Global Allergy Diagnostics and Therapeutics Market By Type (Diagnostics, Therapeutics), By Allergen Type (Food, Inhaled, Drug, Other Allergens), By Test Type (In vivo Test, In vitro Test), Region and Companies – Industry Segment Outlook, Market Assessment, Competition Scenario, Trends and Forecast 2023-2032
- Published date: March 2024
- Report ID: 117335
- Number of Pages: 340
- Format:
-
keyboard_arrow_up
Quick Navigation
Report Overview
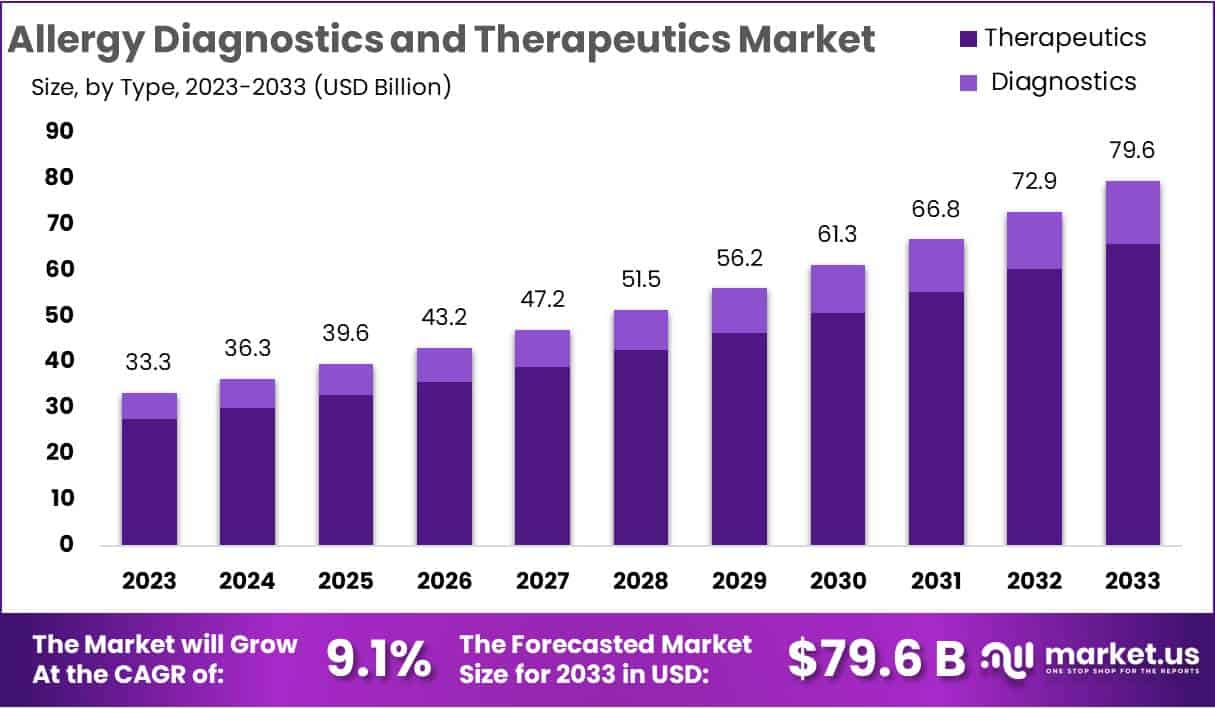
The Global Allergy Diagnostics and Therapeutics Market size is expected to be worth around USD 79.6 Billion by 2033, from USD 33.3 Billion in 2023, growing at a CAGR of 9.1% during the forecast period from 2024 to 2033.
The allergy diagnostics and therapeutics market is a vital segment within the broader healthcare industry, focused on the identification, management, and treatment of allergic conditions. This market encompasses a range of products and services aimed at diagnosing allergies and providing effective therapeutic interventions. Allergy diagnostics involve various techniques such as skin prick tests, blood tests (e.g., IgE testing), and molecular diagnostics to identify specific allergens triggering allergic reactions in individuals. Advanced technologies like allergen microarrays and immunocap assays enhance diagnostic accuracy and enable personalized allergy management.
On the therapeutics side, the market includes medications such as antihistamines, corticosteroids, decongestants, and immunotherapy options like allergy shots and sublingual immunotherapy (SLIT). Immunotherapy plays a significant role in desensitizing individuals to allergens, offering long-term relief from allergic symptoms. The market also sees innovations in biologic therapies targeting specific immune pathways involved in allergic responses, providing more targeted and effective treatments for severe allergies.
Factors driving market growth include the increasing prevalence of allergies worldwide, heightened awareness and diagnosis rates, advancements in diagnostic technologies, and the development of novel therapeutics. However, challenges such as regulatory complexities, reimbursement issues, and the high cost of advanced allergy treatments can impact market dynamics. Overall, the allergy diagnostics and therapeutics market continues to evolve with ongoing research, technological innovations, and a focus on personalized medicine to meet the diverse needs of allergy patients globally.
- According to the Global Allergy & Airways Patient Platform, there has been a consistent rise in the prevalence of allergic diseases on a global scale, with approximately 30-40% of the world’s population currently experiencing one or more allergic conditions.
- According to the American Academy of Allergy, Asthma and Immunology, adverse drug reactions, i.e. drug allergies, could impact around 10% of the global population and affect approximately 20% of hospitalized individuals.
- A research in the Pediatric Allergy and Immunology journal states that Food hypersensitivity (FH) is a rising health concern on a global scale, impacting over 10% of the general population with approximately 1 in 10 adults and 1 in 12 children being susceptible to food allergies.
Key Takeaways
- In 2023, the market for Allergy Diagnostics and Therapeutics generated a revenue of USD 33.3 billion and is poised to reach USD 79.6 billion by the year 2033, with a CAGR of 9.1%.
- Based on type, the therapeutics segment held a significant revenue share of over 82.8% in 2023.
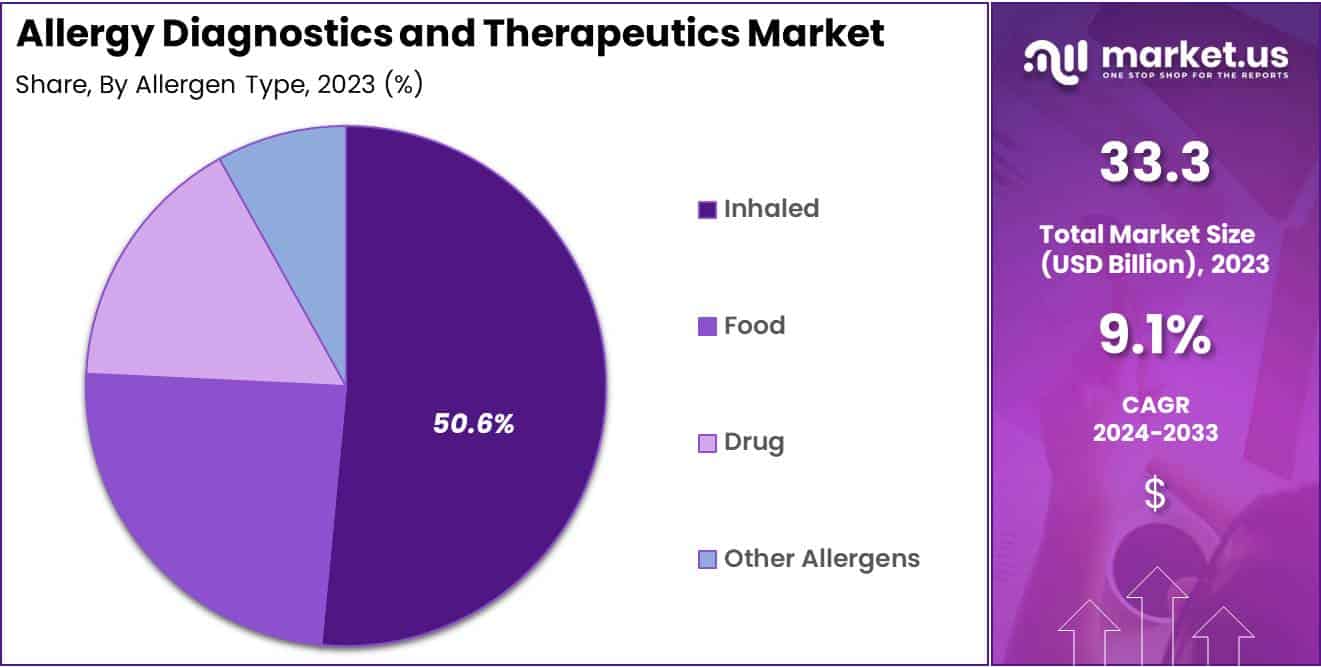
- Among the allergy types, the inhaled allergy segment emerged as the largest contributor, with a share exceeding 50.6% in the same year.
- Additionally, the in vitro test segment accounted for a market share of 53.6% in 2023.
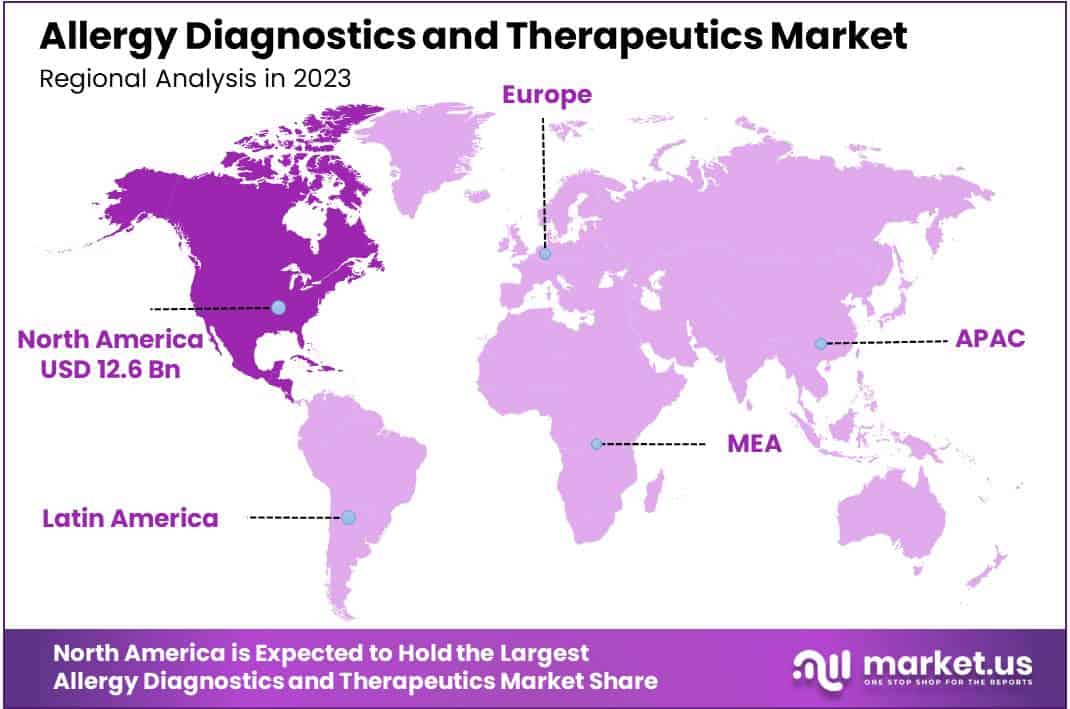
- Region wise, North America maintained stronghold on the market, with a revenue share of 38.1% in 2023.
Type Analysis
In terms of categorization, the market is divided into therapeutics and diagnostics, with the therapeutics segment accounting for a substantial revenue share of more than 82.8% in 2023. This dominance is attributed to therapeutics’ primary focus on treating allergic conditions and providing relief to patients. Among therapeutics, epinephrine emerged as the leading product, commanding a significant portion of the market due to its rapid and potent response, particularly in severe allergic reactions like anaphylaxis.
Epinephrine’s ability to mimic the body’s fight-or-flight response, swiftly alleviating symptoms such as respiratory distress and low blood pressure, along with its widespread availability, cost-effectiveness, and established efficacy over decades, reinforces its position as a vital tool in emergency allergy management.
Allergy Type Analysis
The inhaled allergy segment emerged as the largest revenue contributor, with a share exceeding 50.6% in 2023, driven by the rising prevalence of respiratory allergies linked to industrialization and escalating air pollution levels. According to an article published in the Allergo Journal International, around 400 million individuals worldwide experience allergic rhinitis (AR), while approximately 150 million are impacted by allergic asthma.
The expanding incidence of allergic rhinitis is anticipated to create growth prospects in the future. Meanwhile, drug allergies are projected to witness a higher compound annual growth rate (CAGR) in the upcoming years. Common triggers like chemotherapy drugs, sulfonamide drugs, and antiepileptic drugs are associated with various conditions such as contact dermatitis, a form of skin inflammation. This segment is poised for positive growth due to increased treatment options availability and heightened patient awareness regarding drug allergies.
Test Type Analysis
The in vitro test segment accounted for a market share of 53.6% in 2023. This test is utilized to identify allergen-specific immunoglobulin E (IgE). Serum IgE demonstrates a higher positive predictive value and better specificity in detecting aeroallergen sensitization, such as pollen and insect allergens. These aspects are expected to significantly influence the penetration of the allergy diagnostics market during the forecast period.
Moreover, food allergies are increasingly becoming a public health concern, with escalating rates observed in both developed and developing nations. The in vivo test segment is projected to witness substantial growth with a significant compound annual growth rate (CAGR) over the forecast period.
This growth is attributed to the growing adoption of various in vivo tests that offer convenience, cost-effectiveness, safety, and reliability compared to other methods. The rising demand for rapid, simple, and efficient testing has further propelled the growth of in vivo diagnostic tests.
Key Market Segments
By Type
- Therapeutics
- Antihistamines
- Decongestants
- Corticosteroids
- Mast Cell Stabilizers
- Leukotriene Inhibitors
- Nasal Anti-cholinergic
- Immuno-modulators
- Epinephrine
- Immunotherapy
- Diagnostics
- Instruments
- Consumables
- Services
By Allergen Type
- Inhaled
- Food
- Dairy Products
- Poultry Product
- Tree Nuts
- Peanuts
- Shellfish
- Wheat
- Soys
- Other Food Allergens
- Drug
- Other Allergens
By Test Type
- In vitro Test
- In vivo Test
- Skin Prick Test
- Intradermal Test
- Patch Test
Market Drivers
Rising Prevalence of Allergies
The rising incidence of allergies, encompassing respiratory allergies, food allergies, and drug allergies, is a significant driver in the Allergy Diagnostics and Therapeutics Market. This trend is attributed to various factors such as environmental changes, increased exposure to allergens, genetic predispositions, and lifestyle factors. As allergy rates continue to escalate globally, there is a growing demand for advanced diagnostics that can accurately identify allergens and effective therapeutic solutions to manage allergic conditions, driving market growth in the healthcare sector.
Increasing Healthcare Spending and Infrastructure
The growth in healthcare expenditure, especially in developed regions like North America and Europe, along with expanding healthcare infrastructure in emerging markets like Asia Pacific, is supporting market expansion by increasing access to allergy diagnostics and therapeutics. Higher healthcare spending allows for investment in advanced diagnostic technologies, such as molecular diagnostics and allergen-specific IgE testing, improving accuracy in identifying allergic conditions and guiding appropriate treatment strategies.
Restraints
Regulatory challenges
One of the primary restraints in the Allergy Diagnostics and Therapeutics Market is the complex regulatory landscape governing the development, approval, and commercialization of allergy diagnostics and therapeutics. Regulatory agencies such as the U.S. Food and Drug Administration (FDA), the European Medicines Agency (EMA), and other regional regulatory bodies impose stringent requirements on the safety, efficacy, and quality of diagnostic tests and therapeutic products.
Obtaining regulatory approval for new allergy diagnostics and therapeutics involves extensive clinical trials, preclinical studies, and regulatory submissions. The approval process is time-consuming, costly, and often unpredictable, leading to delays in market entry for innovative products.
High Costs
Another significant restraint in the Allergy Diagnostics and Therapeutics Market is the high costs associated with developing, manufacturing, and accessing advanced diagnostic tests and therapeutic interventions for allergies. Developing new allergy diagnostics and therapeutics requires substantial investment in research, clinical trials, and product development. R&D expenses account for a significant portion of the overall costs in bringing innovative products to market.
Opportunities
One of the main opportunities in the Allergy Diagnostics and Therapeutics Market lies in the increasing focus on precision medicine and personalized healthcare. Advances in molecular diagnostics, genetic testing, and biomarker analysis are paving the way for more accurate and targeted allergy diagnoses and treatments tailored to individual patient needs.
Furthermore, the integration of digital health technologies, such as telemedicine and remote monitoring, offers opportunities to enhance patient engagement, improve treatment outcomes, and expand access to allergy care. Embracing these opportunities can lead to more effective and efficient allergy management strategies, benefiting both patients and healthcare providers.
Impact of Macroeconomic / Geopolitical Factors
Government policies play a crucial role in shaping the impact of rising allergies on the Allergy Diagnostics and Therapeutics Market. Regulations governing healthcare practices, drug approvals, and insurance coverage can influence market dynamics. Trade agreements and taxation policies can impact the availability and cost of allergy diagnostics and therapeutics.
Additionally, healthcare expenditure allocations by governments directly impact the accessibility and affordability of allergy treatments. These factors collectively shape the market environment, affecting the demand, pricing, and innovation in allergy diagnostics and therapeutics.
Latest Trends
The latest trends in the Allergy Diagnostics and Therapeutics Market include the increasing adoption of advanced diagnostic technologies such as molecular diagnostics and allergen microarrays for precise allergen identification.
Additionally, there is a growing focus on personalized medicine approaches, leveraging genetic testing and biomarker analysis to tailor allergy treatments to individual patient needs. Furthermore, the development of biologic therapies targeting specific immune pathways and the integration of digital health solutions for remote monitoring and management of allergies are driving innovation in the market.
Regional Analysis
North America is leading the Allergy Diagnostics and Therapeutics Market
North America’s market dominance, capturing a 38.1% revenue share in 2023, is attributed to several factors including a rise in allergy cases and supportive government initiatives. The region’s advanced healthcare infrastructure and substantial healthcare spending also contributed significantly to its market share. Moreover, U.S.-based companies’ substantial investments in allergy drug development and the entry of new players with innovative products have heightened competition in North America.
The Asia Pacific region is expected to experience the highest CAGR during the forecast period
The Asia Pacific market is expected to witness rapid growth due to increasing healthcare reforms, expanding infrastructure, growing populations, and the entry of local companies. Key markets in the region include Japan, China, and India, where strategic initiatives by key players are driving market growth.
Key Regions and Countries.
- North America
- US
- Canada
- Europe
- Germany
- France
- The UK
- Spain
- Italy
- Russia
- Netherland
- Rest of Europe
- Asia Pacific
- China
- Japan
- South Korea
- India
- New Zealand
- Singapore
- Thailand
- Vietnam
- Rest of APAC
- Latin America
- Brazil
- Mexico
- Rest of Latin America
- Middle East & Africa
- South Africa
- Saudi Arabia
- UAE
- Rest of MEA
Key Players Analysis
The Allergy Diagnostics and Therapeutics Market is dominated by key players such as Thermo Fisher Scientific Inc., and Siemens Healthineers AG. These companies hold significant market shares due to their extensive product portfolios, innovative technologies, and strategic partnerships. Other notable players in the market include Omega Diagnostics Group PLC, bioMérieux SA, and HOB Biotech Group Corp. These players collectively drive market growth and innovation in allergy diagnostics and therapeutics.
Recent Developments
- In September 2023, Nestlé has sold its peanut allergy drug Palforzia to Swiss biopharma company Stallergenes Greer after previously investing significant funds in acquiring the treatment. As per the announcement on 4 September, Nestlé will receive undisclosed milestone payments and ongoing royalties from Stallergenes Greer as part of the agreement.
- In March 2024, bioMérieux has initiated a research partnership with the U.S. Food and Drug Administration’s (FDA) Center for Food Safety and Applied Nutrition (CFSAN) in collaboration with the Offices of Applied Research and Safety Assessment (OARSA) and Regulatory Science (ORS). Their joint aim is to create tools that can effectively address and combat foodborne pathogens.
Top Key Players in Allergy Diagnostics and Therapeutics Market
- Thermo Fisher Scientific, Inc.
- Siemens Healthcare GmbH
- R-Biopharm AG
- DASIT Group SPA
- bioMérieux
- Stallergenes Greer
- HYCOR Biomedical
- Minaris Medical America, Inc.
- Omega Diagnostics Group PLC
- Lincoln Diagnostics
- HOB Biotech Group Corp.
- Other Key Players
Report Scope
Report Features Description Market Value (2023) USD 33.3 billion Forecast Revenue (2033) USD 79.6 billion CAGR (2024-2033) 9.1% Base Year for Estimation 2023 Historic Period 2018-2022 Forecast Period 2024-2033 Report Coverage Revenue Forecast, Market Dynamics, COVID-19 Impact, Competitive Landscape, Recent Developments Segments Covered By Type – Diagnostics, Therapeutics; By Allergen Type – Food, Inhaled, Drug, Other Allergens; By Test Type – In vivo Test, In vitro Test Regional Analysis North America – US, Canada; Europe – Germany, France, The UK, Spain, Italy, Russia, Netherlands, Rest of Europe; Asia Pacific – China, Japan, South Korea, India, New Zealand, Singapore, Thailand, Vietnam, Rest of APAC; Latin America – Brazil, Mexico, Rest of Latin America; Middle East & Africa – South Africa, Saudi Arabia, UAE, Rest of MEA Competitive Landscape R-Biopharm AG, Thermo Fisher Scientific, Inc., DASIT Group SPA, AESKU.GROUP GmbH, bioMérieux, Siemens Healthcare GmbH, Stallergenes Greer, HYCOR Biomedical, Minaris Medical America, Inc., Omega Diagnostics Group PLC, Lincoln Diagnostics, Inc., HOB Biotech Group Corp., Ltd., and Other Key Players Customization Scope Customization for segments, region/country-level will be provided. Moreover, additional customization can be done based on the requirements. Purchase Options We have three licenses to opt for: Single User License, Multi-User License (Up to 5 Users), Corporate Use License (Unlimited User and Printable PDF)  Allergy Diagnostics and Therapeutics MarketPublished date: March 2024add_shopping_cartBuy Now get_appDownload Sample
Allergy Diagnostics and Therapeutics MarketPublished date: March 2024add_shopping_cartBuy Now get_appDownload Sample -
-
- Thermo Fisher Scientific, Inc.
- Siemens Healthcare GmbH
- R-Biopharm AG
- DASIT Group SPA
- bioMérieux
- Stallergenes Greer
- HYCOR Biomedical
- Minaris Medical America, Inc.
- Omega Diagnostics Group PLC
- Lincoln Diagnostics
- HOB Biotech Group Corp.
- Other Key Players









