Global Medical Specimen Tracking Systems Market By Component (Hardware, Software, Services) By Technology (Barcode Scanning, Radio Frequency Identification (RFID), Near Field Communication (NFC), Others) By Application (Laboratory Specimens, Anatomical Pathology Specimens, Surgical and Biopsy Specimens, Blood and Blood Products, Reproductive Tissue and Gametes, Others) By End User (Hospitals, Diagnostic Laboratories, Blood Banks, Biobanks and Research Institutes, Pathology Laboratories, Others) By System Type (Standalone Systems, Integrated Systems), Region and Companies – Industry Segment Outlook, Market Assessment, Competition Scenario, Trends and Forecast 2026-2035
- Published date: April 2026
- Report ID: 185386
- Number of Pages: 352
- Format:
-
keyboard_arrow_up
Quick Navigation
Report Overview
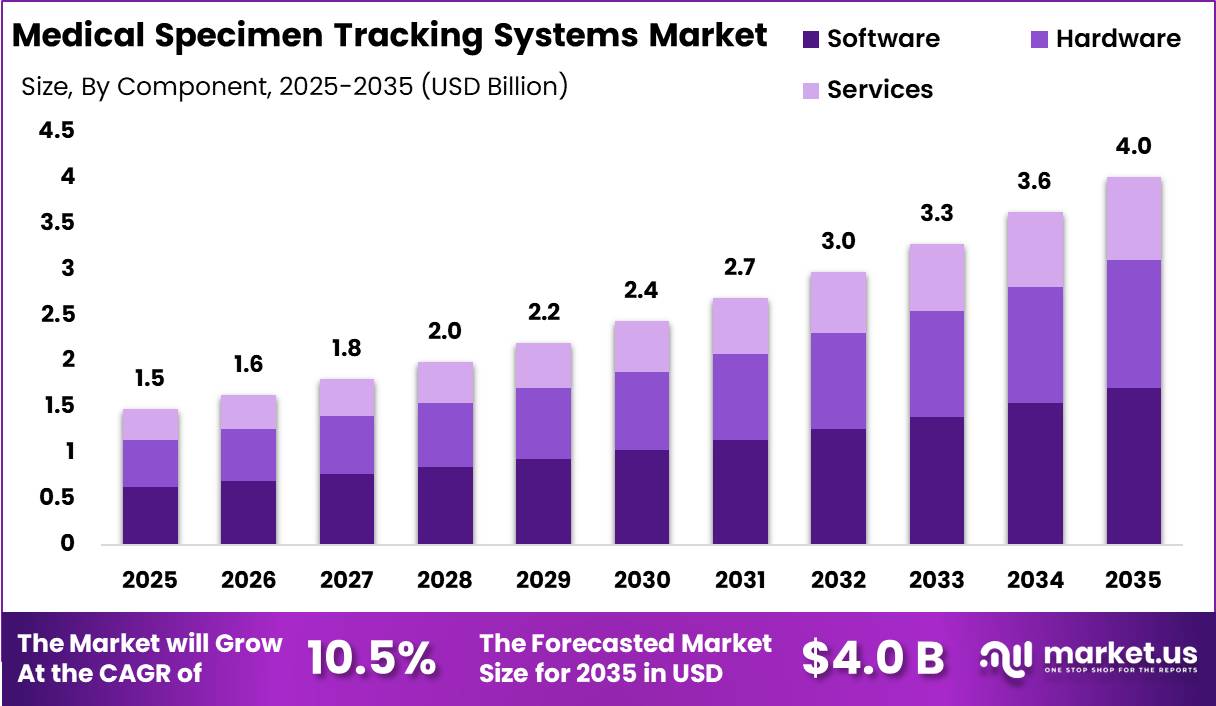
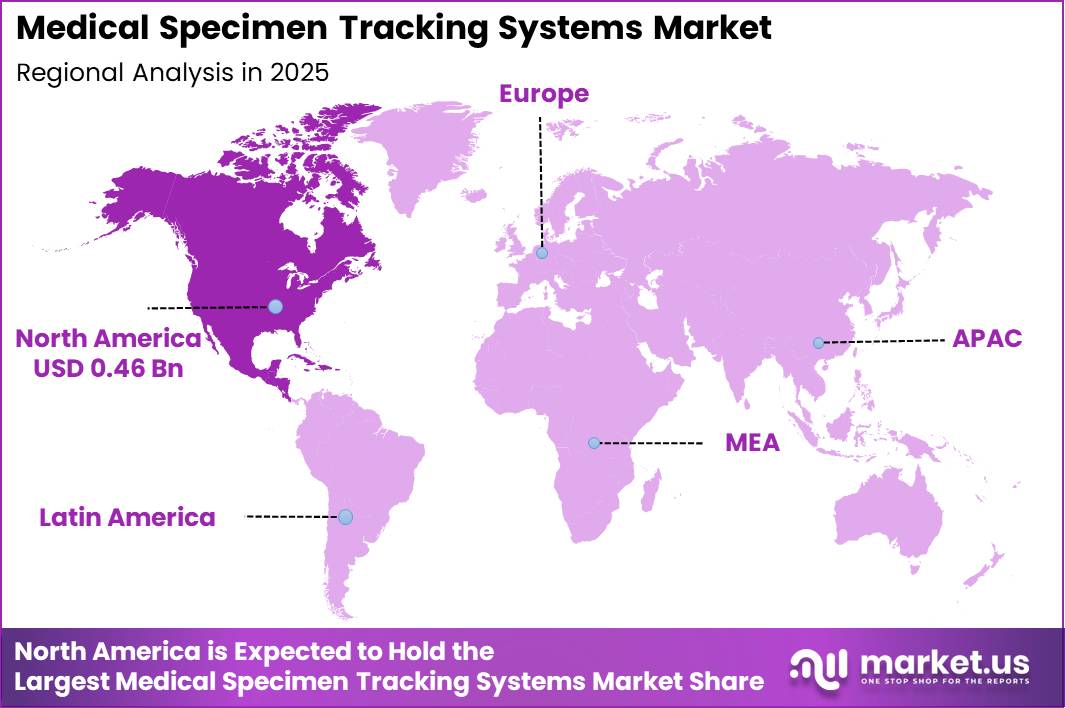
Global Medical Specimen Tracking Systems Market size is expected to be worth around US$ 4.0 Billion by 2035 from US$ 1.5 Billion in 2025, growing at a CAGR of 10.5% during the forecast period from 2026 to 2035. In 2025, North America led the market, achieving over 40.46% share with a revenue of US$ 0.46 Billion.
The medical specimen tracking system represents a critical component within modern healthcare infrastructure, designed to ensure accurate identification, secure handling, and traceability of biological samples throughout their lifecycle. These systems support the end-to-end process, including specimen collection, labeling, transportation, laboratory analysis, and storage, thereby strengthening patient safety and diagnostic reliability.
From a healthcare operations perspective, specimen tracking systems are increasingly being adopted to address persistent challenges related to identification errors. According to the Centers for Disease Control and Prevention, specimen labeling errors remain a significant concern, with studies indicating that identification errors can occur in approximately 0.1% to 5% of specimens in clinical settings.
Furthermore, a national laboratory report highlights that misidentification events were observed in approximately 1 out of every 1,986 specimens, emphasizing the need for robust tracking mechanisms. Such errors can lead to misdiagnosis, delayed treatment, and adverse clinical outcomes.
The integration of advanced technologies such as barcode systems has demonstrated measurable improvements in accuracy. Evidence-based evaluations by the Centers for Disease Control and Prevention show that barcode implementation significantly reduces specimen identification errors, with strong statistical effectiveness observed across multiple healthcare settings .
In practical applications, the introduction of electronic ordering systems combined with barcode labeling has reduced specimen processing error rates from 0.42% to 0.11%, representing a 74% relative reduction.
In addition, digital transformation trends such as cloud-based platforms and automated laboratory workflows are enabling real-time tracking, improved data integration, and enhanced chain-of-custody compliance. These systems are widely utilized across hospitals, diagnostic laboratories, and research institutions, where operational efficiency and regulatory compliance are essential.
Overall, the adoption of medical specimen tracking systems is being driven by the need to minimize human error, improve diagnostic accuracy, and comply with stringent healthcare standards. Continued technological advancements and standardization initiatives are expected to further strengthen system reliability and support improved patient outcomes.
Key Takeaways
- Market Size: Global Medical Specimen Tracking Systems Market size is expected to be worth around US$ 4.0 Billion by 2035 from US$ 1.5 Billion in 2025.
- Market Share: The market growing at a CAGR of 10.5% during the forecast period from 2026 to 2035.
- Component Analysis: The software segment accounts for the largest share, estimated at 42.5% in 2025.
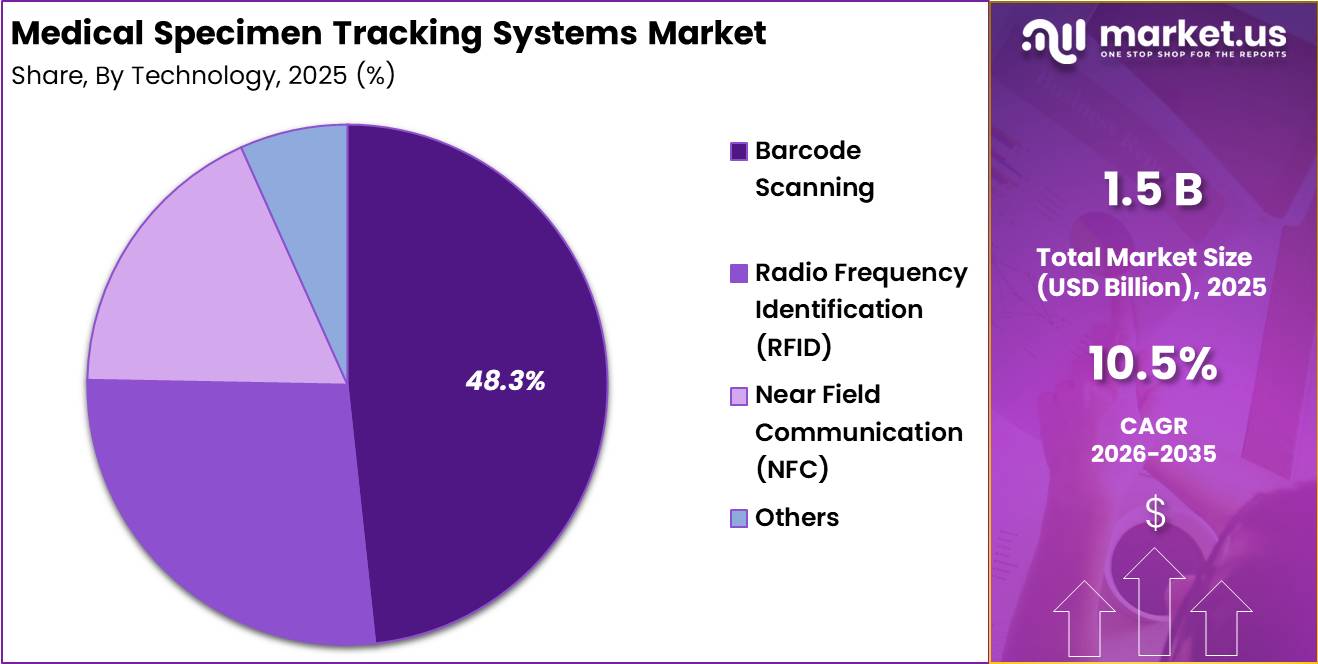
- Technology Analysis: Barcode scanning technology is projected to dominate the market with a 48.3%.
- Application Analysis: Laboratory specimens are expected to hold the largest share, accounting for 34.7% in 2025.
- End user Analysis: Hospitals are anticipated to dominate the market, holding a 41.9%.
- System Type Analysis: Standalone systems are projected to dominate the market with a 57.6% share in 2025.
- Regional Analysis: In 2025, North America led the market, achieving over 40.46% share with a revenue of US$ 0.46 Billion.
Component Analysis
The medical specimen tracking systems market is segmented into software, hardware, and services. It has been observed that the software segment accounts for the largest share, estimated at 42.5% in 2025. This dominance is attributed to the increasing adoption of laboratory information management systems (LIMS), data integration platforms, and real-time tracking applications that enhance workflow efficiency and reduce identification errors.
Software solutions are being widely implemented to ensure compliance with regulatory standards and improve traceability across healthcare settings. The hardware segment, comprising barcode printers, scanners, RFID readers, and labeling systems, represents a significant share, driven by the need for accurate specimen identification and automation in laboratories.
Meanwhile, the services segment, including system integration, maintenance, and training, is witnessing steady growth due to the rising complexity of healthcare IT infrastructure.
The demand for managed services is also increasing as healthcare providers seek cost optimization and operational efficiency. Overall, the component landscape is being shaped by the growing emphasis on digitization and error reduction in specimen management processes.
Technology Analysis
Based on technology, the market is categorized into barcode scanning, radio frequency identification (RFID), near field communication (NFC), and others. Barcode scanning technology is projected to dominate the market with a 48.3% share in 2025. This leadership position is supported by its cost-effectiveness, ease of implementation, and widespread adoption across laboratories and hospitals.
Barcode systems are considered reliable for minimizing labeling errors and improving specimen tracking accuracy. RFID technology is gaining traction due to its ability to enable real-time tracking without line-of-sight requirements, thereby enhancing operational efficiency in high-volume environments.
NFC technology, although at a nascent stage, is being explored for secure and contactless data exchange in specimen handling. The “others” segment includes emerging technologies such as Bluetooth-based tracking and IoT-enabled systems, which are expected to gain momentum over the forecast period.
Overall, technological advancements are contributing to improved patient safety, streamlined workflows, and enhanced data visibility across healthcare facilities.
Application Analysis
The application segment includes laboratory specimens, anatomical pathology specimens, surgical and biopsy specimens, blood and blood products, reproductive tissue and gametes, and others. Laboratory specimens are expected to hold the largest share, accounting for 34.7% in 2025. This dominance is driven by the high volume of routine diagnostic testing and the need for accurate sample identification and tracking.
Anatomical pathology specimens represent a significant segment due to the complexity and critical nature of tissue-based diagnostics. Surgical and biopsy specimens also contribute substantially, as precise tracking is essential to avoid diagnostic errors and ensure patient safety. Blood and blood products tracking is gaining importance with the increasing demand for transfusion safety and regulatory compliance.
Additionally, reproductive tissue and gametes tracking is witnessing growth due to advancements in fertility treatments and stringent handling requirements. The “others” category includes clinical research samples and forensic specimens. The application landscape reflects a strong focus on accuracy, compliance, and patient-centric care delivery.
End user Analysis
The end user segment comprises hospitals, diagnostic laboratories, blood banks, biobanks and research institutes, pathology laboratories, and others. Hospitals are anticipated to dominate the market, holding a 41.9% share in 2025. This can be attributed to the high patient inflow, increasing diagnostic procedures, and the need for integrated specimen tracking systems to reduce medical errors.
Diagnostic laboratories represent a key segment due to their reliance on high-throughput sample processing and stringent quality standards. Blood banks are increasingly adopting tracking systems to ensure the safety and traceability of blood products. Biobanks and research institutes are also significant contributors, driven by the growing focus on personalized medicine and long-term sample storage.
Pathology laboratories require robust tracking solutions to manage complex workflows and maintain accuracy in diagnostics. The “others” segment includes ambulatory care centers and specialty clinics. Overall, the end user landscape is being influenced by the rising demand for efficiency, accuracy, and regulatory compliance in healthcare operations.
System Type Analysis
The market is segmented by system type into standalone systems and integrated systems. Standalone systems are projected to dominate the market with a 57.6% share in 2025. This dominance is primarily due to their ease of deployment, lower initial investment, and suitability for small to mid-sized healthcare facilities. Standalone solutions are often preferred for their simplicity and ability to function independently without complex IT infrastructure.
However, integrated systems are gaining traction, particularly in large hospitals and healthcare networks, as they enable seamless data exchange across multiple departments and systems such as electronic health records (EHR) and laboratory information systems.
Integrated systems offer enhanced visibility, improved workflow coordination, and better decision-making capabilities. Despite higher implementation costs, their long-term benefits are encouraging adoption. The system type landscape is evolving with the increasing need for interoperability, scalability, and real-time data access in healthcare environments.
Key Market Segments
By Component
- Hardware
- Software
- Services
By Technology
- Barcode Scanning
- Radio Frequency Identification (RFID)
- Near Field Communication (NFC)
- Others
By Application
- Laboratory Specimens
- Anatomical Pathology Specimens
- Surgical and Biopsy Specimens
- Blood and Blood Products
- Reproductive Tissue and Gametes
- Others
By End User
- Hospitals
- Diagnostic Laboratories
- Blood Banks
- Biobanks and Research Institutes
- Pathology Laboratories
- Others
By System Type
- Standalone Systems
- Integrated Systems
Driving Factors
The growth of the medical specimen tracking systems market is primarily driven by the increasing need to reduce diagnostic errors and enhance patient safety across healthcare systems. According to clinical evidence, approximately 0.5%–1.0% of specimen handling processes involve labeling or identification errors, which can directly impact diagnostic accuracy and treatment outcomes.
Additionally, studies indicate that around 1 in 1,000 cases are affected by mislabeling, while misdirected reports occur in nearly 2 per 1,000 medical records, highlighting systemic inefficiencies in specimen handling. Healthcare systems are under regulatory pressure to ensure traceability and compliance, especially in high-volume laboratory environments where billions of samples are processed annually.
Specimen tracking systems, using barcode and RFID technologies, enable real-time monitoring and chain-of-custody control. These systems significantly reduce turnaround time and error rates, improving clinical decision-making, which is estimated to depend on laboratory diagnostics in nearly 70% of cases, reinforcing their critical importance.
Trending Factors
A key trend shaping the market is the integration of advanced digital technologies such as cloud computing, automation, and RFID-based real-time tracking. Healthcare systems are increasingly adopting end-to-end digital workflows to enhance laboratory efficiency and data transparency. Barcode technology currently dominates due to affordability, while RFID adoption is rising rapidly due to its ability to track multiple specimens simultaneously in real time.
In addition, automation in pathology laboratories is expanding, driven by rising diagnostic volumes and workforce constraints. Globally, laboratories process extremely high testing volumes, with estimates suggesting billions of specimens annually, necessitating scalable and automated solutions.
Another observable trend is the integration of specimen tracking with laboratory information systems (LIS) and electronic health records (EHR), enabling seamless data exchange and compliance monitoring.
Cloud-based platforms further allow remote access, audit trails, and predictive analytics, supporting quality control and operational optimization. These technological advancements are contributing to improved turnaround times, reduced manual intervention, and enhanced traceability across healthcare networks.
Restraint
Despite strong adoption potential, high implementation and integration costs remain a significant restraint for the market. Advanced specimen tracking systems require investment in hardware (RFID tags, scanners), software platforms, and workforce training, which can create financial barriers, particularly for small and mid-sized healthcare facilities.
Initial deployment costs are further increased by the need to integrate these systems with existing hospital infrastructure such as laboratory information management systems (LIMS) and electronic health records. According to industry observations, such integration complexity and cost burdens limit adoption in resource-constrained settings.
Additionally, ongoing maintenance, software upgrades, and compliance requirements contribute to the total cost of ownership. In developing healthcare systems, budget constraints and uneven digital infrastructure slow the transition from manual to automated tracking.
This financial barrier is particularly critical in regions with lower healthcare expenditure per capita, where priority is often given to basic diagnostic capacity rather than advanced tracking technologies, thereby restricting market penetration.
Opportunity
Significant growth opportunities are emerging due to the rising global demand for diagnostic testing and expanding healthcare infrastructure, particularly in emerging economies. The increasing prevalence of chronic diseases such as cancer and diabetes has led to a surge in diagnostic procedures, creating demand for efficient specimen management systems.
For instance, high diagnostic dependency is evident as laboratory data influences approximately 70% of medical decisions, emphasizing the need for reliable tracking solutions. Furthermore, healthcare systems in Asia-Pacific and other developing regions are investing in digital health infrastructure, with improved laboratory capacity and automation adoption.
The region is expected to witness strong growth due to increasing patient volumes and healthcare modernization initiatives. Opportunities also exist in the adoption of AI-driven analytics, blockchain for data security, and mobile-enabled tracking systems, which enhance transparency and reduce specimen loss.
These innovations can significantly improve operational efficiency, reduce costs associated with errors, and support large-scale diagnostic networks, positioning the market for sustained expansion.
Regional Analysis
In 2025, North America accounted for a leading share of over 40.46% in the medical specimen tracking systems market, with revenue reaching approximately US$ 0.46 billion. This dominance has been primarily supported by the presence of well-established healthcare infrastructure and advanced diagnostic networks across the region. A significant rise in chronic disease prevalence has increased the demand for accurate and efficient diagnostic procedures, thereby accelerating the adoption of specimen tracking solutions.
Early adoption of healthcare technologies has played a critical role in strengthening regional market performance. Continuous investments in research and development have enabled the integration of advanced technologies, including automation systems, blockchain-based data management, and RFID-enabled tracking solutions. These innovations have significantly enhanced operational efficiency, minimized errors, and improved traceability across laboratory workflows.
Furthermore, strategic collaborations between healthcare providers, laboratories, and technology companies have contributed to market expansion. Such partnerships have facilitated technology deployment at scale, improved system interoperability, and supported the development of more reliable and secure specimen tracking frameworks across North America.
Key Regions and Countries
North America
- The US
- Canada
Europe
- Germany
- France
- The U.K.
- Italy
- Spain
- Russia & CIS
- Rest of Europe
Asia Pacific
- China
- India
- Japan
- South Korea
- ASEAN
- Australia & New Zealand
- Rest of Asia Pacific
Middle East & Africa
- GCC
- South Africa
- Rest of Middle East & Africa
Latin America
- Brazil
- Mexico
- Rest of Latin America
Key Players Analysis
The competitive landscape of the medical specimen tracking systems market is characterized by the presence of several specialized technology providers and logistics-focused companies, contributing to innovation and operational efficiency.
Key players such as RMS Omega and Peak Technologies (Vision ID) focus on advanced identification technologies, including barcode and RFID systems, enabling accurate specimen labeling and tracking across healthcare facilities. GAO Group strengthens the market through IoT-enabled tracking systems, enhancing real-time monitoring capabilities.
Meanwhile, TrakCel plays a critical role in providing digital orchestration platforms, particularly for complex therapy logistics. Logistics-driven companies such as Kuehne + Nagel (Quick International Courier) and Cryoport Systems, LLC contribute through specialized cold-chain and time-sensitive transportation services for biological specimens.
Overall, market participants are increasingly emphasizing technological innovation, strategic partnerships, and integrated solutions to enhance traceability, reduce errors, and improve patient safety outcomes across healthcare and research environments.
Market Key Players
- RMS Omega
- Peak Technologies (Vision ID)
- GAO Group
- TrakCel
- Kuehne + Nagel (Quick International Courier)
- Cryoport Systems, LLC
- LabConnect
- Taylor Data Systems
- BioIT Solutions
- BROOKS AUTOMATION
- Others
Recent Developments
- Cryoport Systems LLC (January 2025): The company strengthened its positioning in temperature-controlled specimen logistics through expanded global supply chain capabilities, focusing on advanced cryogenic tracking and monitoring solutions for cell and gene therapy shipments. This development aligns with increased demand for precision tracking in biopharma logistics.
- TrakCel (March 2025): A new enhancement to its digital tracking platform was introduced, improving real-time visibility and chain-of-identity tracking for personalized medicine workflows. The upgrade reflects industry movement toward integrated digital ecosystems supporting clinical trial logistics.
- Brooks Automation (June 2025): Expansion of automated biostorage and specimen management solutions was announced, with increased focus on scalable sample tracking systems for large biobanks. The initiative supports growing global demand for high-throughput sample storage and traceability.
- Peak Technologies (August 2025): The company advanced its barcode and RFID-based specimen tracking offerings by integrating mobile-enabled tracking capabilities, enabling healthcare providers to enhance workflow efficiency and reduce specimen identification errors.
Report Scope
Report Features Description Market Value (2025) US$ 1.5 Billion Forecast Revenue (2035) US$ 4.0 Billion CAGR (2026-2035) 10.5% Base Year for Estimation 2025 Historic Period 2020-2024 Forecast Period 2026-2035 Report Coverage Revenue Forecast, Market Dynamics, Competitive Landscape, Recent Developments Segments Covered By Component (Hardware, Software, Services) By Technology (Barcode Scanning, Radio Frequency Identification (RFID), Near Field Communication (NFC), Others) By Application (Laboratory Specimens, Anatomical Pathology Specimens, Surgical and Biopsy Specimens, Blood and Blood Products, Reproductive Tissue and Gametes, Others) By End User (Hospitals, Diagnostic Laboratories, Blood Banks, Biobanks and Research Institutes, Pathology Laboratories, Others) By System Type (Standalone Systems, Integrated Systems) Regional Analysis North America – The US, Canada; Europe – Germany, France, U.K., Italy, Spain, Russia & CIS, Rest of Europe; Asia Pacific – China, India, Japan, South Korea, ASEAN, Australia & New Zealand, Rest of Asia Pacific; Middle East & Africa – GCC, South Africa, Rest of Middle East & Africa; Latin America – Brazil, Mexico, Rest of Latin America Competitive Landscape RMS Omega, Peak Technologies (Vision ID), GAO Group, TrakCel, Kuehne + Nagel (Quick International Courier), Cryoport Systems, LLC, LabConnect, Taylor Data Systems, BioIT Solutions, BROOKS AUTOMATION, Others Customization Scope Customization for segments, region/country-level will be provided. Moreover, additional customization can be done based on the requirements. Purchase Options We have three licenses to opt for: Single User License, Multi-User License (Up to 5 Users), Corporate Use License (Unlimited User and Printable PDF)  Medical Specimen Tracking Systems MarketPublished date: April 2026add_shopping_cartBuy Now get_appDownload Sample
Medical Specimen Tracking Systems MarketPublished date: April 2026add_shopping_cartBuy Now get_appDownload Sample -
-
- RMS Omega
- Peak Technologies (Vision ID)
- GAO Group
- TrakCel
- Kuehne + Nagel (Quick International Courier)
- Cryoport Systems, LLC
- LabConnect
- Taylor Data Systems
- BioIT Solutions
- BROOKS AUTOMATION
- Others










