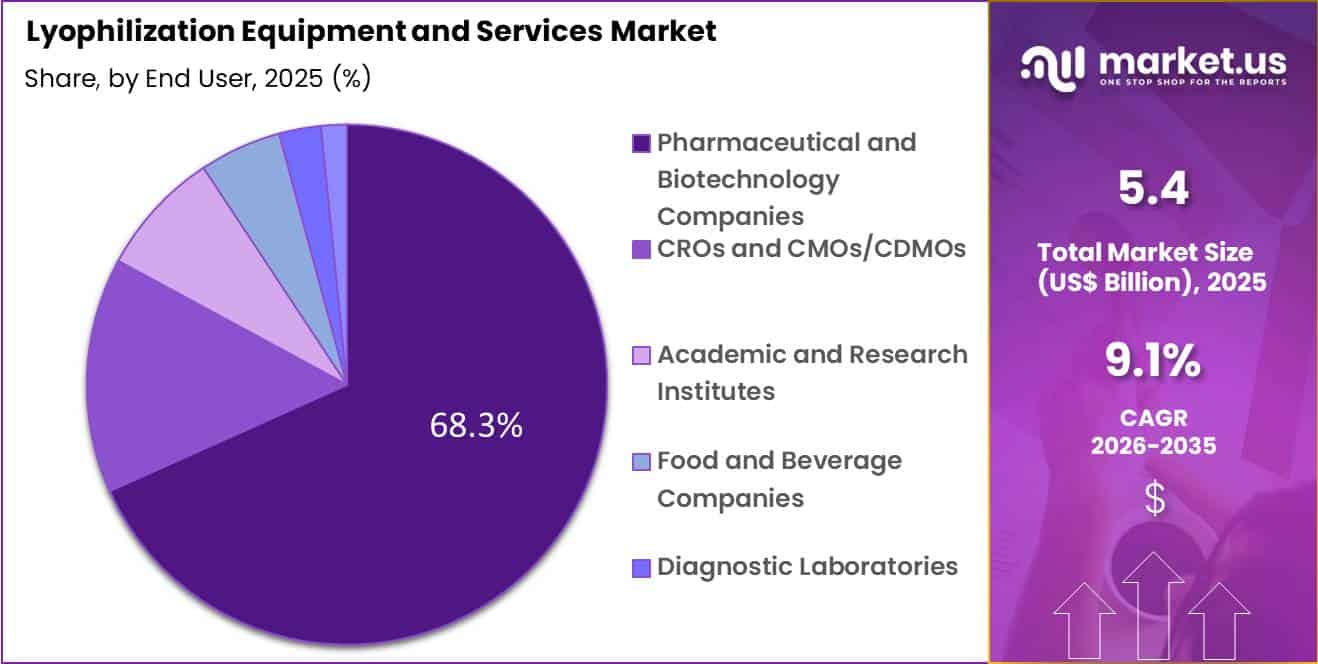
- Furthermore, concerning the end user segment, the market is segregated into pharmaceutical and biotechnology companies, CROs and CMOs/CDMOs, academic and research institutes, food and beverage companies, diagnostic laboratories and others. The pharmaceutical and biotechnology companies sector stands out as the dominant player, holding the largest revenue share of 68.3% in the market.
- North America led the market by securing a market share of 39.4%.
Product Type Analysis
Lyophilizers accounted for 58.1% of growth within product type and dominate the lyophilization equipment and services market due to their central role in freeze-drying processes across pharmaceutical and biotechnology applications. These systems enable removal of moisture under controlled temperature and pressure conditions, which preserves product stability and extends shelf life.
Pharmaceutical manufacturers rely heavily on lyophilizers for biologics, vaccines, and injectable drugs that require long-term storage without degradation. The segment is expected to expand as demand for biologics and complex drug formulations continues to increase globally.
Lyophilizers are likely to remain preferred because they ensure product integrity and support sterile manufacturing environments. The segment benefits from technological advancements such as automated control systems and improved energy efficiency.
Increasing regulatory emphasis on product stability and quality is projected to support adoption. As pharmaceutical production scales up, lyophilizers are estimated to maintain their dominant position in this market.
Application Analysis
Pharmaceutical and biotechnology accounted for 72.4% of growth within application and dominate the lyophilization equipment and services market due to the widespread use of freeze-drying in drug development and manufacturing. Biopharmaceutical products such as vaccines, monoclonal antibodies, and peptides require stabilization to maintain efficacy, which drives demand for lyophilization processes.
The global increase in biologics development and vaccine production strengthens this segment significantly. Pharmaceutical applications are expected to grow as research pipelines expand and more advanced therapies enter clinical stages. Companies are likely to adopt lyophilization to improve product shelf life and transport stability.
The segment benefits from rising investment in drug innovation and increasing regulatory requirements for product preservation. As demand for temperature-sensitive drugs continues to rise, pharmaceutical and biotechnology applications are anticipated to retain their dominant position in this market.
End-User Analysis
Pharmaceutical and biotechnology companies accounted for 68.3% of growth within end user and dominate the lyophilization equipment and services market due to their extensive involvement in drug development, manufacturing, and commercialization. These companies invest heavily in advanced processing technologies to ensure product quality and compliance with regulatory standards.
Lyophilization plays a critical role in their production workflows, especially for biologics and injectable formulations. This segment is expected to expand as pharmaceutical pipelines continue to grow and production capacities increase.
Companies are likely to adopt advanced lyophilization systems to enhance efficiency and scalability. The segment benefits from strong R&D investment and increasing demand for high-quality pharmaceutical products. As innovation in drug development accelerates, pharmaceutical and biotechnology companies are estimated to maintain their dominant position in this market.
Key Market Segments
By Product Type
- Lyophilizers
- Vacuum Pumps
- Loading and Unloading Systems
- Accessories and Consumables
By Application
- Pharmaceutical and Biotechnology
- Food Processing
- Diagnostic Testing
- Environmental Testing
- Cosmetics and Nutraceuticals
- Others
By End User
- Pharmaceutical and Biotechnology Companies
- CROs and CMOs/CDMOs
- Academic and Research Institutes
- Food and Beverage Companies
- Diagnostic Laboratories
- Others
Drivers
Increasing demand for stable biologics and injectable formulations is driving the Lyophilization Equipment and Services market.
The expansion of biopharmaceutical pipelines has elevated the necessity for reliable preservation methods that maintain the integrity of sensitive molecules during storage and transport. Lyophilization enables long-term stability of vaccines, monoclonal antibodies, and recombinant proteins by removing moisture while preserving structural and functional properties.
Regulatory approvals for lyophilized injectables have shown consistent growth, with the U.S. Food and Drug Administration approving approximately 32 lyophilized drug products in 2022 alone, predominantly in oncology and infectious disease categories.
Between 2012 and 2022, regulatory agencies approved a total of 336 lyophilized drug products. This upward trajectory in approvals underscores the reliance on freeze-drying technology for complex therapeutics that cannot tolerate liquid formulations.
Contract development and manufacturing organizations have expanded capacity to meet sponsor requirements for scalable lyophilization services. The process supports global distribution by eliminating the need for continuous cold-chain logistics in many cases.
Pharmaceutical manufacturers invest in advanced equipment to achieve precise control over critical process parameters such as shelf temperature and chamber pressure. These capabilities directly address formulation challenges associated with heat-sensitive and moisture-labile compounds.
Consequently, the growing biologics sector establishes a foundational driver sustaining market expansion during the 2022–2025 period.
Restraints
High capital investment and operational complexity are restraining the Lyophilization Equipment and Services market.
Acquisition and installation of industrial-scale lyophilizers involve substantial upfront expenditures for specialized chambers, vacuum systems, and control instrumentation. Ongoing maintenance, validation, and compliance with current good manufacturing practice standards add considerable recurring costs that burden smaller manufacturers and emerging biotechnology firms.
Integration of new equipment with existing production lines demands extensive engineering resources and downtime, extending project timelines significantly. The process itself requires skilled operators proficient in cycle development and in-process monitoring to ensure batch consistency and product quality.
Regulatory expectations for robust process validation and documentation further increase administrative overhead. Alternative drying technologies, such as spray drying, present lower-cost options for certain applications, diverting potential demand away from lyophilization in select segments.
Resource constraints in facilities limit the ability to adopt continuous or microwave-assisted systems despite their efficiency advantages. These economic and technical barriers moderate the pace of capacity expansion and technology upgrades across the industry. As a result, such factors impose measurable restraint on accelerated market growth throughout the 2022–2025 timeframe.
Opportunities
Expansion of contract lyophilization services and outsourcing models is creating growth opportunities in the Lyophilization Equipment and Services market.
Contract manufacturing organizations provide specialized expertise and pre-validated infrastructure, allowing sponsors to accelerate development timelines without committing to large-scale internal capital investments. Opportunities arise for end-to-end services encompassing formulation development, cycle optimization, scale-up, and commercial manufacturing under a single provider.
This model supports smaller biotechnology companies entering regulated markets by mitigating risks associated with equipment ownership and regulatory filings. Geographic expansion of contract facilities in North America, Europe, and Asia-Pacific regions facilitates localized production and reduces logistics complexities.
Integration of advanced process analytical technologies enables real-time monitoring and improved yield consistency across batches. Potential exists for modular and flexible equipment designs that accommodate diverse product portfolios within shared facilities.
Alignment with personalized medicine and small-batch orphan drug production enhances versatility and revenue potential. These outsourcing arrangements foster long-term partnerships and recurring service engagements. Overall, the shift toward specialized contract capabilities generates substantial prospects for scalable growth and broader industry participation.
Impact of Macroeconomic / Geopolitical Factors
Macroeconomic shifts and geopolitical developments are exerting a pronounced influence on capital investment, outsourcing trends, and operational efficiency in the lyophilization equipment and services market.
Expansion of biologics, vaccines, and injectable drug pipelines is sustaining demand for freeze-drying technologies, as these products require controlled preservation to maintain stability and shelf life. At the same time, inflationary pressure is increasing the cost of high-value equipment, energy consumption, and validation processes, which is prompting pharmaceutical companies to favor contract service providers over in-house infrastructure.
Currency volatility and global trade uncertainty are complicating procurement of refrigeration systems, vacuum components, and precision instrumentation that rely on cross-border supply networks. Geopolitical tensions are also affecting international collaboration and delaying equipment delivery timelines, particularly for large-scale manufacturing units.
Current US tariffs on imported industrial equipment, electronic systems, and specialized components are increasing capital expenditure for new installations and upgrades, which may slow expansion decisions among smaller manufacturers. These cost pressures can shift demand further toward outsourcing models and consolidated production environments.
However, such trade dynamics are encouraging regional manufacturing, energy-efficient system development, and stronger domestic supply chains. In strategic terms, despite near-term cost escalation and supply constraints, the growing reliance on biologics and contract manufacturing is expected to sustain a structurally resilient and expanding market outlook.
Latest Trends
Adoption of continuous and process analytical technology-enhanced lyophilization represents a recent trend in the Lyophilization Equipment and Services market.
In 2024 and 2025, manufacturers have increasingly implemented continuous freeze-drying systems and advanced process analytical technologies to improve efficiency, reduce cycle times, and enhance product uniformity.
Regulatory submissions analyzed between 2020 and 2023 demonstrate growing utilization of sophisticated in-process controls for primary and secondary drying phases across new drug applications, abbreviated new drug applications, and biologics license applications.
This evolution supports compliance with quality-by-design principles promoted by the U.S. Food and Drug Administration Pharmaceutical Quality for the 21st Century Initiative. Microwave-assisted and automated loading/unloading technologies have gained traction to minimize contamination risks and operator exposure.
Industry implementations during this period emphasize data-rich manufacturing that facilitates predictive modeling and deviation management. The trend aligns with demands for higher throughput while maintaining stringent sterility and stability requirements for injectable biologics.
Continued refinement of sensor integration and control algorithms has enabled more robust scale-up from laboratory to commercial production. Prominent in 2024–2025, these technological advancements continue to modernize lyophilization practices and elevate manufacturing standards within the biopharmaceutical sector.
Regional Analysis
North America is leading the Lyophilization Equipment and Services Market
North America accounted for 39.4% of the lyophilization equipment and services market in 2025, supported by the region’s strong biopharmaceutical manufacturing base and continued expansion of biologics and vaccine production.
Drug manufacturers across the United States are increasingly relying on freeze-drying technologies to stabilize sensitive formulations such as monoclonal antibodies, peptides, and injectable therapeutics.
The U.S. Food and Drug Administration recorded dozens of novel drug approvals in recent years, many of which involve complex biologic formulations that require advanced preservation techniques to maintain efficacy and shelf life. Pharmaceutical companies are investing in large-scale lyophilization systems to support commercial production as well as contract manufacturing operations.
In parallel, service providers are expanding capabilities in process development, cycle optimization, and analytical testing to meet evolving regulatory expectations. The growing pipeline of temperature-sensitive therapies has increased reliance on freeze-drying to ensure product stability during storage and distribution.
Automation and continuous monitoring technologies are improving process efficiency and reproducibility across manufacturing facilities. Academic and research institutions are also utilizing lyophilization in drug development and formulation studies. These factors have collectively reinforced steady growth of freeze-drying technologies across North America in 2025.
The Asia Pacific region is expected to experience the highest CAGR during the forecast period
Asia Pacific is expected to demonstrate strong expansion over the forecast period as pharmaceutical manufacturing capacity and biotechnology research activity accelerate across the region. Countries such as China, India, Japan, and South Korea are strengthening their roles in global drug production through investments in advanced manufacturing infrastructure.
The World Health Organization underscores the importance of stable pharmaceutical formulations in ensuring global access to safe and effective medicines, particularly for vaccines and biologics that require controlled preservation methods.
Regional manufacturers are adopting freeze-drying technologies to enhance product stability and extend shelf life in diverse climatic conditions. Governments are supporting local pharmaceutical industries through incentives, regulatory reforms, and infrastructure development.
Contract development and manufacturing organizations are expanding lyophilization service offerings to meet growing international demand. Research institutions are also advancing formulation science to improve efficiency and reduce processing time.
Equipment suppliers are introducing scalable and cost-efficient systems tailored to emerging markets. These developments are expected to drive sustained adoption of lyophilization technologies across Asia Pacific.
Key Regions and Countries
North America
- The US
- Canada
Europe
- Germany
- France
- The U.K.
- Italy
- Spain
- Russia & CIS
- Rest of Europe
Asia Pacific
- China
- India
- Japan
- South Korea
- ASEAN
- Australia & New Zealand
- Rest of Asia Pacific
Middle East & Africa
- GCC
- South Africa
- Rest of Middle East & Africa
Latin America
- Brazil
- Mexico
- Rest of Latin America
Key Players Analysis
Key participants in the Lyophilization Equipment and Services Market expand growth by developing advanced freeze-drying systems, strengthening partnerships with pharmaceutical and biotechnology companies, and enhancing process optimization services that support sterile drug manufacturing.
Companies invest in automated lyophilizers, scalable production solutions, and energy-efficient technologies that improve drying efficiency and product stability. They also focus on validation services, regulatory compliance support, and contract manufacturing capabilities that accelerate biologics and vaccine production timelines.
GEA Group represents a prominent participant in the Lyophilization Equipment and Services Market and operates as a Germany-based industrial technology company that develops freeze-drying equipment, process engineering solutions, and pharmaceutical production systems for global clients.
The company emphasizes innovation in drying technology and integrated manufacturing solutions to support high-quality drug production. Industry competitors continue to introduce advanced lyophilization systems, expand service offerings, and strengthen pharmaceutical collaborations to drive adoption and sustain long-term market growth.
Top Key Players
- SP Scientific (SP Industries Inc. / ATS Corporation)
- GEA Group AG
- IMA S.p.A.
- BTL Industries Inc.
- Millrock Technology Inc.
- Telstar Life Science Solutions (Azbil Group)
- Labconco Corporation
- Azbil Corporation
- MechaTech Systems Ltd.
- Praxair Surface Technologies (Linde plc)
- Thermo Fisher Scientific Inc.
- Zirbus Technology GmbH
Recent Developments
- In January 2026, GEA Group reported stronger-than-expected financial performance for 2025, supported by rising demand for energy-efficient processing systems. Its LYOVAC ECO Mode and LYOAIR technologies are gaining traction, particularly among European clients, as they offer meaningful reductions in energy consumption. These solutions are increasingly relevant in the context of elevated energy costs and stricter environmental regulations, driving the adoption of sustainable freeze-drying systems.
- Following the revised Annex 11 data integrity guidelines introduced in Q2 2025 by the European Medicines Agency, companies such as Telstar and IMA Life experienced a surge in demand for digitally enabled systems. By early 2026, the market focus has shifted toward advanced digital capabilities, including SCADA integration and IoT-connected sensors. These technologies enhance monitoring precision, optimize sublimation processes, and ensure full traceability of production data, supporting compliance and operational transparency.
Report Scope
Report Features Description Market Value (2025) US$ 5.4 Billion Forecast Revenue (2035) US$ 12.9 Billion CAGR (2026-2035) 9.1% Base Year for Estimation 2025 Historic Period 2020-2024 Forecast Period 2026-2035 Report Coverage Revenue Forecast, Market Dynamics, COVID-19 Impact, Competitive Landscape, Recent Developments Segments Covered By Product Type (Lyophilizers, Vacuum Pumps, Loading and Unloading Systems and Accessories and Consumables), By Application (Pharmaceutical and Biotechnology, Food Processing, Diagnostic Testing, Environmental Testing, Cosmetics and Nutraceuticals and Others), By End User (Pharmaceutical and Biotechnology Companies, CROs and CMOs/CDMOs, Academic and Research Institutes, Food and Beverage Companies, Diagnostic Laboratories and Others) Regional Analysis North America – The US, Canada; Europe – Germany, France, The U.K., Italy, Spain, Russia & CIS, Rest of Europe; Asia Pacific – China, India, Japan, South Korea, ASEAN, Australia & New Zealand, Rest of Asia Pacific; Middle East & Africa – GCC, South Africa, Rest of Middle East & Africa; Latin America – Brazil, Mexico, Rest of Latin America Competitive Landscape SP Scientific, GEA Group AG, IMA S.p.A., BTL Industries Inc., Millrock Technology Inc., Telstar Life Science Solutions, Labconco Corporation, Azbil Corporation, MechaTech Systems Ltd., Praxair Surface Technologies, Thermo Fisher Scientific Inc., Zirbus Technology GmbH. Customization Scope Customization for segments, region/country-level will be provided. Moreover, additional customization can be done based on the requirements. Purchase Options We have three licenses to opt for: Single User License, Multi-User License (Up to 5 Users), Corporate Use License (Unlimited User and Printable PDF)  Lyophilization Equipment and Services MarketPublished date: April 2026add_shopping_cartBuy Now get_appDownload Sample
Lyophilization Equipment and Services MarketPublished date: April 2026add_shopping_cartBuy Now get_appDownload Sample -
-
- SP Scientific (SP Industries Inc. / ATS Corporation)
- GEA Group AG
- IMA S.p.A.
- BTL Industries Inc.
- Millrock Technology Inc.
- Telstar Life Science Solutions (Azbil Group)
- Labconco Corporation
- Azbil Corporation
- MechaTech Systems Ltd.
- Praxair Surface Technologies (Linde plc)
- Thermo Fisher Scientific Inc.
- Zirbus Technology GmbH










