Global Automated Sample Storage Systems Market By Storage Temperature (Automated Cold Storage (Ambient to -20°C), Automated Ultra-Cold Storage (−80 °C ULT) and Automated Cryogenic Storage (Down to −190 °C)), By Component (System Platforms/Hardware, Software and Others), By Sample Type (Chemical & Compound Samples, Biological Samples and Others), By Application (Long-Term Sample Preservation, High-Throughput Sample Retrieval and Handling, Regulatory-Compliant Sample Management and Others), By End-User (Pharmaceutical and Biotechnology Companies, Academic and Clinical Research Laboratories, Biobanks and Others), Region and Companies – Industry Segment Outlook, Market Assessment, Competition Scenario, Trends and Forecast 2025-2034
- Published date: Dec 2025
- Report ID: 172758
- Number of Pages: 339
- Format:
-
keyboard_arrow_up
Quick Navigation
- Report Overview
- Key Takeaways
- Storage Temperature Analysis
- Component Analysis
- Sample Type Analysis
- Application Analysis
- End-User Analysis
- Key Market Segments
- Drivers
- Restraints
- Opportunities
- Impact of Macroeconomic / Geopolitical Factors
- Latest Trends
- Regional Analysis
- Key Players Analysis
- Recent Developments
- Report Scope
Report Overview
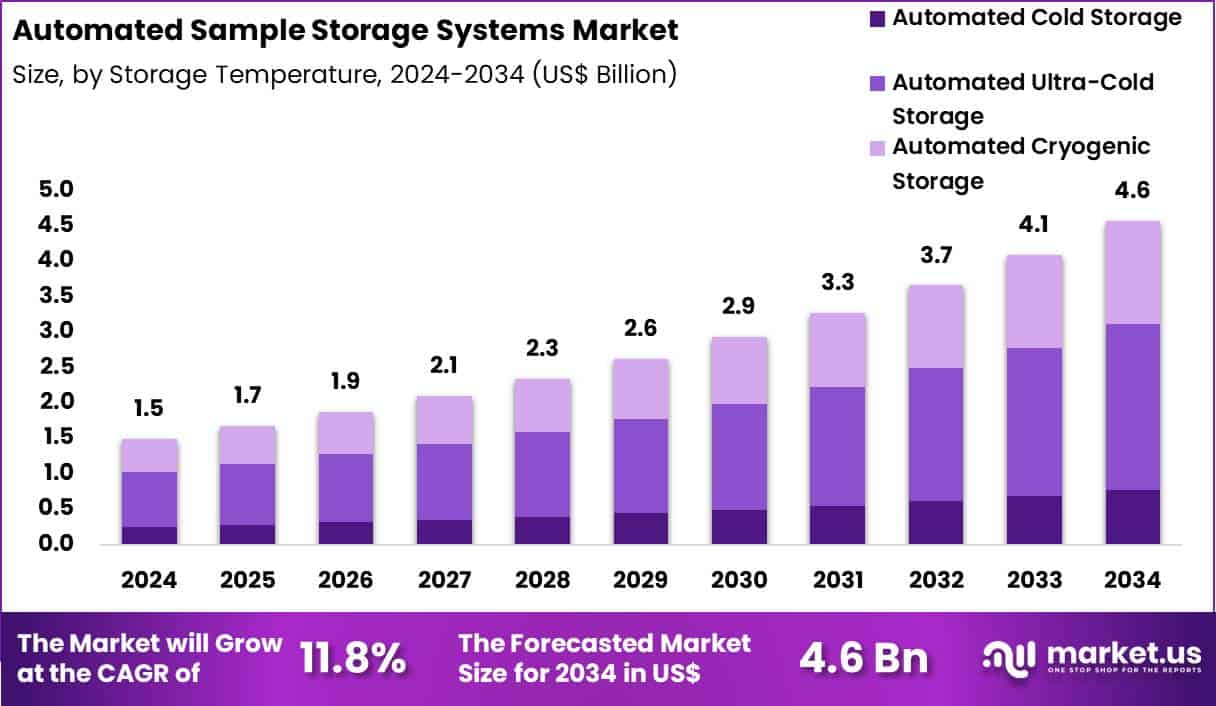
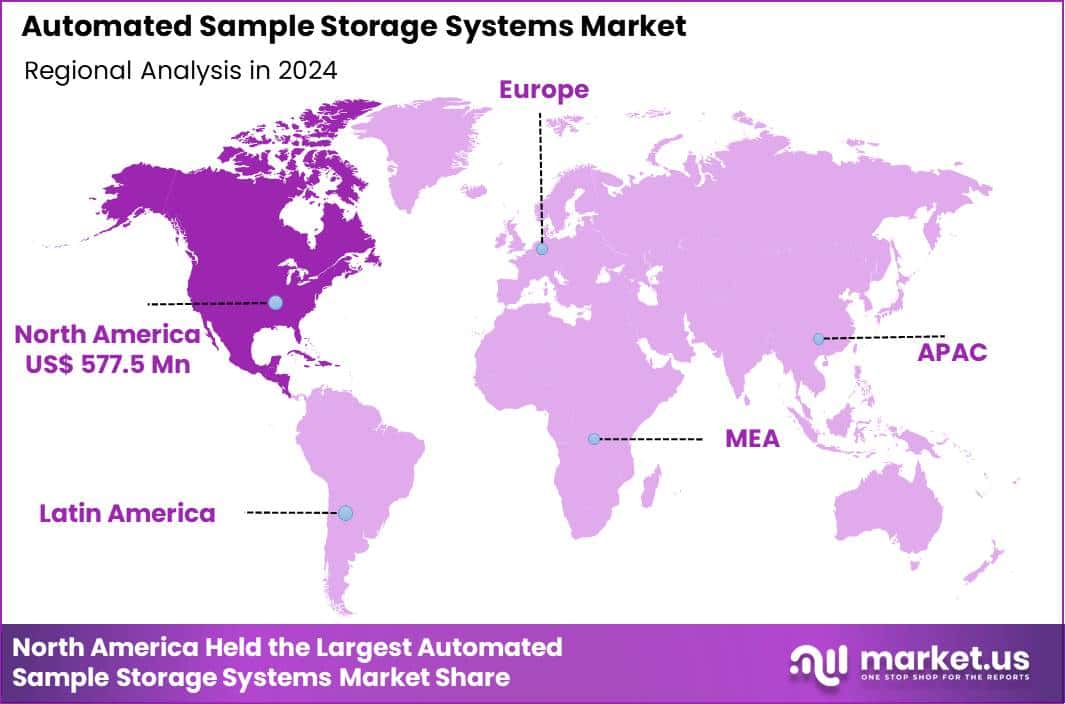
The Global Automated Sample Storage Systems Market size is expected to be worth around US$ 4.6 Billion by 2034 from US$ 1.5 Billion in 2024, growing at a CAGR of 11.8% during the forecast period 2025 to 2034. In 2024, North America led the market, achieving over 38.5% share with a revenue of US$ 577.5 Million.
Growing volumes of biological specimens in research and clinical settings compel laboratories to deploy automated sample storage systems that ensure integrity and traceability over extended periods. Biobanks increasingly rely on these platforms to preserve tissue samples for genomic studies, maintaining viability for downstream sequencing and biomarker discovery.
Pharmaceutical developers utilize automated systems to archive compound libraries, enabling rapid retrieval for high-throughput screening in drug candidate evaluation. These technologies support cell line repositories by controlling cryogenic conditions, safeguarding master cell banks essential for biologic production. Forensic facilities employ these storage solutions to manage DNA evidence, preventing degradation and facilitating long-term evidentiary access.
In March 2025, the BioArc Ultra received recognition as the Best New Sustainable Lab Product at the Scientists’ Choice Awards. The award reflects growing industry emphasis on energy-efficient, automated biobanking technologies as laboratories seek to reduce environmental impact while scaling sample storage capacity.
Laboratory operators pursue opportunities to implement modular automated storage units that accommodate varying tube formats, enhancing flexibility in vaccine development repositories during clinical trial phases. Manufacturers design scalable systems with robotic cherry-picking capabilities, streamlining access to rare disease biospecimens for collaborative research networks. These platforms expand applications in regenerative medicine by integrating temperature monitoring for stem cell storage, optimizing conditions for therapeutic product manufacturing.
Opportunities arise in toxicology testing archives, where automated retrieval minimizes exposure risks during repeat analyses of environmental samples. Companies advance hybrid ambient-to-cryogenic configurations, allowing seamless transitions for progressive freezing protocols in organoid banking. Firms invest in cloud-linked inventory management, improving audit trails for regulatory-compliant storage in personalized medicine initiatives.
Industry innovators introduce air-cycle refrigeration in automated systems, eliminating chemical refrigerants to achieve lower energy consumption in large-scale biobanking operations. Developers enhance sensor networks for real-time viability tracking, ensuring optimal preservation of live cell samples in immunotherapy research. Market participants refine compact footprint designs that maximize density, supporting efficient storage in space-constrained diagnostic laboratories.
Companies prioritize integration with laboratory information management systems, automating workflows for seamless sample tracking in proteomics studies. Innovators embed predictive maintenance algorithms to preempt downtime, maintaining uninterrupted access in critical pathogen repositories. Ongoing developments emphasize user-configurable compartments, adapting storage environments for diverse applications in metabolomics and microbiome sample archiving.
Key Takeaways
- In 2024, the market generated a revenue of US$ 1.5 Billion, with a CAGR of 11.8%, and is expected to reach US$ 4.6 billion by the year 2034.
- The storage temperature segment is divided into automated cold storage, automated ultra-cold storage and automated cryogenic storage, with automated ultra-cold storage (−80 °c ult)taking the lead in 2024 with a market share of 51.1%.
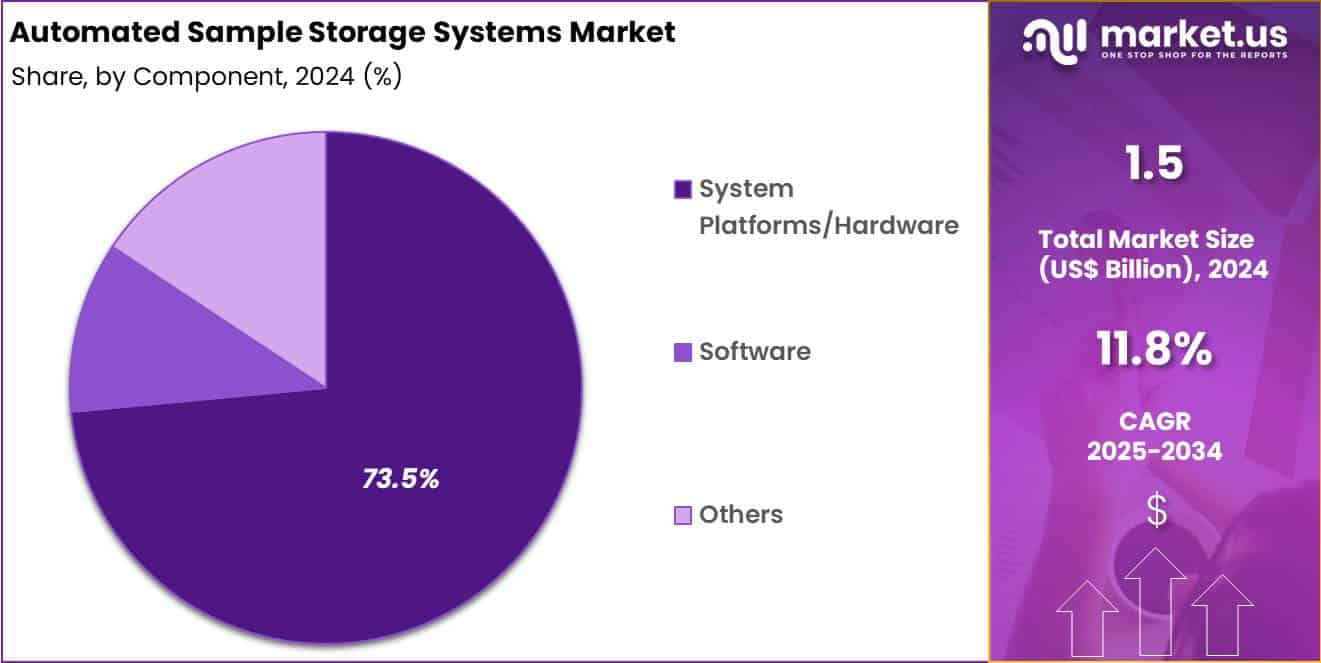
- Considering component, the market is divided into system platforms/hardware, software and others. Among these, system platforms/hardware held a significant share of 73.5%.
- Furthermore, concerning the sample type segment, the market is segregated into chemical & compound samples, biological samples and others. The chemical & compound samples sector stands out as the dominant player, holding the largest revenue share of 65.5% in the market.
- The application segment is segregated into long-term sample preservation, high-throughput sample retrieval and handling, regulatory-compliant sample management and others, with the long-term sample preservation segment leading the market, holding a revenue share of 39.2%.
- The end-user segment is segregated into pharmaceutical and biotechnology companies, academic and clinical research laboratories, biobanks and others, with the pharmaceutical and biotechnology companies segment leading the market, holding a revenue share of 43.0%.
- North America led the market by securing a market share of 38.5% in 2024.
Storage Temperature Analysis
Automated ultra cold storage at −80 °C accounted for 51.1% of the automated sample storage systems market, driven by its critical role in preserving temperature sensitive research materials. Pharmaceutical and biotechnology laboratories rely on −80 °C environments to maintain molecular stability and sample integrity over extended periods.
Increasing biologics research and precision medicine programs expand demand for reliable ultra cold storage. Automation reduces manual freezer access, which lowers contamination risk and temperature fluctuations. Laboratories prioritize ultra cold automation to improve energy efficiency and operational safety. Regulatory expectations for sample integrity strengthen adoption of controlled ultra cold systems.
Growth in global biorepositories further supports long term demand. Technological improvements enhance system uptime and recovery assurance. Integration with digital monitoring platforms improves compliance tracking. This segment is projected to grow steadily due to its essential role in advanced life science research.
Component Analysis
System platforms or hardware represented 73.5% of the automated sample storage systems market, reflecting high capital investment requirements for physical automation infrastructure. End users prioritize robotic handling, storage towers, and retrieval mechanisms to reduce human error. Hardware driven automation improves sample traceability and operational throughput.
Rising laboratory workloads encourage investment in scalable physical systems. Manufacturers focus on modular designs that support expansion without workflow disruption. High durability requirements favor premium hardware procurement. Hardware integration supports seamless software and data management deployment.
Increased focus on laboratory automation strengthens purchasing decisions. Replacement cycles for aging manual storage further expand demand. This segment is anticipated to maintain dominance due to its foundational role in automation deployment.
Sample Type Analysis
Chemical and compound samples accounted for 65.5% of the automated sample storage systems market, supported by extensive use in drug discovery and screening programs. Pharmaceutical companies manage millions of compounds that require precise cataloging and long term stability. Automated storage ensures consistent environmental conditions for compound libraries. High throughput screening workflows depend on rapid and accurate compound retrieval.
Growth in combinatorial chemistry expands storage volumes significantly. Automation improves sample tracking accuracy across discovery pipelines. Chemical samples require less extreme storage compared to biologics, which supports higher volume adoption. Integration with screening robotics improves operational efficiency. Centralized compound management enhances R and D productivity. As a result, this segment is expected to sustain leadership due to scale driven demand in drug development.
Application Analysis
Long term sample preservation represented 39.2% of the automated sample storage systems market, driven by increasing emphasis on research reproducibility and data integrity. Regulatory frameworks require consistent storage conditions for archived samples. Biopharmaceutical development timelines extend sample retention periods significantly. Automation minimizes degradation risks associated with manual handling. Longitudinal studies and clinical research increase preservation needs.
Laboratories adopt automated systems to reduce operational variability. Digital inventory control improves long term tracking accuracy. Preservation focused systems support disaster recovery planning. Growing biobanking initiatives further increase storage duration requirements. This application segment is likely to expand steadily due to compliance and research continuity priorities.
End-User Analysis
Pharmaceutical and biotechnology companies held a 43.0% share of the automated sample storage systems market, reflecting their high volume and high value sample management needs. Drug discovery and development pipelines generate extensive sample inventories. Automation supports faster research cycles and improved laboratory efficiency.
These companies invest heavily in infrastructure to protect intellectual property assets. Regulatory compliance requirements drive adoption of validated storage solutions. Integration with laboratory information systems improves workflow transparency. Expansion of biologics and personalized medicine increases storage complexity.
Capital availability supports large scale automation projects. Strategic focus on digital laboratories accelerates adoption. Consequently, this end user segment is projected to remain dominant due to sustained R and D intensity and compliance driven investment.
Key Market Segments
By Storage Temperature
- Automated Cold Storage (Ambient to -20°C)
- Automated Ultra-Cold Storage (−80 °C ULT)
- Automated Cryogenic Storage (down to −190 °C)
By Component
- System Platforms/Hardware
- Software
- Others
By Sample Type
- Chemical & Compound Samples
- Biological Samples
- Others
By Application
- Long-Term Sample Preservation
- High-Throughput Sample Retrieval and Handling
- Regulatory-Compliant Sample Management
- Others
By End-User
- Pharmaceutical and Biotechnology Companies
- Academic and Clinical Research Laboratories
- Biobanks
- Others
Drivers
Increasing biobanking activities is driving the market
The automated sample storage systems market is driven by the growing number of biobanking activities, which require reliable systems for preserving biological samples at controlled temperatures to support long-term research. Biobanks rely on automated systems to handle large volumes of samples, ensuring integrity for studies in genetics, oncology, and infectious diseases.
Healthcare institutions invest in these systems to facilitate efficient retrieval and minimize manual errors in sample management. Government-funded projects emphasize standardized storage to enable data sharing across international collaborations. Manufacturers develop systems with enhanced capacity to meet the demands of expanding biobank inventories. Clinical researchers utilize automated storage to maintain sample viability for biomarker discovery and personalized medicine initiatives.
Regulatory standards for sample quality further encourage adoption of advanced storage technologies. Academic partnerships advance system features for high-throughput applications in biobanking. The ASPREE Healthy Ageing Biobank collected and stored over 1.4 million aliquots of blood and urine samples at -80°C or under nitrogen vapour for aging biomarker research. This scale demonstrates the essential role of automated systems in supporting large-scale biobanking efforts.
Restraints
High initial installation costs are restraining the market
The automated sample storage systems market is restrained by high initial installation costs, which include expenses for specialized equipment, shielded rooms, and integration with existing laboratory infrastructure. Smaller research facilities often postpone purchases due to limited budgets for capital-intensive technologies.
Manufacturers face challenges in reducing prices while maintaining high-precision components like robotic arms and temperature controls. Regulatory compliance for installation in controlled environments adds to the financial burden. Maintenance contracts for cryogenic systems contribute to ongoing expenses that deter potential buyers. Geographic variations in energy costs influence affordability in different regions. Training programs for staff on system operation represent additional indirect costs.
Vendor financing options remain insufficient to offset upfront investments for many users. Institutional procurement processes prioritize cost-effectiveness, favoring alternative manual storage in resource-constrained settings. These economic factors collectively limit market accessibility and slow adoption among smaller-scale operations.
Opportunities
Growing research in proteomics is creating growth opportunities
The automated sample storage systems market benefits from growing research in proteomics, where large-scale sample repositories are essential for analyzing protein expressions in disease states. Researchers require automated systems to store and retrieve samples efficiently for high-throughput proteomics workflows.
Pharmaceutical companies integrate these systems into proteomics pipelines to accelerate drug discovery. Government initiatives support proteomics projects that demand secure, scalable storage solutions. Developers innovate systems with modular designs to accommodate expanding proteomics datasets. Clinical applications in precision medicine rely on well-preserved samples for proteomic profiling.
Academic consortia utilize automated storage to enable multi-site collaborations in proteomics studies. Regulatory advancements in data standards enhance the value of stored samples for proteomics research. The UK Biobank Pharma Proteomics Project analyzed 5,400 proteins from 600,000 samples to advance understanding of disease mechanisms. This project highlights opportunities for automated systems to facilitate groundbreaking proteomics investigations.
Impact of Macroeconomic / Geopolitical Factors
Rising healthcare expenditures and rapid advancements in biotechnology stimulate robust growth in the automated sample storage systems market, enabling laboratories to invest in sophisticated cryogenic and robotic units that enhance sample integrity and throughput. Key industry players actively pursue expansions in high-capacity models, aligning with the surge in personalized medicine demands from pharmaceutical and research sectors in stable economies.
Elevated inflation rates, nevertheless, amplify expenses for electronic components and energy-efficient designs, leading facilities to scale back on immediate procurements and extend timelines for infrastructure enhancements. Heightened geopolitical rivalries, such as those between the United States and China alongside disruptions in European supply routes, frequently impede the flow of precision engineering parts and software integrations, complicating timelines for system deployments.
Present U.S. tariff structures enforce a baseline reciprocal duty of 10 percent on imported laboratory equipment, supplemented by up to 25% under Section 301 for goods originating from China, thereby raising overall import expenditures and challenging budget allocations for U.S.-based buyers. Such impositions further elicit countermeasures from international partners, which restrict American firms’ access to collaborative ventures and export opportunities in overseas territories.
Despite these hurdles, the tariff regime encourages strategic pivots toward enhanced local assembly operations and partnerships with North American vendors, ultimately reinforcing operational reliability. This shift cultivates a more autonomous and innovative landscape, positioning the market for resilient expansion and superior long-term performance.
Latest Trends
Expansion of longitudinal biobanking collections is a recent trend
In 2024, the automated sample storage systems market has seen a notable trend toward the expansion of longitudinal biobanking collections, enabling repeated sample acquisitions from cohorts over time to track health trajectories. Biobanks incorporate automated systems to manage increasing volumes from follow-up collections, ensuring consistent temperature control. Researchers prioritize systems that support nitrogen vapour storage for long-term sample stability in aging studies.
Clinical teams adopt automated retrieval to facilitate biomarker analysis from sequential samples. Manufacturers enhance system software for tracking sample histories in longitudinal designs. Regulatory guidelines encourage standardized protocols for ongoing collections in biobanks. Academic publications emphasize the value of expanded collections for predictive modeling in chronic diseases.
Collaborative networks share best practices for integrating automation in multi-wave biobanking. Ethical protocols address participant consent for extended sample use in these expansions. The ASPREE Healthy Ageing Biobank initiated a further wave of blood and urine sample collections during 2022–2024 to extend investigations of aging biomarkers.
Regional Analysis
North America is leading the Automated Sample Storage Systems Market
In 2024, North America commanded a 38.5% share of the global automated sample storage systems market, driven by expansive genomic initiatives and the imperative for high-integrity biospecimen preservation in translational research. Pharmaceutical laboratories escalate deployment of robotic retrieval platforms to safeguard compound libraries, ensuring viability for high-throughput drug screening in accelerated discovery pipelines.
Clinical trial sponsors integrate cryogenic automated vaults to maintain chain-of-custody integrity for diverse specimen types, supporting multi-site biomarker studies amid regulatory scrutiny. Biotechnology hubs refine ultra-low temperature architectures with redundant monitoring, mitigating risks of degradation in long-term repositories for rare disease cohorts. Public-private consortia leverage modular storage arrays to harmonize collections across networks, facilitating data-sharing for population health analytics.
Venture infusions propel cryogenic-free alternatives, optimizing energy efficiency in sustainable facility designs. Guideline harmonizations prioritize automated inventory tracking, enhancing auditability in compliance-driven environments. These advancements underscore a robust ecosystem committed to specimen longevity and research reproducibility. The NIH All of Us Research Program had received more than 524,000 biosamples by November 2023.
The Asia Pacific region is expected to experience the highest CAGR during the forecast period
Stakeholders project accelerated adoption of automated sample storage infrastructures in Asia Pacific over the forecast period, as nations fortify biorepository capacities to underpin precision health agendas. Research institutes procure scalable robotic freezers to accommodate burgeoning collections for hereditary disorder investigations in genetically diverse populations. Pharmaceutical manufacturers install high-density racking systems, streamlining workflows for natural product-derived lead compounds in humid subtropical settings.
Governments fund centralized cryogenic facilities, centralizing specimens to expedite infectious disease surveillance and vaccine efficacy trials. Biotech clusters deploy sensor-networked units, enabling predictive maintenance to avert failures in resource-variable locales. Academic alliances standardize liquid nitrogen automation, fostering cross-institutional collaborations for metabolic syndrome explorations.
Export-oriented firms customize compact modules, aligning with stringent international transport regulations for collaborative genomics. These investments harness rapid scientific expansion, securing vital assets for emergent therapeutic breakthroughs. The Westlake BioBank for Chinese pilot project encompassed genomic analyses from 10,376 individuals.
Key Regions and Countries
North America
- US
- Canada
Europe
- Germany
- France
- The UK
- Spain
- Italy
- Russia
- Netherland
- Rest of Europe
Asia Pacific
- China
- Japan
- South Korea
- India
- Australia
- New Zealand
- Singapore
- Thailand
- Vietnam
- Rest of APAC
Latin America
- Brazil
- Mexico
- Rest of Latin America
Middle East & Africa
- South Africa
- Saudi Arabia
- UAE
- Rest of MEA
Key Players Analysis
Key players in the Automated Sample Storage Systems market drive growth by offering high-density, temperature-controlled automation that safeguards biospecimens while accelerating retrieval for biobanks, pharma, and clinical laboratories. Companies in the Automated Sample Storage Systems market invest in robotics, barcode or RFID traceability, and error-reduction software to support large sample volumes with minimal human handling.
Commercial strategies emphasize long-term service agreements, uptime guarantees, and consumables programs that create stable recurring revenue. Technology leaders in the Automated Sample Storage Systems market differentiate through seamless LIMS and data-platform integration that strengthens compliance and audit readiness. Product teams broaden applicability by supporting diverse vial formats and ultra-low-temperature workflows aligned with genomics and cell-therapy pipelines.
Geographic expansion targets regions building centralized biorepository infrastructure and regulated research capacity. Energy efficiency and system scalability increasingly influence procurement decisions in the Automated Sample Storage Systems market. Azenta Life Sciences exemplifies a key player by delivering automated cold-storage platforms and global sample-management services backed by deep expertise in secure, traceable biospecimen handling.
Top Key Players
- Thermo Fisher Scientific, Inc.
- Azenta Life Sciences
- SPT Labtech Ltd
- Hamilton Company
- Danaher
- MICRONIC
- LiCONiC AG
- Haier Biomedical
- ASKION GmbH
- MEGAROBO
- Brooks Automation, Inc.
- Angelantoni Life Science
- Biotron Healthcare
- Celltrio
- HighRes Bio
- MGI Tech
Recent Developments
- In February 2024, Azenta Life Sciences introduced the BioArc Ultra, an automated ultra-low temperature storage system designed for high-density sample preservation at minus 80 degrees Celsius. The system uses a natural air-based cooling approach rather than conventional mechanical refrigeration, resulting in a substantial reduction in energy use and supporting more sustainable laboratory operations.
- In May 2024, Hamilton Storage completed the deployment of four large-scale automated BiOS storage systems at the UK Biobank’s new facility. The installation enables fully automated handling of millions of biological samples, strengthening the infrastructure required for long-term population health studies and large international research collaborations.
Report Scope
Report Features Description Market Value (2024) US$ 1.5 Billion Forecast Revenue (2034) US$ 4.6 Billion CAGR (2025-2034) 11.8% Base Year for Estimation 2024 Historic Period 2020-2023 Forecast Period 2025-2034 Report Coverage Revenue Forecast, Market Dynamics, COVID-19 Impact, Competitive Landscape, Recent Developments Segments Covered By Storage Temperature (Automated Cold Storage (Ambient to -20°C), Automated Ultra-Cold Storage (−80 °C ULT) and Automated Cryogenic Storage (Down to −190 °C)), By Component (System Platforms/Hardware, Software and Others), By Sample Type (Chemical & Compound Samples, Biological Samples and Others), By Application (Long-Term Sample Preservation, High-Throughput Sample Retrieval and Handling, Regulatory-Compliant Sample Management and Others), By End-User (Pharmaceutical and Biotechnology Companies, Academic and Clinical Research Laboratories, Biobanks and Others) Regional Analysis North America – US, Canada; Europe – Germany, France, The UK, Spain, Italy, Russia, Netherlands, Rest of Europe; Asia Pacific – China, Japan, South Korea, India, Australia, New Zealand, Singapore, Thailand, Vietnam, Rest of APAC; Latin America – Brazil, Mexico, Rest of Latin America; Middle East & Africa – South Africa, Saudi Arabia, UAE, Rest of MEA Competitive Landscape Thermo Fisher Scientific, Inc., Azenta Life Sciences, SPT Labtech Ltd, Hamilton Company, Danaher, MICRONIC, LiCONiC AG, Haier Biomedical, ASKION GmbH, MEGAROBO, Brooks Automation, Inc., Angelantoni Life Science, Biotron Healthcare, Celltrio, HighRes Bio, MGI Tech Customization Scope Customization for segments, region/country-level will be provided. Moreover, additional customization can be done based on the requirements. Purchase Options We have three licenses to opt for: Single User License, Multi-User License (Up to 5 Users), Corporate Use License (Unlimited User and Printable PDF)  Automated Sample Storage Systems MarketPublished date: Dec 2025add_shopping_cartBuy Now get_appDownload Sample
Automated Sample Storage Systems MarketPublished date: Dec 2025add_shopping_cartBuy Now get_appDownload Sample -
-
- Thermo Fisher Scientific, Inc.
- Azenta Life Sciences
- SPT Labtech Ltd
- Hamilton Company
- Danaher
- MICRONIC
- LiCONiC AG
- Haier Biomedical
- ASKION GmbH
- MEGAROBO
- Brooks Automation, Inc.
- Angelantoni Life Science
- Biotron Healthcare
- Celltrio
- HighRes Bio
- MGI Tech










