Global Pharmaceutical Contract Packaging Market Size, Share, Growth Analysis By Service Type (Primary, Secondary, and Tertiary), By Drug Formulation (Solid Dosage, Oral Liquids, and Injectable), By End-Use (Big Pharma, Generics and Biosimilar Companies, Emerging Biotech and Start-ups, CRO and CDMO Partners, and Others), By Region and Companies - Industry Segment Outlook, Market Assessment, Competition Scenario, Trends and Forecast 2025-2035
- Published date: May 2026
- Report ID: 186051
- Number of Pages: 371
- Format:
-
keyboard_arrow_up
Quick Navigation
Report Overview
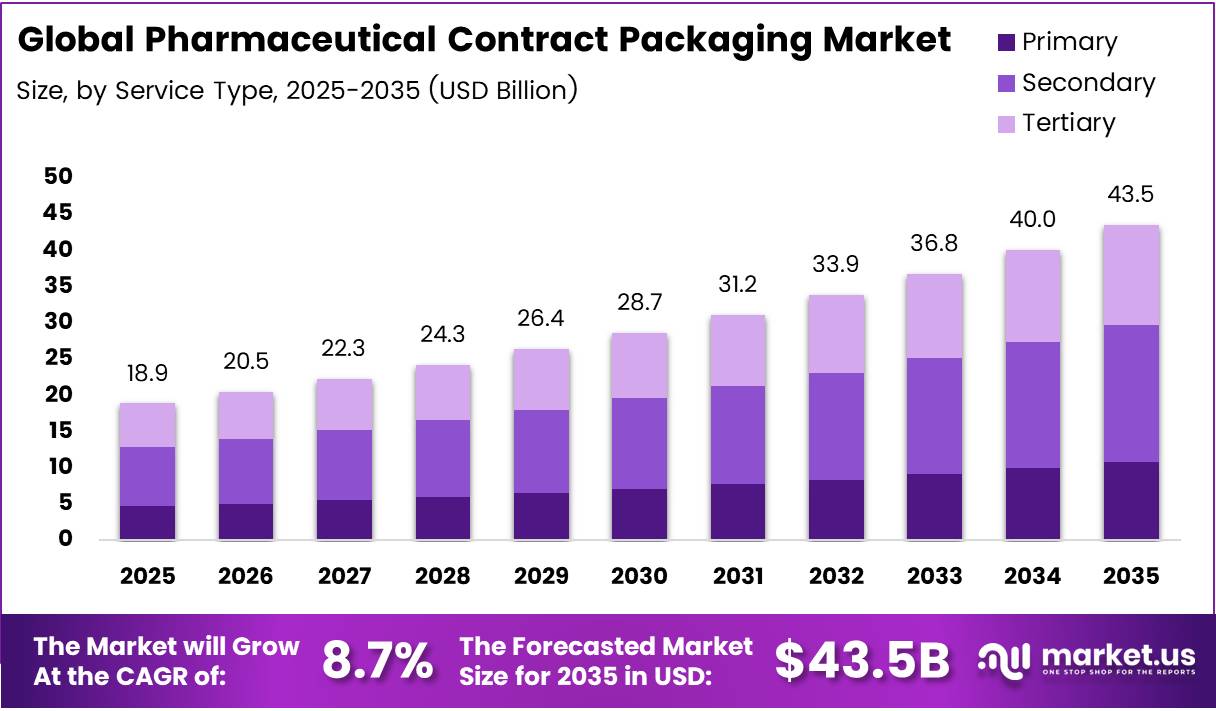
In 2025, the Global Pharmaceutical Contract Packaging Market was valued at US$18.9 billion, and between 2026 and 2035, this market is estimated to register a CAGR of 8.7%, reaching about US$43.5 billion by 2035.
Pharmaceutical contract packaging is the practice of outsourcing the final stage of drug production, the packaging and labeling, to a specialized third-party firm known as a Contract Packaging Organization (CPO). The pharmaceutical contract packaging market is shaped by operational efficiency, regulatory complexity, and product specialization.
Outsourcing is driven by cost reduction and flexibility, allowing manufacturers to shift fixed capital and labor to third-party providers while maintaining compliance with frameworks such as the Drug Supply Chain Security Act and the Falsified Medicines Directive. Secondary packaging is most widely used due to its regulatory adaptability, lower technical barriers, and logistical advantages, while solid dosage forms dominate because of their stability, scalability, and minimal aseptic requirements.
Large pharmaceutical companies are the primary users, benefiting from high-volume portfolios, multi-market customization, and stringent compliance demands, whereas generics, biosimilars, biotech start-ups, and CRO/CDMO partners rely less on contract packaging. Opportunities are increasing in specialized biologics and cold-chain products, and sustainable packaging initiatives are gaining traction. Geographic concentration is notable in North America, where regulatory rigor, high prescription volumes, and advanced infrastructure support extensive outsourcing.
Key Takeaways
- The global pharmaceutical contract packaging market was valued at US$18.9 billion in 2025.
- The global pharmaceutical contract packaging market is projected to grow at a CAGR of 8.7% and is estimated to reach US$43.5 billion by 2035.
- On the basis of service types, secondary packaging dominated the market, constituting 43.5% of the total market share.
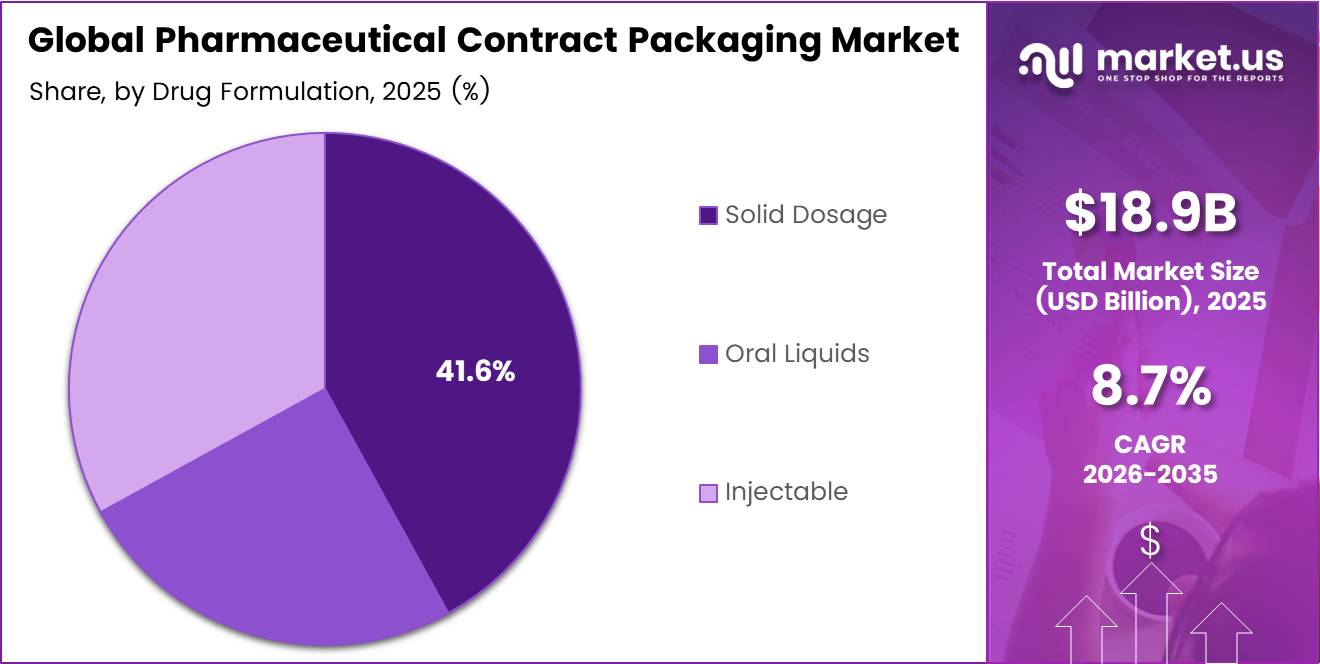
- Based on the drug formulation, solid dosage packaging led the market, comprising 41.6% of the total market.
- Among the end-uses of pharmaceutical contract packaging, the big pharma industry is the most considerable within the market, accounting for around 31.9% of the revenue.
- In 2025, North America was the most dominant region in the pharmaceutical contract packaging market, accounting for 37.6% of the total global consumption.
Service Type Analysis
Secondary Packaging dominates with 43.5% due to regulatory adaptability and logistical advantages without direct product-contact constraints.
In 2025, Secondary Packaging held a dominant market position in the By Service Type segment of the Global Pharmaceutical Contract Packaging Market, with a 43.5% share. Requirements such as serialization under the Drug Supply Chain Security Act and the Falsified Medicines Directive are typically implemented at the secondary level — cartons and labels — enabling contract packagers to add compliance features without altering primary packaging validated for drug stability. Secondary packaging supports late-stage customization, reducing inventory risk and enabling rapid market-specific adjustments.
Unlike primary packaging, secondary packaging does not require sterile environments or direct product compatibility validation, making outsourcing simpler and faster. Secondary packaging enables bundling, barcoding, and tamper-evidence, directly supporting distribution and retail requirements. This combination of regulatory function and operational flexibility positions secondary packaging as the default outsourcing entry point for pharmaceutical manufacturers seeking compliance without capital-intensive infrastructure investment.
Primary Packaging encompasses direct product-contact formats including bottles, plastic and glass containers, vials, ampoules, and blister packs. This segment requires stringent material compatibility validation and, in many cases, cleanroom or aseptic processing environments. Contract packagers serving the primary segment command premium pricing due to the technical barriers and regulatory qualification requirements associated with each format.
Bottles serve as the dominant primary packaging format for oral solid and liquid dosage forms, offering scalable high-speed filling across both plastic and glass substrates. Their compatibility with automated counting, filling, and capping lines makes them a preferred outsourcing format for large-volume generics and OTC products. Glass bottles remain preferred for sensitive formulations where chemical inertness and barrier performance are critical.
Plastic primary packaging offers lightweight, shatter-resistant containment for a broad range of solid and liquid pharmaceutical products. High-density polyethylene and polypropylene formats dominate cost-sensitive generics packaging, where per-unit material economics are a primary procurement driver. Advances in polymer barrier technology are expanding plastic’s applicability into previously glass-exclusive formulations.
Glass primary packaging retains preference for injectable, parenteral, and chemically sensitive formulations where inertness, transparency, and hermetic sealing are non-negotiable. Borosilicate glass vials and ampoules dominate sterile injectable packaging, particularly for biologics. Supply constraints on pharmaceutical-grade glass vials, highlighted during vaccine distribution programs, have reinforced the strategic importance of qualified glass primary packaging partners.
Vials and Ampoules serve as the primary containment format for injectable drug products, including vaccines, biologics, and high-potency small molecules. Their filling and sealing processes require aseptic manufacturing environments and extensive process validation, placing them among the highest-complexity primary packaging formats. Contract packagers with validated vial and ampoule filling capacity are positioned in the most technically differentiated tier of the outsourcing market.
Blister Packs dominate solid oral dosage primary packaging across branded and generic pharmaceutical portfolios. Unit-dose blistering supports patient compliance, tamper evidence, and product protection across diverse climatic conditions. High-speed blister lines with integrated vision inspection and serialization capability represent a core capital asset for contract packagers serving the solid dosage segment.
Cartons within secondary packaging serve as the primary vehicle for printed labeling, patient information leaflets, serialization data, and tamper-evident features. Their central role in regulatory compliance — particularly under DSCSA and FMD serialization mandates — makes carton packaging a high-activity outsourcing category. Contract packagers with automated carton erection, leaflet insertion, and online serialization printing maintain the highest throughput and compliance reliability in this format.
Labels and Inserts function as the regulatory and informational interface between the drug product and its end user, carrying mandatory content including dosage instructions, contraindications, and traceability identifiers. Multi-market pharmaceutical portfolios require frequent label variations by language, reimbursement code, and national regulatory requirement, making label management a recurring outsourcing driver. Digital printing and variable data integration capabilities are increasingly differentiating contract packagers in this sub-segment.
Tertiary Packaging covers outer shipping configurations including corrugated cases, palletization, and distribution-ready bundling that protect primary and secondary packages through logistics chains. While less regulated than inner packaging formats, tertiary configurations are increasingly subject to sustainability requirements and supply chain traceability integration. Contract packagers offering end-to-end packaging services that extend into tertiary formats provide clients with single-source accountability from unit packaging to distribution-ready shipment.
Drug Formulation Analysis
Solid Dosage dominates with 41.6% due to ambient stability, scalability, and minimal aseptic handling requirements.
In 2025, Solid Dosage held a dominant market position in the By Drug Formulation segment of the Global Pharmaceutical Contract Packaging Market, with a 41.6% share. Tablets and capsules are generally stable under ambient conditions and do not require aseptic handling, unlike injectables governed by sterile processing expectations. This reduces the need for specialized cleanroom infrastructure in outsourced settings, making solid dosage the most accessible and cost-efficient format for contract packaging engagement.
Solid dosages are less sensitive to contamination, oxidation, or microbial growth compared with oral liquids, which often require preservatives and tight closure systems to maintain integrity. High-speed blistering and bottling lines allow efficient large-batch outsourcing with minimal revalidation. Logistics are simpler — solids typically do not require cold-chain distribution, unlike many injectables, reducing compliance burden and making third-party packaging more operationally efficient.
Capsules represent a significant sub-segment within solid dosage packaging, offering formulation flexibility for both immediate and modified-release drug delivery applications. Their compatibility with powder, pellet, and liquid-fill formats makes capsules suitable for a broad range of active pharmaceutical ingredients. Contract packagers with multi-size capsule filling and sealing lines serve both innovator and generic drug manufacturers across global markets.
Tablets constitute the highest-volume solid dosage format globally, supported by well-established compression, coating, and blister packaging technology. Tablet packaging lines offer the highest throughput rates among all pharmaceutical dosage formats, making them the default outsourcing format for high-volume generics and branded oral products. Serialization-integrated tablet blistering represents the convergence of volume efficiency and regulatory compliance in contract packaging operations.
Oral Liquids require more complex primary containment than solid dosages, including preservative systems, child-resistant closures, and precise fill-volume control. Their sensitivity to microbial contamination and oxidation demands tighter process controls during contract filling and packaging. Oral liquid contract packaging serves pediatric, geriatric, and dysphagia patient populations where solid dosage forms are clinically inappropriate.
Injectable drug formulations represent the highest-complexity and highest-value segment within pharmaceutical contract packaging, encompassing large-volume and small-volume parenteral products. Aseptic filling, sterility assurance, and cold-chain packaging requirements create significant technical barriers that concentrate injectable outsourcing among a limited number of qualified contract packagers. The growth of biologics, biosimilars, and gene therapies is expanding the injectable contract packaging segment at a faster rate than any other drug formulation category.
Large-Volume Parenterals include intravenous fluids, irrigation solutions, and nutritional products packaged in flexible bags or rigid bottles requiring aseptic filling at high throughput. Their manufacturing and packaging demand dedicated, purpose-built facilities with validated filling lines and comprehensive environmental monitoring programs. Contract packagers operating large-volume parenteral lines serve hospital systems, compounding operations, and pharmaceutical manufacturers supplying critical care settings.
Small-Volume Parenterals encompass unit-dose injectables, prefilled syringes, cartridges, and vials containing potent or biologically derived drug substances. This sub-segment commands the highest per-unit packaging complexity and pricing within the injectable category. The increasing adoption of self-injection devices and prefilled syringe formats — driven by patient convenience and biologic drug delivery requirements — is expanding outsourcing demand for small-volume parenteral packaging specialists.
End-Use Analysis
Big Pharma dominates with 31.9% due to scale, regulatory burden, and multi-market portfolio complexity.
In 2025, Big Pharma held a dominant market position in the By End-Use segment of the Global Pharmaceutical Contract Packaging Market, with a 31.9% share. Large firms manage multi-market product portfolios with frequent label, language, and pack variations, making outsourced secondary packaging efficient for late-stage customization and global distribution. Compliance with serialization and traceability mandates under frameworks such as the Drug Supply Chain Security Act and the Falsified Medicines Directive requires advanced data systems and validated processes, which contract packagers provide at scale.
Large companies prioritize R&D and manufacturing while externalizing packaging to reduce internal complexity and fixed infrastructure. Established firms use multiple qualified packaging partners to ensure continuity across regions, an approach less common among smaller firms with limited product volumes or more integrated operating models. This multi-vendor qualification strategy reinforces sustained outsourcing volume from big pharma clients regardless of product pipeline cycles.
Generics and Biosimilar Companies engage contract packaging providers primarily to manage cost efficiency and rapid market entry across multiple geographies. Generic manufacturers operating on compressed margins benefit from variable-cost outsourcing models that avoid fixed packaging infrastructure investment. Biosimilar companies, navigating complex regulatory approval pathways, require packaging partners with validated biologic handling and serialization capabilities that internal operations may not cost-effectively support.
Emerging Biotech and Start-ups represent a structurally dependent end-use segment, as early-stage companies typically lack the capital and regulatory infrastructure to operate proprietary packaging facilities. Contract packagers serve as an essential service bridge for biotech companies moving from clinical to commercial-scale packaging without committing to fixed infrastructure. The growth of cell and gene therapy pipelines is creating demand for highly specialized, small-batch contract packaging that emerging biotech firms cannot efficiently internalize.
CRO and CDMO Partners engage pharmaceutical contract packaging providers as part of integrated drug development and manufacturing service chains. Clinical research organizations require clinical trial packaging — including blinding, randomization labeling, and temperature-controlled distribution — that specialized contract packagers deliver with validated compliance. CDMOs increasingly bundle contract packaging within end-to-end service offerings, either through in-house capability or qualified packaging subcontractors.
Others within the end-use segment include hospital pharmacies, veterinary pharmaceutical manufacturers, nutraceutical producers, and government health agencies procuring contract packaging for essential medicines programs. These end-users generate specialized, often lower-volume packaging demand that niche contract packagers serve with flexible line configurations. Government procurement programs for pandemic preparedness and essential medicine stockpiling represent an emerging demand source within this category.
Key Market Segments
By Service Type
- Primary
- Bottles
- Plastic
- Glass
- Vials & Ampoules
- Blister Packs
- Secondary
- Cartons
- Labels & Inserts
- Tertiary
By Drug Formulation
- Solid Dosage
- Capsules
- Tablets
- Oral Liquids
- Injectable
- Large-volume Parenterals
- Small-volume Parenterals
By End-Use
- Big Pharma
- Generics and Biosimilar Companies
- Emerging Biotech and Start-ups
- CRO and CDMO Partners
- Others
Drivers
Outsourcing to Cut Costs and Enhance Efficiency Drives the Pharmaceutical Contract Packaging Market
Pharmaceutical organizations increasingly utilize Contract Packaging Organizations (CPOs) to transition from capital-intensive internal operations to flexible, specialized models. This driver is anchored in two primary operational imperatives — mitigation of high compliance and fixed costs, and enhancement of operational efficiency and agility. Outsourcing allows firms to bypass massive capital investment in specialized machinery, such as that required for pre-filled syringes or cold-chain biologics.
Contract packaging eliminates the need for investment in dedicated packaging lines, validation, and skilled labor, reducing fixed capital intensity and freeing resources for R&D and commercialization. This shifts expenditure from fixed to variable cost structures, improving asset utilization and lowering per-unit packaging costs at scale. The financial logic is particularly compelling for manufacturers managing multi-SKU portfolios where internal line changeovers and revalidation cycles impose disproportionate overhead costs.
Furthermore, efficiency gains are multi-dimensional. Outsourcing improves speed-to-market, reduces lead times, and enhances supply-chain flexibility through scalable capacity and established supplier networks. Additionally, specialized providers handle high-complexity formats, such as cold-chain biologics packaging, which internal facilities may not efficiently support. This capability gap between what manufacturers can operate internally versus what specialized contract packagers provide continues to widen as drug formats grow more technically demanding.
Restraints
Dynamic and Complex Regulatory Frameworks Pose Challenges to the Conventional Pharmaceutical Contract Packaging Market
Dynamic and increasingly stringent regulatory frameworks impose material operational and compliance challenges on pharmaceutical contract packaging providers. Regulatory mandates such as the EU Falsified Medicines Directive (Directive 2011/62/EU) and the U.S. Drug Supply Chain Security Act (DSCSA) require unit-level serialization, verification, and reporting across the supply chain, significantly increasing process complexity. Compliance burden is significantly intensive across both major regulatory jurisdictions.
In the EU, almost all prescription medicines must carry unique identifiers and tamper-evident features since 2019, with each pack encoded using multiple data elements such as product code, serial number, batch, and expiry. This requires packaging lines to integrate high-resolution printing, vision inspection, and data management systems capable of handling large-scale serialization datasets. The capital and operational cost of maintaining these systems falls directly on contract packagers who serve multi-market pharmaceutical clients.
Similarly, operational inefficiencies arise from regulatory adaptation. Serialization introduces line slowdowns, additional quality checks, and increased per-pack processing steps, alongside mandatory data uploads to centralized repositories such as the European Medicines Verification System. While harmonized at a regional level, regulations vary in implementation details — including national reimbursement codes and reporting rules — requiring contract packagers to maintain multiple compliance configurations simultaneously. This multi-configuration burden raises operating costs and limits line throughput efficiency.
Growth Factors
Specialized Packaging for Biologics and Complex Drugs Creates Opportunities in the Pharmaceutical Contract Packaging Market
Specialized packaging for biologics and complex drugs constitutes a distinct opportunity for pharmaceutical contract packaging providers, driven by stringent stability requirements and measurable handling risks. Biologics, including monoclonal antibodies, vaccines, and gene therapies, require precise temperature control across defined ranges to maintain efficacy. Technical complexity is significantly high due to these specialized conditions.
Regulatory guidance mandates validated cold-chain systems capable of maintaining 2–8°C for at least 96 hours with integrated monitoring devices per shipment, increasing packaging sophistication and compliance burden. Several temperature-sensitive pharmaceutical shipments experience excursions, directly risking product degradation and regulatory non-compliance. This necessitates insulated systems, active cooling, and real-time data logging, all typically provided by specialized contract packagers with validated cold-chain infrastructure.
Moreover, product fragility reinforces the outsourcing case. Even short deviations can cause irreversible loss of potency in biologics, often without visible indicators, requiring validated packaging, monitoring, and handling protocols. Contract packagers who invest in cold-chain capability, biologics-compatible primary packaging, and real-time excursion monitoring position themselves in the highest-value, highest-barrier tier of the pharmaceutical outsourcing market — where switching costs and qualification requirements create durable client relationships.
Emerging Trends
Shift Towards Sustainable and Eco-Friendly Materials
The shift toward sustainable and eco-friendly materials is an increasingly codified trend in pharmaceutical contract packaging, driven by regulatory mandates, material innovation, and quantifiable environmental pressures. Public policy frameworks such as the EU Circular Economy Action Plan require 100% of packaging to be reusable or recyclable by 2030, directly influencing pharmaceutical packaging design and material selection. Compliance with this mandate is not optional — it will restructure material procurement across the contract packaging supply chain.
Pharmaceutical packaging contributes substantially to global medical plastic use, with estimates of approximately 15 million tons of plastic waste annually, prompting substitution toward recyclable and biodegradable alternatives. Material substitution is measurable and technology-driven. Conventional multi-material laminates are being replaced with mono-material structures and recyclable polymers such as HDPE and PET, enabling easier recovery and reuse while maintaining barrier properties required for drug stability and regulatory compliance.
Similarly, paper-based blister packs and fiber-based formats have demonstrated about 80% lower CO₂ footprints compared to traditional plastic formats, indicating significant decarbonization potential. Contract packagers who develop validated sustainable format capabilities ahead of regulatory deadlines gain a first-mover advantage in client retention and new business development. Pharmaceutical manufacturers increasingly specify sustainability credentials as part of contract packager qualification criteria, making eco-format capability a commercial differentiator rather than a voluntary commitment.
Geopolitical Impact Analysis
Geopolitical Tensions Have Led to Regionalized Production in the Pharma Industry
Geopolitical instability and trade restrictions have introduced systemic volatility into the pharmaceutical contract packaging market, primarily through supply chain fragmentation and input cost inflation. For certain Western markets, up to 80% of active pharmaceutical ingredients (APIs) and key primary packaging components such as specialized glass vials originate from the Asia Pacific region, creating high exposure to geopolitical risk and trade disruptions. The heightened geopolitical risks have prompted a shift from globalized to regionalized packaging hubs to ensure medicine security.
Additionally, 60–70% of global API production is concentrated in Asia, reinforcing single-region dependency. This concentration directly impacts contract packaging through irregular input availability and scheduling uncertainty. Under the U.S. Executive Order 14017, federal agencies have identified domestic packaging capacity as a critical vulnerability, leading to a 25% increase in domestic manufacturing grants for essential medicine components to mitigate foreign reliance.
Similarly, trade restrictions and tariffs increase packaging costs. Policy actions including tariffs have affected several countries and key industrial inputs — such as steel and aluminum — used in pharmaceutical equipment and packaging systems. Such measures elevate material and logistics costs for contract packagers. These combined pressures are accelerating investment in regional supplier diversification, nearshore packaging hubs, and higher safety stock requirements as structural responses to geopolitical supply chain exposure.
Regional Analysis
North America Dominates the Global Pharmaceutical Contract Packaging Market with a Market Share of 37.6%
In 2025, North America dominated the global pharmaceutical contract packaging market, holding about 37.6% of the total global consumption, supported by high pharmaceutical throughput, stringent regulations, and technological intensity. The U.S. healthcare distribution system handles over 6.5 billion prescription units annually, necessitating extensive primary and secondary packaging operations, including labeling, serialization, and tamper-evident systems. This volume directly sustains demand for outsourced packaging capacity and specialized service providers.
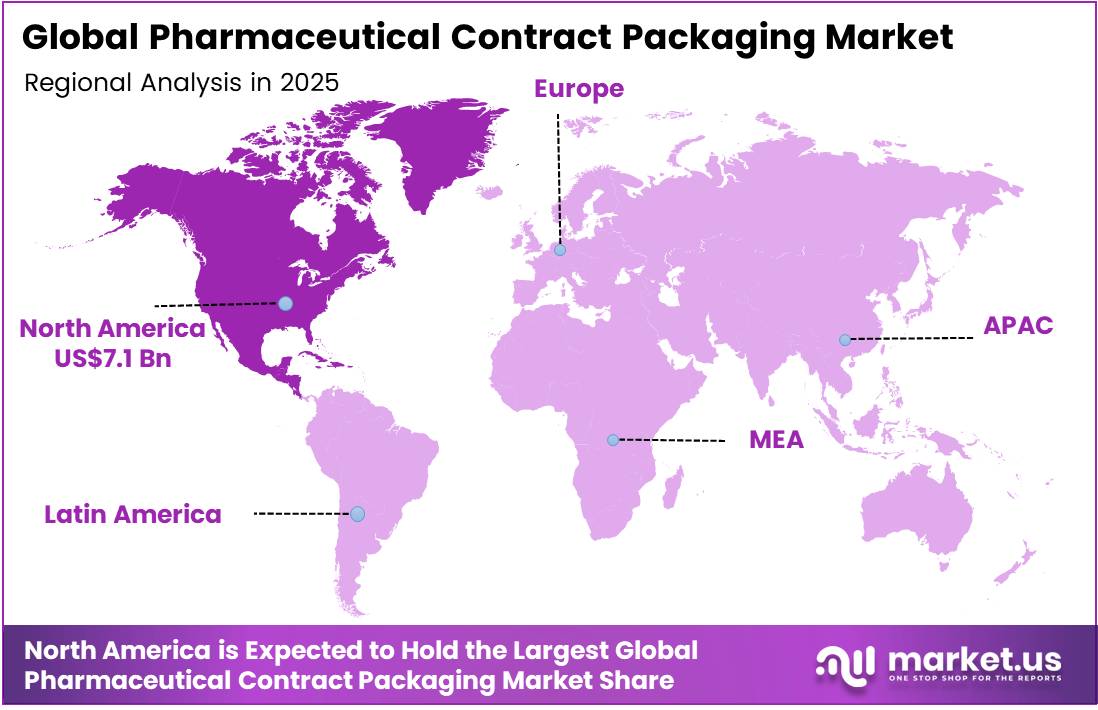
The U.S. Drug Supply Chain Security Act (DSCSA) mandates unit-level traceability and serialization for nearly all prescription drugs, requiring each package to carry standardized identifiers such as product, serial, batch, and expiry. This creates sustained demand for contract packagers with compliant infrastructure and data capabilities. The U.S. accounts for the majority of global biopharmaceutical R&D activity, increasing the share of complex drug formats requiring advanced packaging — including sterile, temperature-controlled, and combination products — that specialized contract packagers are best positioned to deliver.
Key Regions and Countries
North America
- The US
- Canada
Europe
- Germany
- France
- The UK
- Spain
- Italy
- Russia & CIS
- Rest of Europe
Asia Pacific
- China
- Japan
- South Korea
- India
- ASEAN
- Rest of APAC
Latin America
- Brazil
- Mexico
- Rest of Latin America
Middle East & Africa
- GCC
- South Africa
- Rest of MEA
Key Company Insights
Manufacturers of pharmaceutical contract packaging focus on capability expansion, regulatory compliance, and technology integration. Firms invest in high-speed, flexible packaging lines, automation, and digital serialization systems to comply with requirements under frameworks such as the Drug Supply Chain Security Act and the Falsified Medicines Directive. These investments enable multi-format packaging, reduce errors, and support track-and-trace obligations.
Companies expand capabilities in cold-chain packaging, sterile handling, and complex formats. Establishing regionally distributed facilities improves resilience and reduces lead times amid trade disruptions. Continuous upgrades to GMP-compliant processes, audit readiness, and data integrity systems enhance reliability and client retention.
Recent Developments
- June 2024 — PCI Pharma Services announced it would build an 82,000 sq ft facility at CityNorth Business Campus, Stamullen, Ireland, under a 25-year lease. Featuring sustainable design, it is to expand production capacity.
- July 2024 — Catalent completed a US$25 million expansion of its Schorndorf, Germany, clinical supply facility, adding 32,000 square feet, temperature-controlled storage, and automated bottle filling to enhance packaging, storage, and distribution services across Europe.
Key Players
- PCI Pharma Services
- Catalent Inc.
- Sharp Packaging Services
- Almac Group
- Wasdell Group
- Ropack Inc.
- Reed-Lane Inc.
- Jones Healthcare Group
- Recipharm AB
- Tjoapack Netherlands B.V.
- AmeriPac (Veritiv Corporation)
- Silgan Unicep
- Nelipak
- Aphena Pharma Solutions
- Central Pharma Contract Packing
- Quantrelle Packaging Solutions
- Variopack GmbH
- Sepha Ltd.
- Assemblies Unlimited
- DaklaPack Group
- Tripak Pharmaceuticals
- MPH Co-Packing
- Southwest Packaging
- MJS Packaging
- Jam Jams Group
- Asiapack (Elanders Group)
- Other Key Players
Report Scope
Report Features Description Market Value (2025) US$18.9 Bn Forecast Revenue (2035) US$43.5 Bn CAGR (2026-2035) 8.7% Base Year for Estimation 2025 Historic Period 2020-2024 Forecast Period 2026-2035 Report Coverage Revenue Forecast, Market Dynamics, Competitive Landscape, Recent Developments Segments Covered By Service Type (Primary, Secondary, and Tertiary), By Drug Formulation (Solid Dosage, Oral Liquids, and Injectable), By End-Use (Big Pharma, Generics and Biosimilar Companies, Emerging Biotech and Start-ups, CRO and CDMO Partners, and Others) Regional Analysis North America – The US & Canada; Europe – Germany, France, The UK, Spain, Italy, Russia & CIS, Rest of Europe; APAC – China, Japan, South Korea, India, ASEAN & Rest of APAC; Latin America – Brazil, Mexico & Rest of Latin America; Middle East & Africa – GCC, South Africa, & Rest of MEA Competitive Landscape PCI Pharma Services, Catalent Inc., Sharp Packaging Services, Almac Group, Wasdell Group, Ropack Inc., Reed-Lane Inc., Jones Healthcare Group, Recipharm AB, Tjoapack Netherlands B.V., AmeriPac (Veritiv Corporation), Silgan Unicep, Nelipak, Aphena Pharma Solutions, Central Pharma Contract Packing, Quantrelle Packaging Solutions, Variopack GmbH, Sepha Ltd., Assemblies Unlimited, DaklaPack Group, Tripak Pharmaceuticals, MPH Co-Packing, Southwest Packaging, MJS Packaging, Jam Jams Group, Asiapack (Elanders Group), and Other Players. Customization Scope Customization for segments, region/country-level will be provided. Moreover, additional customization can be done based on the requirements. Purchase Options We have three licenses to opt for: Single User License, Multi-User License (Up to 5 Users), Corporate Use License (Unlimited Users and Printable PDF)  Pharmaceutical Contract Packaging MarketPublished date: May 2026add_shopping_cartBuy Now get_appDownload Sample
Pharmaceutical Contract Packaging MarketPublished date: May 2026add_shopping_cartBuy Now get_appDownload Sample -
-
- PCI Pharma Services
- Catalent Inc.
- Sharp Packaging Services
- Almac Group
- Wasdell Group
- Ropack Inc.
- Reed-Lane Inc.
- Jones Healthcare Group
- Recipharm AB
- Tjoapack Netherlands B.V.
- AmeriPac (Veritiv Corporation)
- Silgan Unicep
- Nelipak
- Aphena Pharma Solutions
- Central Pharma Contract Packing
- Quantrelle Packaging Solutions
- Variopack GmbH
- Sepha Ltd.
- Assemblies Unlimited
- DaklaPack Group
- Tripak Pharmaceuticals
- MPH Co-Packing
- Southwest Packaging
- MJS Packaging
- Jam Jams Group
- Asiapack (Elanders Group)
- Other Key Players










