Global Leadless Pacemakers Market By Product Type (Single-chamber Leadless Pacemakers, Dual-chamber Leadless Pacemakers) By Application (Bradycardia, Atrial Fibrillation, Heart Block, Others) By Patient Type (Pediatric, Adult, Geriatric) By End User (Hospitals, Ambulatory Surgical Centers, Others), Region and Companies – Industry Segment Outlook, Market Assessment, Competition Scenario, Trends and Forecast 2026-2035
- Published date: May 2026
- Report ID: 185711
- Number of Pages: 224
- Format:
-
keyboard_arrow_up
Quick Navigation
Report Overview
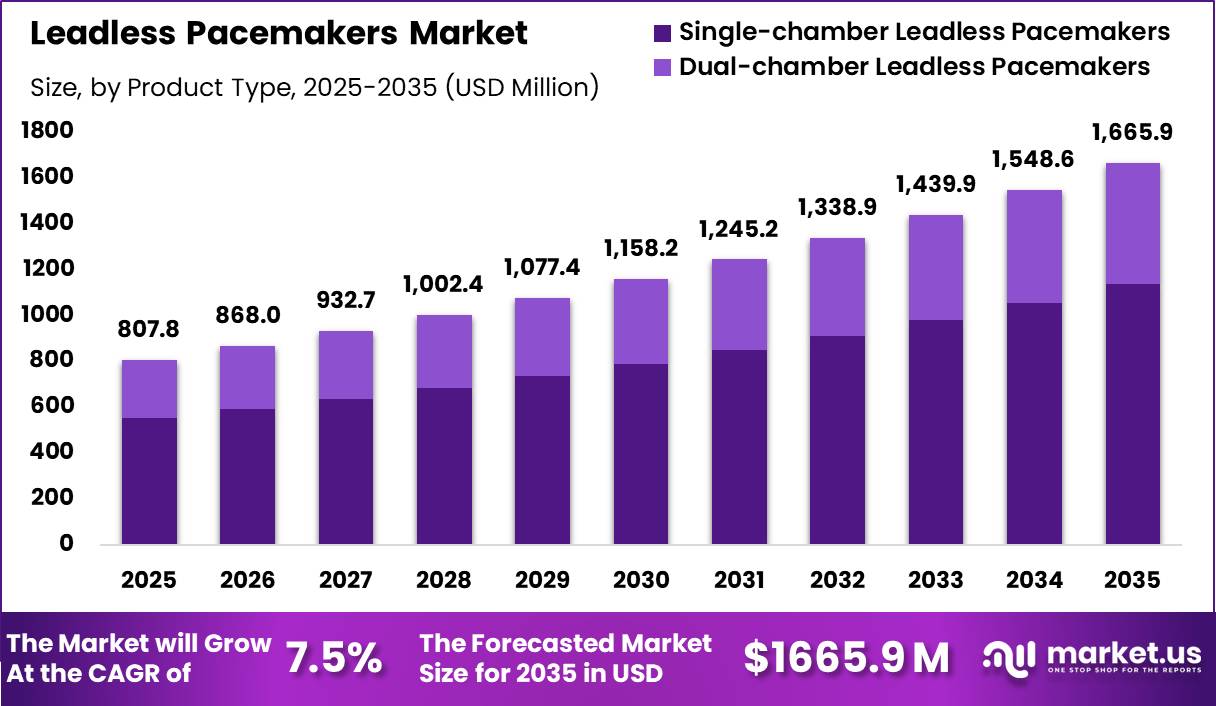
Global Leadless Pacemakers Market size is expected to be worth around US$ 1665.9 Million by 2035 from US$ 807.8 Million in 2025, growing at a CAGR of 7.5% during the forecast period from 2026 to 2035. In 2025, North America led the market, achieving over 43.3% share with a revenue of US$ 178.3 Million.
The global leadless pacemakers market is being shaped by the rising clinical burden of cardiovascular diseases (CVDs) and the increasing need for safer and minimally invasive cardiac rhythm management solutions.
According to data from the World Health Organization, cardiovascular diseases remain the leading cause of mortality worldwide, accounting for nearly 17.9 million deaths annually. This growing disease burden, combined with aging populations and higher incidence of arrhythmias such as atrial fibrillation and bradycardia, has significantly increased the demand for implantable pacing technologies.
Leadless pacemakers have emerged as a technologically advanced alternative to conventional transvenous systems. These devices are directly implanted into the heart via catheter-based procedures, eliminating the need for leads and subcutaneous pockets.
Clinical evidence from peer-reviewed medical literature indicates that traditional pacemaker systems are often associated with complications such as lead dislodgement, infection, venous obstruction, and pneumothorax.
In contrast, leadless systems demonstrate improved safety outcomes, with studies highlighting comparable or lower short-term complication rates and reduced device-related issues. Additionally, real-world clinical studies have reported major complication rates as low as approximately 3–4% in leadless pacemaker recipients, reinforcing their safety profile.
Technological advancements are further accelerating market expansion. The development of dual-chamber and atrioventricular (AV) synchronous pacing capabilities has significantly broadened the clinical applicability of leadless systems.
Devices such as modular and dual-chamber leadless pacemakers enable treatment of more complex rhythm disorders, including sinus node dysfunction and AV block, which were previously managed primarily with traditional systems.
Furthermore, improvements in battery longevity, device miniaturization, and remote monitoring capabilities are enhancing long-term patient outcomes and device efficiency. From a healthcare system perspective, the adoption of leadless pacemakers is supported by the increasing focus on reducing hospital-acquired infections and minimizing repeat interventions.
The elimination of leads, which are a major source of mechanical failure and infection, aligns with clinical guidelines emphasizing patient safety and cost efficiency. Moreover, the expansion of electrophysiology infrastructure, particularly in emerging markets, is facilitating greater accessibility to advanced cardiac implant technologies.
Overall, the market is expected to witness sustained growth, driven by clinical advantages, technological innovation, and the escalating global burden of cardiovascular diseases.
Key Takeaways
- Market Size: Global Leadless Pacemakers Market size is expected to be worth around US$ 1665.9 Million by 2035 from US$ 807.8 Million in 2025.
- Market Share: The market growing at a CAGR of 7.5% during the forecast period from 2026 to 2035.
- Product Type Analysis: Single-chamber devices, accounting for approximately 68.2% of the total market share in 2025.
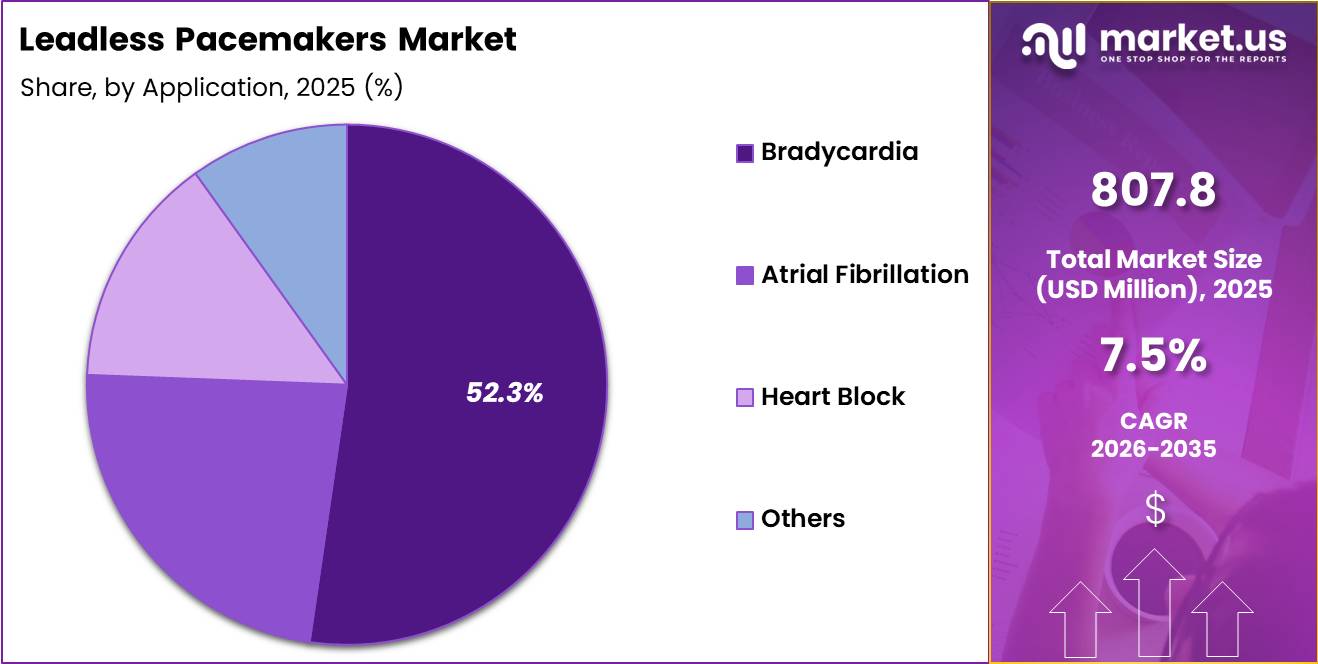
- Application Analysis: Bradycardia treatment, which holds a dominant share of 52.3% in 2025.
- Patient Type Analysis: Pediatric segment holds the largest share, accounting for approximately 49.5% in 2025
- End User Analysis: Hospitals dominate the leadless pacemakers market, capturing 55.9% of the total share in 2025.
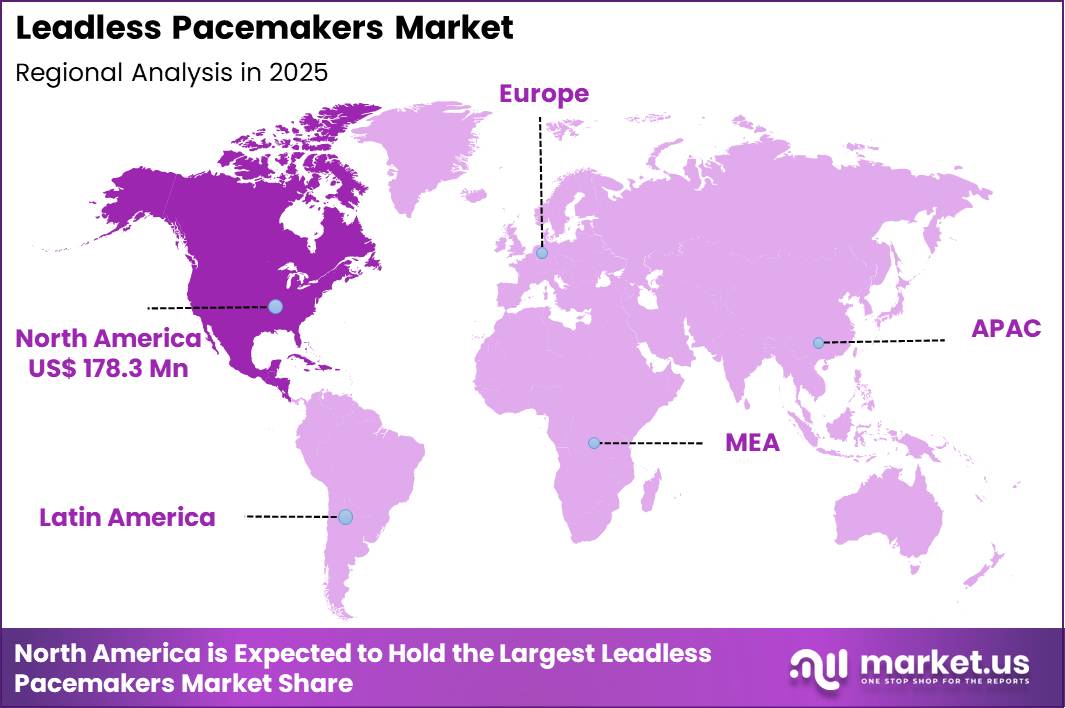
- Regional Analysis: In 2025, North America led the market, achieving over 43.3% share with a revenue of US$ 178.3 Million.
Product Type Analysis
The leadless pacemakers market, when segmented by product type, demonstrates a strong dominance of single-chamber devices, accounting for approximately 68.2% of the total market share in 2025. This dominance is primarily attributed to their simplified design, reduced procedural complexity, and widespread clinical acceptance for treating patients with standard pacing requirements.
Single-chamber leadless pacemakers are extensively utilized in cases where pacing is required in only one heart chamber, typically the right ventricle, making them suitable for a large patient pool. Additionally, their lower cost compared to more advanced systems contributes to higher adoption across emerging and cost-sensitive markets.
In contrast, dual-chamber leadless pacemakers represent an emerging segment with significant growth potential. These devices are designed to coordinate pacing between both the atrium and ventricle, thereby offering improved physiological pacing and better clinical outcomes in complex cardiac conditions.
Although currently limited by higher costs and technological challenges, ongoing advancements and clinical trials are expected to enhance their adoption. The segment is projected to witness accelerated growth as innovation improves device synchronization capabilities and expands indications.
Application Analysis
Based on application, the leadless pacemakers market is led by bradycardia treatment, which holds a dominant share of 52.3% in 2025. This segment’s prominence is driven by the high global prevalence of bradycardia, particularly among aging populations, and the clinical effectiveness of leadless pacemakers in managing slow heart rhythms.
The minimally invasive nature of these devices, combined with reduced risk of infection and complications, has significantly supported their adoption in bradycardia management. Favorable clinical outcomes and increasing physician preference for leadless systems further reinforce segment growth.
Other key application segments include atrial fibrillation, heart block, and a category classified as others. The atrial fibrillation segment is witnessing steady growth due to the rising incidence of arrhythmias globally and increasing awareness regarding advanced cardiac treatment options.
Heart block applications also contribute notably, as leadless pacemakers provide reliable pacing solutions in conduction disorders. The “others” category encompasses less common cardiac conditions but remains relevant due to ongoing expansion in clinical indications. Collectively, these segments are expected to grow as technological advancements broaden therapeutic applicability.
Patient Type Analysis
The market segmentation by patient type indicates that the pediatric segment holds the largest share, accounting for approximately 49.5% in 2025. This dominance can be attributed to the increasing preference for minimally invasive cardiac procedures in younger populations, where long-term complications associated with traditional pacemakers are a major concern.
Leadless pacemakers offer significant advantages such as reduced infection risk, absence of leads, and improved cosmetic outcomes, making them particularly suitable for pediatric patients. Additionally, advancements in device miniaturization have enhanced their applicability in smaller anatomies.
The adult segment also represents a substantial portion of the market, driven by the rising incidence of cardiovascular diseases and increasing adoption of advanced cardiac technologies. Adults benefit from improved procedural safety and reduced hospital stays associated with leadless systems.
Meanwhile, the geriatric segment is expected to witness notable growth due to the higher prevalence of arrhythmias and age-related cardiac conditions. However, adoption in elderly populations may be moderated by comorbidities and procedural risks. Overall, all segments are projected to expand, supported by increasing clinical confidence and technological progress.
End User Analysis
In terms of end users, hospitals dominate the leadless pacemakers market, capturing 55.9% of the total share in 2025. This dominance is primarily due to the availability of advanced infrastructure, skilled cardiologists, and comprehensive post-procedural care facilities within hospital settings.
Hospitals are typically the primary centers for complex cardiac procedures, including the implantation of leadless pacemakers, ensuring higher patient inflow and procedural volume. Additionally, favorable reimbursement policies and access to advanced imaging and diagnostic tools further strengthen hospital-based adoption.
Ambulatory surgical centers (ASCs) represent a growing segment, driven by the increasing shift toward outpatient procedures and cost-effective treatment options. ASCs offer advantages such as reduced hospitalization time and lower procedural costs, making them an attractive option for selected patient groups.
The “others” category, including specialty clinics and cardiac care centers, also contributes to market growth by expanding access to specialized treatment. As healthcare systems increasingly emphasize efficiency and cost containment, the role of ASCs and specialized centers is expected to grow, although hospitals will likely maintain their leading position in the near term.
Key Market Segments
By Product Type
- Single-chamber Leadless Pacemakers
- Dual-chamber Leadless Pacemakers
By Application
- Bradycardia
- Atrial Fibrillatio
- Heart Block
- Others
By Patient Type
- Pediatric
- Adult
- Geriatric
By End User
- Hospitals
- Ambulatory Surgical Centers
- Others
Driving Factors
Rising burden of cardiac arrhythmias and procedural advantages
The growth of the leadless pacemakers market is primarily driven by the increasing global burden of cardiac rhythm disorders such as bradycardia and atrial fibrillation. According to the U.S. Food and Drug Administration, nearly 1 million pacemakers are implanted annually worldwide, indicating a large and expanding patient base requiring pacing therapies.Traditional pacemakers rely on transvenous leads, which are associated with complications such as infections, lead dislodgement, and device failure. Leadless systems eliminate these risks by being fully self-contained and implanted directly into the heart, improving clinical outcomes.
Clinical data evaluated by the FDA showed 98% effective pacing performance with leadless devices and complication rates below 7%, demonstrating strong safety and efficacy. Additionally, increasing adoption of minimally invasive procedures is supporting demand, as leadless pacemakers are delivered via catheter rather than open surgery.
The combination of high disease prevalence, improved safety profiles, and procedural efficiency is significantly accelerating the adoption of leadless pacing technologies across healthcare systems.
Trending Factors
Technological advancements and dual-chamber innovation
A key trend shaping the leadless pacemakers market is rapid technological advancement, particularly the transition from single-chamber to dual-chamber systems. The American College of Cardiology reported that the first dual-chamber leadless pacemaker received regulatory approval with an implant success rate of 98.3% and over 97% atrioventricular synchrony, marking a major milestone in device capability.Earlier generations of leadless pacemakers were limited to single-chamber pacing, restricting their use in broader patient populations. However, innovations such as wireless communication between implanted units and retrievable device designs are expanding clinical applications. Miniaturization trends have also resulted in devices comparable in size to a AAA battery, enhancing patient comfort and reducing implantation risks.
Furthermore, improvements in battery longevity—often exceeding 10–15 years in clinical practice—are strengthening long-term value propositions. These advancements indicate a transition toward more versatile, durable, and patient-centric cardiac devices, supporting sustained market expansion and increasing physician acceptance globally.
Restraining Factors
High cost and limited patient eligibility
Despite technological progress, the leadless pacemakers market faces constraints related to high procedural costs and limited eligibility criteria. Leadless pacemaker implantation remains significantly more expensive than conventional systems, particularly in emerging economies.For instance, recent clinical adoption in India indicates procedure costs reaching ₹17–25 lakh (approximately USD 20,000–30,000), which restricts accessibility for a large patient population. Additionally, reimbursement policies are still evolving; the Centers for Medicare & Medicaid Services has implemented conditional coverage frameworks requiring evidence development, indicating cautious payer adoption.
From a clinical standpoint, current leadless devices are primarily indicated for single-chamber ventricular pacing, limiting their use in patients requiring more complex pacing therapies.
Certain anatomical constraints and contraindications, such as incompatible implanted devices or vascular limitations, further restrict adoption. These economic and clinical limitations collectively act as barriers, slowing widespread penetration despite strong technological advantages.
Opportunity
Expansion in minimally invasive cardiology and emerging markets
Significant opportunities exist in the expansion of minimally invasive cardiology and increasing healthcare investments in emerging markets. Leadless pacemakers align strongly with global healthcare priorities focused on reducing hospital stays and improving recovery outcomes.Clinical evidence indicates that minimally invasive implantation enables faster discharge, with some patients released within 3 days post-procedure, highlighting efficiency gains.
Government healthcare institutions are also beginning to adopt these technologies; for example, public hospitals in India have successfully implemented leadless pacemaker procedures, indicating growing institutional acceptance.
Furthermore, advancements in device design, including retrievability and modular systems, are expected to expand indications beyond current limitations. As healthcare infrastructure improves in Asia-Pacific and Latin America, and awareness of advanced cardiac care increases, demand for innovative pacing solutions is projected to rise.
The integration of digital monitoring and long-life battery systems further enhances value, positioning leadless pacemakers as a transformative solution within next-generation cardiac rhythm management.
Regional Analysis
The regional distribution of the Leadless Pacemakers Market demonstrates a strong concentration in North America, which emerged as the dominant region in 2025. A market share exceeding 43.3% was recorded, with total revenue reaching approximately US$ 178.3 million.
This leadership position can be attributed to the presence of advanced healthcare infrastructure, high adoption of minimally invasive cardiac procedures, and favorable reimbursement frameworks. In addition, the growing prevalence of cardiovascular diseases and increasing awareness regarding innovative cardiac rhythm management devices have supported market expansion in the region.
Europe is identified as the second-largest market, driven by rising geriatric population levels and increased demand for technologically advanced pacemaker solutions. Supportive regulatory policies and ongoing clinical research initiatives have further contributed to regional growth.
The Asia-Pacific region is expected to exhibit the fastest growth rate over the forecast period. This growth is supported by improving healthcare facilities, expanding patient population, and increasing investments in medical technology. Countries such as China, India, and Japan are witnessing higher adoption rates due to rising healthcare expenditure and awareness.
Meanwhile, Latin America and the Middle East & Africa are projected to experience steady growth, supported by gradual improvements in healthcare access and infrastructure development.
Key Regions and Countries
North America
- The US
- Canada
Europe
- Germany
- France
- The U.K.
- Italy
- Spain
- Russia & CIS
- Rest of Europe
Asia Pacific
- China
- India
- Japan
- South Korea
- ASEAN
- Australia & New Zealand
- Rest of Asia Pacific
Middle East & Africa
- GCC
- South Africa
- Rest of Middle East & Africa
Latin America
- Brazil
- Mexico
- Rest of Latin America
Key Players Analysis
The competitive landscape of the Leadless Pacemakers Market is characterized by high consolidation and strong technological intensity, with a limited number of global players dominating market revenues.
Leading companies such as Medtronic plc and Abbott Laboratories collectively account for a significant share of the market, supported by their commercially established product platforms, including Micra and Aveir. These companies maintain leadership through continuous innovation, strong clinical evidence, and extensive global distribution networks.
Medtronic is recognized as the first mover in the segment, leveraging its early commercialization advantage and large-scale adoption of its leadless pacing systems. Meanwhile, Abbott has strengthened its competitive position through advancements in dual-chamber leadless pacemakers, expanding the addressable patient population.
Other key participants, including Boston Scientific Corporation, EBR Systems, Inc., and MicroPort Scientific Corporation, are focusing on product pipeline development and clinical trials to enhance their market presence.
Strategic initiatives such as mergers and acquisitions, partnerships, and geographic expansion are widely adopted to strengthen competitive positioning. Additionally, emerging players are investing in miniaturization, battery longevity, and improved device performance to compete with established leaders. Overall, the market is expected to remain innovation-driven, with technological differentiation serving as a key competitive factor.
Market Key Players
- Medtronic plc
- Abbott Laboratories
- Boston Scientific Corporation
- MicroPort Scientific Corporation
- Biotronik SE & Co. KG
- Osypka Medical GmbH
- Lepu Medical Technology Co., Ltd.
- Shree Pacetronix Ltd.
- Cook Medical, Inc.
- OSCOR Inc.
- IMARC
- CathRx Ltd.
- Norton Healthcare
- Pacetronix
- LivaNova PLC
- Others
Recent Developments
- Boston Scientific Corporation (February 2026): In February 2026, Boston Scientific highlighted progress on its Empower leadless pacemaker program as part of its global pacemaker portfolio push, positioning Empower as a next-generation miniaturized device aimed at reducing lead-related complications and improving long-term device performance in complex rhythm patients.
- Medtronic plc (February 2026): In February 2026, Medtronic continued to scale its Micra AV and Micra VR leadless pacemakers globally, using new clinical evidence on reduced infection risk and shorter hospital stays to deepen penetration in high-volume cardiac centers and reinforce its leadership in transcatheter pacing systems
- Abbott Laboratories (February 2026): In February 2026, Abbott accelerated commercial and clinical momentum for its Aveir VR leadless pacemaker platform, focusing on centers of excellence in North America and Europe and emphasizing future roadmap potential for modular, dual-chamber leadless pacing architectures.
- Biotronik SE & Co. KG (January 2025): In January 2025, Biotronik entered a research partnership with European electrophysiology experts to explore MRI-compatible leadless pacing concepts, targeting future systems that maintain full diagnostic imaging flexibility while lowering the risk profile associated with conventional leads.
Report Scope
Report Features Description Market Value (2025) US$ 807.8 Million Forecast Revenue (2035) US$ 1665.9 Million CAGR (2026-2035) 7.5% Base Year for Estimation 2025 Historic Period 2020-2024 Forecast Period 2026-2035 Report Coverage Revenue Forecast, Market Dynamics, Competitive Landscape, Recent Developments Segments Covered By Product Type (Single-chamber Leadless Pacemakers, Dual-chamber Leadless Pacemakers) By Application (Bradycardia, Atrial Fibrillation, Heart Block, Others) By Patient Type (Pediatric, Adult, Geriatric) By End User (Hospitals, Ambulatory Surgical Centers, Others) Regional Analysis North America – The US, Canada; Europe – Germany, France, U.K., Italy, Spain, Russia & CIS, Rest of Europe; Asia Pacific – China, India, Japan, South Korea, ASEAN, Australia & New Zealand, Rest of Asia Pacific; Middle East & Africa – GCC, South Africa, Rest of Middle East & Africa; Latin America – Brazil, Mexico, Rest of Latin America Competitive Landscape Medtronic plc, Abbott Laboratories, Boston Scientific Corporation, MicroPort Scientific Corporation, Biotronik SE & Co. KG, Osypka Medical GmbH, Lepu Medical Technology Co., Ltd., Shree Pacetronix Ltd., Cook Medical, Inc., OSCOR Inc., IMARC, CathRx Ltd., Norton Healthcare, Pacetronix, LivaNova PLC, Others, Customization Scope Customization for segments, region/country-level will be provided. Moreover, additional customization can be done based on the requirements. Purchase Options We have three licenses to opt for: Single User License, Multi-User License (Up to 5 Users), Corporate Use License (Unlimited User and Printable PDF) 
-
-
- Medtronic plc
- Abbott Laboratories
- Boston Scientific Corporation
- MicroPort Scientific Corporation
- Biotronik SE & Co. KG
- Osypka Medical GmbH
- Lepu Medical Technology Co., Ltd.
- Shree Pacetronix Ltd.
- Cook Medical, Inc.
- OSCOR Inc.
- IMARC
- CathRx Ltd.
- Norton Healthcare
- Pacetronix
- LivaNova PLC
- Others










