Global Biosimilar Contract Manufacturing Market By Product Type (Monoclonal Antibodies, Recombinant Proteins, Peptides, Granulocyte Colony-Stimulating Factor, Insulin, Epoetin, Interferon, Follitropin, Others) By Application, (Oncology, Autoimmune Diseases, Chronic Diseases, Infectious Diseases, Blood Disorders, Others) By Service Type (Process Development, Analytical Services, Fill and Finish, Regulatory Support, Commercial Manufacturing) By End User (Pharmaceutical Companies, Biotechnology Companies, Biosimilar Developers), Region and Companies – Industry Segment Outlook, Market Assessment, Competition Scenario, Trends and Forecast 2026-2035
- Published date: April 2026
- Report ID: 185449
- Number of Pages: 238
- Format:
-
keyboard_arrow_up
Quick Navigation
Report Overview
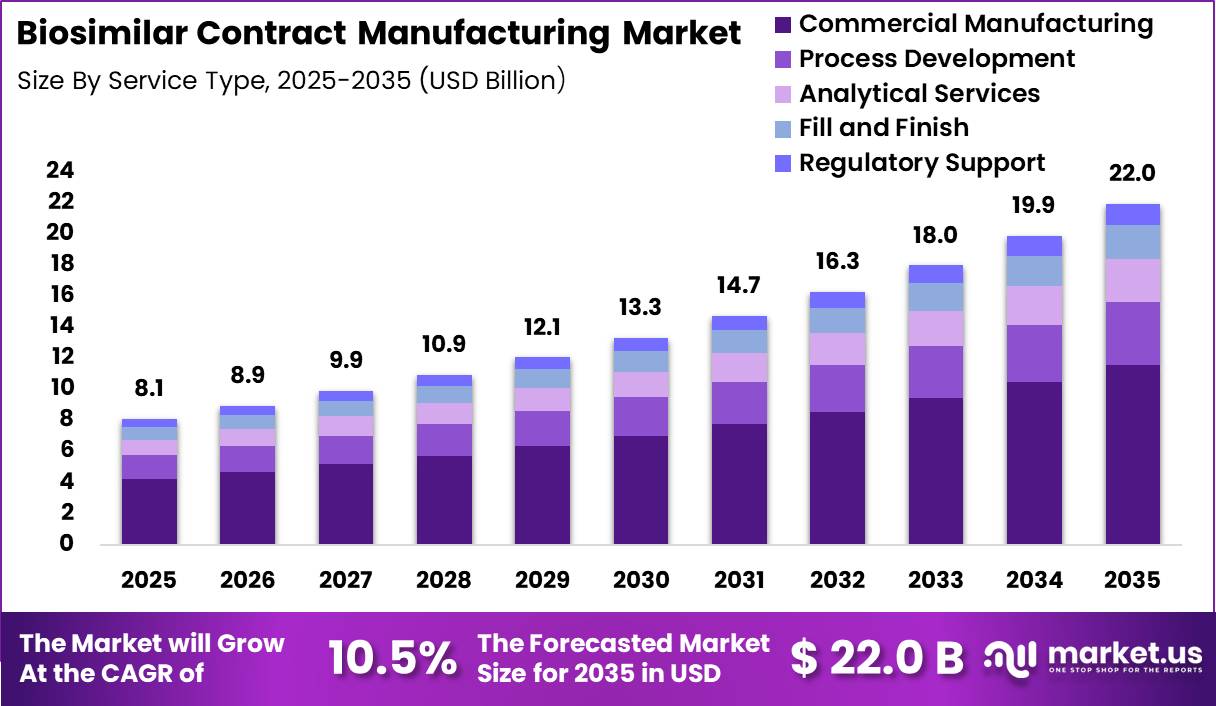
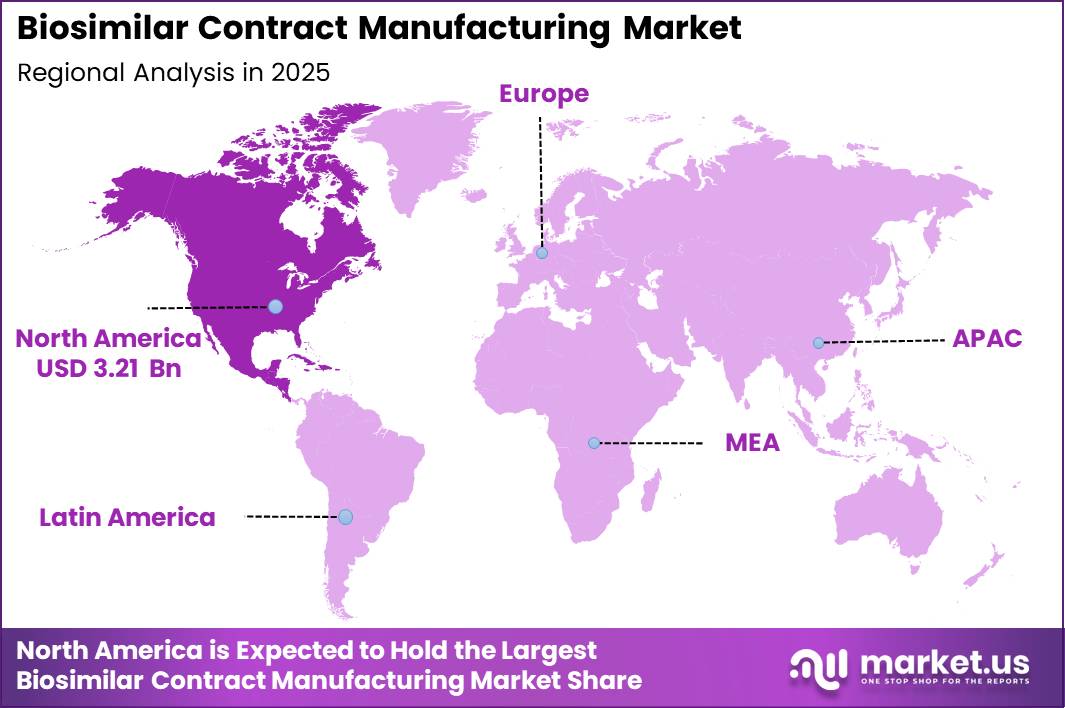
Global Biosimilar Contract Manufacturing Market size is expected to be worth around US$ 22.0 Billion by 2035 from US$ 8.1 Billion in 2025, growing at a CAGR of 10.5% during the forecast period from 2026 to 2035. In 2025, North America led the market, achieving over 42.50% share with a revenue of US$ 3.21 Billion.
Biosimilar contract manufacturing refers to the outsourcing of complex biologic drug production to specialized contract manufacturing organizations (CMOs). Biosimilars are defined by the World Health Organization as highly similar versions of approved biologic medicines, with no clinically meaningful differences in safety, purity, and potency.
Unlike small-molecule generics, biosimilars are produced using living cells, making their manufacturing processes highly sophisticated and capital-intensive.
The role of CMOs has become critical in this ecosystem, as they provide advanced capabilities such as cell line development, process optimization, large-scale bioreactor production, and regulatory support.
These capabilities are essential because biosimilar manufacturing requires stringent quality control and compliance with global regulatory frameworks, including those established by the U.S. Food and Drug Administration. As of 2026, the FDA has approved over 80 biosimilars, reflecting increasing regulatory acceptance and maturity of the sector.
The growing demand for biosimilars is strongly linked to the high cost of biologic therapies. According to FDA data, biologics account for approximately 5% of prescriptions but nearly 51% of total drug spending in the United States.
This cost imbalance has accelerated the adoption of biosimilars as cost-effective alternatives, thereby increasing reliance on contract manufacturing for scalable production. Additionally, more than 300 biosimilar candidates are currently in clinical development globally, indicating a robust future pipeline.
Technological advancements in cell culture, analytical characterization, and bioprocessing have further strengthened CMO capabilities. Strategic collaborations such as partnerships between biopharmaceutical companies and CMOs are enabling efficient knowledge sharing, capacity expansion, and faster commercialization. These partnerships are particularly important for complex biologics such as monoclonal antibodies, which accounted for nearly 45% of biosimilar usage in 2025.
However, the sector faces challenges including high initial investment, complex regulatory requirements, and manufacturing variability. Despite these constraints, biosimilar contract manufacturing is expected to expand steadily, driven by increasing global demand for affordable biologics, supportive regulatory policies, and continuous innovation in biomanufacturing technologies.
Key Takeaways
- Market Size: Global Biosimilar Contract Manufacturing Market size is expected to be worth around US$ 22.0 Billion by 2035 from US$ 8.1 Billion in 2025.
- Market Share: The market growing at a CAGR of 10.5% during the forecast period from 2026 to 2035.
- Product type Analysis: Monoclonal antibodies are projected to dominate the segment, accounting for approximately 47.8% of the market share in 2025.
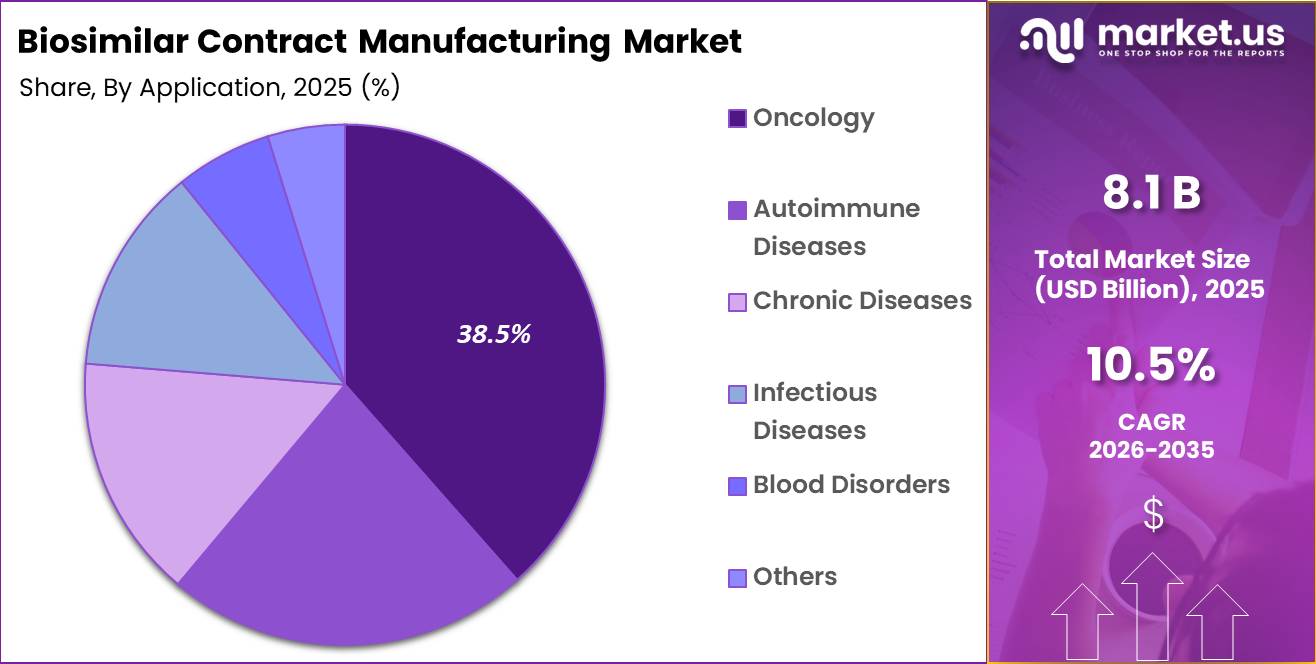
- Application Analysis: Oncology is anticipated to lead the segment, capturing around 38.5% of the total market share.
- Service Type Analysis: Commercial manufacturing is expected to dominate, accounting for approximately 52.6%.
- End User Analysis: Pharmaceutical companies are projected to hold the largest share, accounting for approximately 41.3% in 2025.
- Regional Analysis: In 2025, North America led the market, achieving over 42.50% share with a revenue of US$ 3.21 Billion.
Product type Analysis
The biosimilar contract manufacturing market is segmented by product type into monoclonal antibodies, recombinant proteins, peptides, granulocyte colony-stimulating factor (G-CSF), insulin, epoetin, interferon, follitropin, and others. Monoclonal antibodies are projected to dominate the segment, accounting for approximately 47.8% of the market share in 2025.
This dominance is attributed to their widespread application in treating complex and chronic diseases, particularly oncology and autoimmune conditions, alongside strong patent expirations of originator biologics. The manufacturing complexity and high capital requirements associated with monoclonal antibodies further drive outsourcing to specialized contract manufacturers.
Recombinant proteins and peptides represent significant segments due to their established therapeutic applications and relatively streamlined production processes. Meanwhile, products such as insulin, interferon, and epoetin continue to witness steady demand owing to the rising global burden of diabetes and hematological disorders.
Emerging categories including follitropin and G-CSF are expected to experience moderate growth, supported by increasing adoption in fertility treatments and oncology supportive care. Overall, product diversification is enhancing contract manufacturing opportunities across multiple biologic classes.
Application Analysis
Based on application, the biosimilar contract manufacturing market is categorized into oncology, autoimmune diseases, chronic diseases, infectious diseases, blood disorders, and others. Oncology is anticipated to lead the segment, capturing around 38.5% of the total market share in 2025. This leadership is driven by the increasing global incidence of cancer and the high demand for cost-effective biologic therapies.
Biosimilars offer significant cost advantages, thereby improving patient accessibility and driving outsourcing needs. Autoimmune diseases represent another prominent segment, supported by the growing prevalence of conditions such as rheumatoid arthritis and psoriasis, where biologics remain a primary treatment modality.
Chronic diseases, including diabetes and cardiovascular disorders, contribute steadily to market demand, particularly through insulin and related biologics. Infectious diseases and blood disorders also present notable opportunities, with biosimilars increasingly being utilized in treatment protocols to reduce healthcare expenditure.
The “others” category includes niche therapeutic areas that are gradually gaining traction. Overall, the application landscape reflects a strong alignment with global disease burden trends and cost-containment strategies in healthcare systems.
Service Type Analysis
The market is segmented by service type into commercial manufacturing, process development, analytical services, fill and finish, and regulatory support. Commercial manufacturing is expected to dominate, accounting for approximately 52.6% of the market share in 2025. This dominance is attributed to the increasing number of biosimilars reaching late-stage development and commercialization, necessitating large-scale production capabilities.
Contract manufacturers are being increasingly engaged to manage complex production requirements, reduce operational costs, and ensure regulatory compliance. Process development services play a critical role in optimizing yield, scalability, and cost-efficiency, thereby witnessing steady demand across early and mid-stage development pipelines.
Analytical services are essential for ensuring product comparability, quality assurance, and adherence to stringent regulatory standards. Fill and finish services are gaining importance due to the need for sterile and precise drug formulation and packaging.
Additionally, regulatory support services are expanding as companies navigate complex approval pathways across multiple regions. Collectively, these service segments contribute to a comprehensive outsourcing ecosystem supporting biosimilar commercialization.
End User Analysis
Based on end users, the biosimilar contract manufacturing market is segmented into pharmaceutical companies, biotechnology companies, and biosimilar developers. Pharmaceutical companies are projected to hold the largest share, accounting for approximately 41.3% in 2025. This dominance is driven by their strong financial capabilities, extensive product portfolios, and strategic focus on expanding biosimilar pipelines to offset revenue losses from patent expirations.
These companies increasingly rely on contract manufacturing organizations to enhance production efficiency and accelerate time-to-market. Biotechnology companies represent a significant segment, leveraging specialized expertise in biologics development while outsourcing manufacturing to reduce infrastructure investments.
Biosimilar developers, including smaller and emerging firms, are also contributing to market growth, supported by favorable regulatory frameworks and rising investment in biosimilar research. The growing trend of strategic collaborations and outsourcing partnerships across all end-user categories is reinforcing the role of contract manufacturers as critical enablers in the biosimilar value chain.
Key Market Segments
By Product Type
- Monoclonal Antibodies
- Recombinant Proteins
- Peptides
- Granulocyte Colony-Stimulating Factor
- Insulin
- Epoetin
- Interferon
- Follitropin
- Others
By Application
- Oncology
- Autoimmune Diseases
- Chronic Diseases
- Infectious Diseases
- Blood Disorders
- Others
By Service Type
- Process Development
- Analytical Services
- Fill and Finish
- Regulatory Support
- Commercial Manufacturing
By End User
- Pharmaceutical Companies
- Biotechnology Companies
- Biosimilar Developers
Driving Factors
Increasing biologics demand and cost containment pressure
The growth of the biosimilar contract manufacturing market is strongly driven by the rising global demand for biologic therapies and the need to reduce healthcare expenditure.According to the U.S. Food and Drug Administration, biologics represent a significant share of treatment for chronic diseases such as cancer, diabetes, and autoimmune disorders, but they are complex and expensive to manufacture.
It has been reported that biologic drugs account for nearly 51% of total drug spending in the United States while representing only about 5% of prescriptions, highlighting a major cost imbalance. Biosimilars, typically priced 30–40% lower than reference biologics, offer a cost-efficient alternative, increasing adoption across healthcare systems.
As patents of blockbuster biologics expire, outsourcing to contract manufacturers has increased due to the high complexity of production involving living cells and advanced bioprocessing.
Additionally, regulatory frameworks such as the Biologics Price Competition and Innovation Act have streamlined approvals, supporting manufacturing demand. This combination of cost pressure, rising disease burden, and regulatory support continues to accelerate outsourcing trends in biosimilar production.
Trending Factors
Regulatory streamlining and technological advancement in manufacturing
A significant trend in the biosimilar contract manufacturing market is the ongoing regulatory simplification combined with advancements in bioprocessing technologies. The U.S. Food and Drug Administration has introduced initiatives to reduce development complexity, including proposals to cut certain clinical study requirements and allow the use of global clinical data.These measures are estimated to reduce development costs by up to 50%, equivalent to approximately USD 20 million per biosimilar program. Additionally, regulatory agencies such as the European Medicines Agency have established robust comparability frameworks ensuring safety and efficacy while encouraging faster approvals.
Technological advancements, including single-use bioreactors and continuous manufacturing, are being increasingly adopted to improve scalability and reduce contamination risks. The shift toward contract development and manufacturing organizations (CDMOs) is also evident, as companies aim to optimize capital expenditure and accelerate time-to-market.
This trend reflects a transition toward more efficient, flexible, and globally harmonized biosimilar production ecosystems, strengthening the role of specialized contract manufacturers in the value chain.
Restraining Factors
High manufacturing complexity and stringent regulatory requirements
The biosimilar contract manufacturing market faces significant restraints due to the inherent complexity of biologic production and stringent regulatory expectations. Unlike small-molecule generics, biosimilars are derived from living cells, making them highly sensitive to minor process variations.The U.S. Food and Drug Administration emphasizes that biosimilars must demonstrate “no clinically meaningful differences” in safety and efficacy compared to reference products, requiring extensive analytical, non-clinical, and clinical studies. Manufacturing changes, even minor ones, can impact product quality, leading to increased validation and compliance costs.
Furthermore, development timelines can extend to 5–8 years, significantly longer than traditional generics. High capital investment in bioreactors, purification systems, and quality control infrastructure further limits market entry for smaller players.
Regulatory heterogeneity across regions adds additional complexity, requiring manufacturers to meet multiple compliance standards. These challenges increase operational risks and limit scalability, thereby constraining the growth of contract manufacturing services despite strong demand for biosimilars.
Opportunity
Expanding approvals and global access to biosimilars
The expansion of biosimilar approvals and increasing focus on global healthcare accessibility present significant opportunities for the contract manufacturing market. As of 2024, the U.S. Food and Drug Administration has approved over 60 biosimilars, reflecting a steady increase in market penetration.In addition, regulatory agencies are actively promoting biosimilar adoption to enhance patient access and reduce treatment costs. Biosimilars are expected to improve affordability, with healthcare systems encouraging their use through reimbursement policies and substitution guidelines.
Emerging markets in Asia, Latin America, and Africa are witnessing increased demand due to rising chronic disease prevalence and limited access to high-cost biologics. Contract manufacturers are well-positioned to capitalize on this trend by offering cost-efficient, large-scale production capabilities.
Furthermore, partnerships between pharmaceutical companies and CDMOs are increasing to address capacity constraints and accelerate commercialization timelines. This expanding approval landscape and global demand create a favorable environment for sustained growth in biosimilar contract manufacturing services.
Regional Analysis
North America has been identified as the leading region in the biosimilar contract manufacturing market, accounting for over 42.50% share and generating approximately US$ 3.21 billion in 2025. This dominance is primarily supported by a highly advanced biopharmaceutical ecosystem, strong regulatory frameworks, and significant investment in biologics manufacturing infrastructure.
The presence of the U.S. Food and Drug Administration (FDA) has played a critical role in market expansion. The agency has approved more than 70 biosimilars, reflecting an increasing acceptance of biosimilar therapies and a structured regulatory pathway that supports manufacturing outsourcing and commercialization.
Furthermore, biologics account for a disproportionately high share of drug spending in the United States—around 51% despite representing a small fraction of prescriptions—creating strong demand for cost-effective biosimilars and associated contract manufacturing services.
Additionally, advanced technological capabilities, including cell-line development and bioprocessing, combined with high compliance standards such as Good Manufacturing Practices (GMP), have strengthened the region’s leadership.
Overall, sustained regulatory support, high biologics expenditure, and mature outsourcing infrastructure continue to reinforce North America’s dominant position in the biosimilar contract manufacturing market.
Key Regions and Countries
North America
- The US
- Canada
Europe
- Germany
- France
- The U.K.
- Italy
- Spain
- Russia & CIS
- Rest of Europe
Asia Pacific
- China
- India
- Japan
- South Korea
- ASEAN
- Australia & New Zealand
- Rest of Asia Pacific
Middle East & Africa
- GCC
- South Africa
- Rest of Middle East & Africa
Latin America
- Brazil
- Mexico
- Rest of Latin America
Key Players Analysis
The biosimilar contract manufacturing market is characterized by a concentrated group of global CDMOs and pharmaceutical companies leveraging advanced biologics capabilities and regulatory compliance frameworks. Major players such as Merck KGaA, Thermo Fisher Scientific Inc., AGC Biologics, Pfizer CentreOne, AbbVie, Rentschler Biopharma SE, Biocon Limited, JRS Pharma, and ProBioGen AG operate across integrated value chains, including cell line development, GMP manufacturing, and fill–finish services.
These firms compete primarily on manufacturing scale, process optimization, and compliance with stringent regulatory standards such as those defined by agencies like the FDA and EMA.
Rentschler Biopharma SE and AGC Biologics emphasize specialized biologics production, while Biocon Limited provides cost-efficient biosimilar manufacturing for global markets. Large-scale providers such as Thermo Fisher Scientific Inc. and AbbVie focus on end-to-end solutions and complex monoclonal antibody production.
Overall, strategic partnerships, capacity expansion, and adoption of single-use and continuous bioprocessing technologies are identified as key competitive strategies driving market positioning and long-term growth.
Market Key Players
- Lonza Group AG
- Samsung Biologics Co., Ltd.
- Boehringer Ingelheim BioXcellence
- FUJIFILM Diosynth Biotechnologies
- WuXi Biologics
- Celltrion Inc.
- Merck KGaA
- Thermo Fisher Scientific Inc.
- AGC Biologics
- Pfizer CentreOne
- AbbVie Contract Manufacturing
- Rentschler Biopharma SE
- Biocon Limited
- JRS Pharma
- ProBioGen AG
- Others
Recent Developments
- WuXi Biologics (February 2025): WuXi Biologics signed a trispecific T-cell engager agreement with Candid Therapeutics valued at up to USD 925 million, reinforcing its strategy to leverage advanced biologics modalities and deepen long-term outsourcing relationships that often extend into biosimilar-style lifecycle programs.
- Samsung Biologics (April 2025): Samsung Biologics brought its 180,000-litre Plant 5 in Songdo into full operation, taking its Korean capacity to 785,000 litres and strengthening its ability to take on large, late-stage and commercial biosimilar manufacturing mandates on a truly global scale.
- Lonza Group AG (March 2026): Lonza’s growth strategy update highlighted the operational integration of Roche’s Vacaville site, adding about 700,000 litres of mammalian bioreactor capacity, alongside a planned CHF 500 million investment to expand large-scale capacity in Portsmouth and Stein, which together reinforce Lonza’s position as a go-to partner for late-stage biologics and biosimilar manufacturing at commercial scale.
- Biosimilar CMOs (October 2025): Market trackers highlighted that the biosimilar contract manufacturing segment crossed roughly USD 8.9–9.2 billion in value in 2025, with accelerating outsourcing from big pharma and specialty players as they sought cost-efficient biologics capacity without building greenfield plants.
Report Scope
Report Features Description Market Value (2025) US$ 8.1 Billion Forecast Revenue (2035) US$ 22.0 Billion CAGR (2026-2035) 10.5% Base Year for Estimation 2025 Historic Period 2020-2024 Forecast Period 2026-2035 Report Coverage Revenue Forecast, Market Dynamics, Competitive Landscape, Recent Developments Segments Covered By Product Type (Monoclonal Antibodies, Recombinant Proteins, Peptides, Granulocyte Colony-Stimulating Factor, Insulin, Epoetin, Interferon, Follitropin, Others) By Application, (Oncology, Autoimmune Diseases, Chronic Diseases, Infectious Diseases, Blood Disorders, Others) By Service Type (Process Development, Analytical Services, Fill and Finish, Regulatory Support, Commercial Manufacturing) By End User (Pharmaceutical Companies, Biotechnology Companies, Biosimilar Developers) Regional Analysis North America – The US, Canada; Europe – Germany, France, U.K., Italy, Spain, Russia & CIS, Rest of Europe; Asia Pacific – China, India, Japan, South Korea, ASEAN, Australia & New Zealand, Rest of Asia Pacific; Middle East & Africa – GCC, South Africa, Rest of Middle East & Africa; Latin America – Brazil, Mexico, Rest of Latin America Competitive Landscape Lonza Group AG, Samsung Biologics Co., Ltd., Boehringer Ingelheim BioXcellence, FUJIFILM Diosynth Biotechnologies, WuXi Biologics, Celltrion Inc., Merck KGaA, Thermo Fisher Scientific Inc., AGC Biologics, Pfizer CentreOne, AbbVie Contract Manufacturing, Rentschler Biopharma SE, Biocon Limited, JRS Pharma, ProBioGen AG, Others Customization Scope Customization for segments, region/country-level will be provided. Moreover, additional customization can be done based on the requirements. Purchase Options We have three licenses to opt for: Single User License, Multi-User License (Up to 5 Users), Corporate Use License (Unlimited User and Printable PDF)  Biosimilar Contract Manufacturing MarketPublished date: April 2026add_shopping_cartBuy Now get_appDownload Sample
Biosimilar Contract Manufacturing MarketPublished date: April 2026add_shopping_cartBuy Now get_appDownload Sample -
-
- Lonza Group AG
- Samsung Biologics Co., Ltd.
- Boehringer Ingelheim BioXcellence
- FUJIFILM Diosynth Biotechnologies
- WuXi Biologics
- Celltrion Inc.
- Merck KGaA
- Thermo Fisher Scientific Inc.
- AGC Biologics
- Pfizer CentreOne
- AbbVie Contract Manufacturing
- Rentschler Biopharma SE
- Biocon Limited
- JRS Pharma
- ProBioGen AG
- Others










