Global AI-Driven Retinal Screening Device Market By Component (Hardware, Software) By Type (Diabetic Retinopathy Screening Devices, Glaucoma Detection Devices, Age-related Macular Degeneration (AMD) Screening Devices, Others) By End-User (Hospitals, Ophthalmic Clinics, Ambulatory Surgical Centers, Diagnostic Centers, Others) , Region and Companies – Industry Segment Outlook, Market Assessment, Competition Scenario, Trends and Forecast 2026-2035
- Published date: May 2026
- Report ID: 185929
- Number of Pages: 282
- Format:
-
keyboard_arrow_up
Quick Navigation
Report Overview
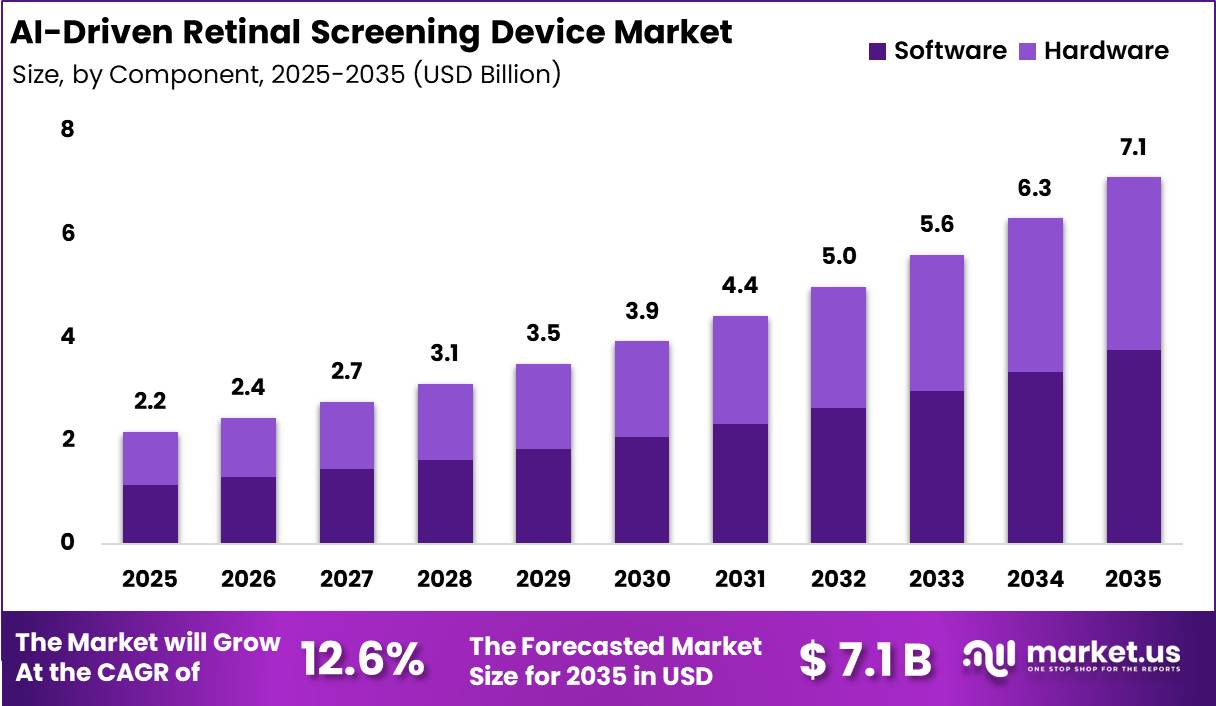
Global AI-Driven Retinal Screening Device Market size is expected to be worth around US$ 7.1 Billion by 2035 from US$ 2.2 Billion in 2025, growing at a CAGR of 12.6% during the forecast period from 2026 to 2035. In 2025, North America led the market, achieving over 41.3% share with a revenue of US$ 0.9 Billion.
The AI-driven retinal screening device market is emerging as a critical segment within the digital healthcare and ophthalmology industry, driven by the increasing prevalence of diabetes and the growing demand for early detection of retinal disorders.
These devices utilize artificial intelligence (AI), deep learning algorithms, and advanced retinal imaging technologies to identify diabetic retinopathy, glaucoma, age-related macular degeneration, and other vision-threatening conditions with high accuracy and speed. The market is gaining strong momentum as healthcare systems increasingly prioritize preventive eye care and scalable screening solutions to reduce the burden of avoidable blindness.
The rising global diabetes population remains one of the primary growth drivers for the market. According to international health estimates, nearly 589 million adults aged 20–79 were living with diabetes in 2024, and the number is projected to reach approximately 853 million by 2050.
Since diabetic retinopathy affects nearly one in three diabetic patients globally, the demand for efficient retinal screening solutions has increased significantly. AI-driven retinal screening devices support healthcare providers by enabling automated image analysis, reducing dependence on specialist ophthalmologists, and improving diagnostic efficiency in both urban and remote healthcare settings.
Technological advancements are further accelerating market expansion. Improvements in AI-based image interpretation, cloud-connected diagnostic platforms, edge-computing retinal cameras, and portable point-of-care retinal devices are enhancing accessibility and diagnostic precision.
In addition, teleophthalmology integration is enabling real-time remote screening and consultation, particularly in underserved and rural regions where access to eye specialists remains limited. Healthcare institutions and public health agencies are also increasingly adopting digital eye-screening initiatives to improve population-level eye disease management.
The market is also benefiting from increasing investments in healthcare digitalization and rising awareness regarding early retinal disease detection. As AI algorithms continue to improve diagnostic sensitivity and workflow efficiency, AI-driven retinal screening devices are expected to become an integral component of future ophthalmic and diabetes care pathways worldwide.
Key Takeaways
- Market Size: Global AI-Driven Retinal Screening Device Market size is expected to be worth around US$ 7.1 Billion by 2035 from US$ 2.2 Billion in 2025.
- Market Share: The market growing at a CAGR of 12.6% during the forecast period from 2026 to 2035.
- Component Analysis: The software segment dominated the market and accounted for 52.7% of the global market share in 2025.
- Type Analysis: Diabetic retinopathy screening devices held the largest market share of 45.8% in 2025.
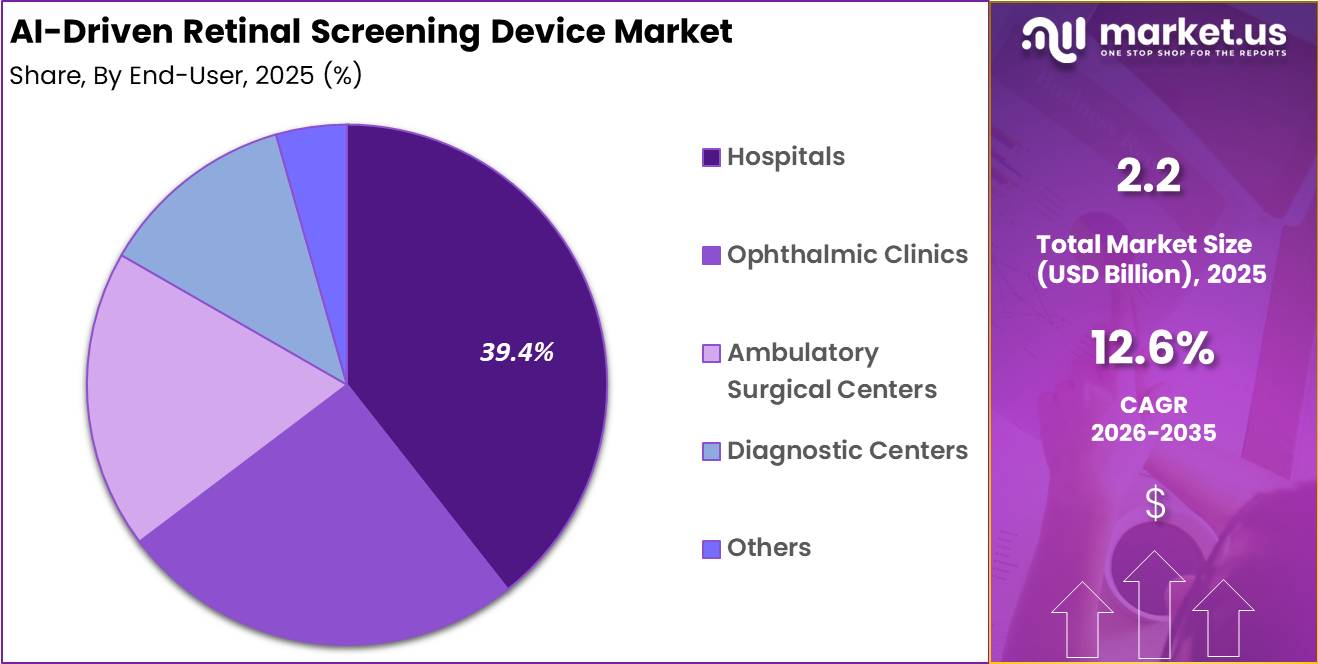
- End-User Analysis: Hospitals dominated the market with a 39.4% share in 2025.
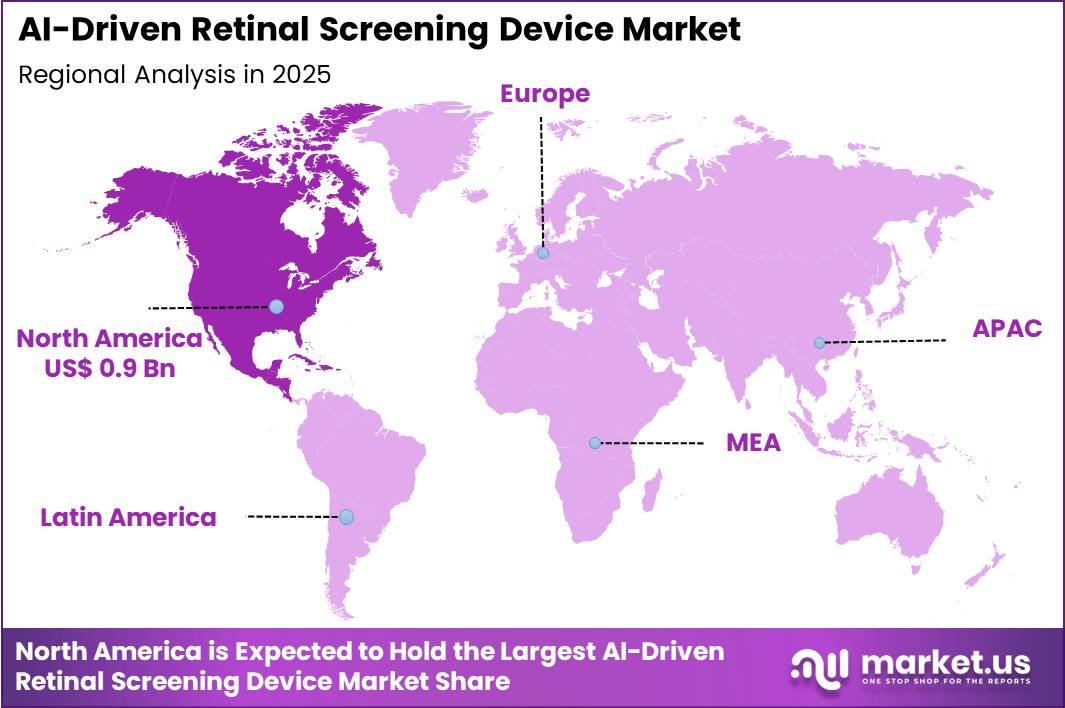
- Regional Analysis: In 2025, North America led the market, achieving over 41.3% share with a revenue of US$ 0.9 Billion.
Component Analysis
The component segment of the AI-Driven Retinal Screening Device Market is divided into software and hardware. The software segment dominated the market and accounted for 52.7% of the global market share in 2025. The dominance of this segment is attributed to the increasing integration of artificial intelligence algorithms for automated retinal image interpretation, disease identification, and clinical workflow optimization.
AI-based software solutions enable rapid screening, improved diagnostic consistency, and reduced dependency on specialist ophthalmologists. Growing adoption of cloud-based analytics platforms and teleophthalmology solutions is further supporting market expansion. In addition, continuous advancements in deep learning technologies and increasing regulatory approvals for AI-assisted diagnostic systems are accelerating software deployment across healthcare facilities.
The hardware segment is also experiencing notable growth due to rising demand for advanced retinal imaging equipment, including fundus cameras, optical coherence tomography systems, and portable retinal scanners. Healthcare providers are increasingly investing in compact and high-resolution imaging devices to improve early-stage retinal disease detection. Furthermore, technological advancements focused on AI-enabled imaging integration and portable screening capabilities are expected to support the long-term growth of the hardware segment.
Type Analysis
Based on type, the AI-Driven Retinal Screening Device Market is segmented into diabetic retinopathy screening devices, services, and instruments. Among these, diabetic retinopathy screening devices held the largest market share of 45.8% in 2025. The segment growth is primarily driven by the increasing global prevalence of diabetes and the growing need for early detection of diabetes-related retinal disorders.
AI-enabled diabetic retinopathy screening systems provide rapid, accurate, and automated diagnosis, improving screening efficiency in both urban and rural healthcare settings. The rising implementation of preventive eye care programs and expanding adoption of telemedicine-based retinal screening are also contributing to segment growth.
The services segment is witnessing steady expansion owing to increasing demand for system installation, maintenance, software upgrades, training, and technical support associated with AI-driven retinal screening solutions. Healthcare facilities are increasingly relying on service providers to ensure efficient integration and operation of AI-enabled diagnostic systems.
Meanwhile, the instruments segment is gaining traction due to ongoing advancements in retinal imaging technologies, including high-resolution fundus cameras and optical coherence tomography devices. The growing focus on precision diagnostics and enhanced imaging quality is expected to support segment development during the forecast period.
End-User Analysis
Based on end user, the AI-Driven Retinal Screening Device Market is segmented into hospitals, ophthalmic clinics, ambulatory surgical centers, diagnostic centers, and others. Hospitals dominated the market with a 39.4% share in 2025, supported by the increasing adoption of AI-powered retinal screening technologies across large healthcare institutions.
Hospitals benefit from advanced healthcare infrastructure, availability of skilled professionals, and high patient volumes, making them key adopters of automated retinal diagnostic systems. Rising cases of diabetic retinopathy, glaucoma, and age-related macular degeneration are further increasing the demand for efficient screening solutions within hospital settings. In addition, integration of AI screening platforms with electronic health record systems is improving workflow efficiency and patient management.
Ophthalmic clinics represent another significant segment due to the growing adoption of specialized AI-assisted retinal diagnostic tools for eye care services. Ambulatory surgical centers are gradually implementing retinal screening systems to support ophthalmic surgical evaluations and postoperative monitoring.
Diagnostic centers are also witnessing increased demand as preventive healthcare screening programs continue to expand. The others segment, including community healthcare centers and mobile screening units, is expected to grow steadily owing to increasing efforts to improve eye care accessibility in underserved and remote regions.
Key Market Segments
By Component
- Hardware
- Software
By Type
- Diabetic Retinopathy Screening Devices
- Glaucoma Detection Devices
- Age-related Macular Degeneration (AMD) Screening Devices
- Others
By End-User
- Hospitals
- Ophthalmic Clinics
- Ambulatory Surgical Centers
- Diagnostic Centers
- Others
Driving Factors
The increasing global burden of diabetes is a major driver for the AI-driven retinal screening device market. Diabetic retinopathy (DR) is one of the leading causes of blindness among working-age adults, creating strong demand for early and scalable screening technologies.
According to the U.S. Centers for Disease Control and Prevention (CDC), approximately 9.6 million people in the United States were living with diabetic retinopathy in 2021, while nearly 1.84 million individuals had vision-threatening DR. In addition, around 26.4% of diabetic patients were affected by some form of retinopathy.
Healthcare systems are increasingly adopting AI-enabled retinal imaging solutions because conventional eye screening requires trained ophthalmologists, who remain limited in many rural and underserved regions. AI devices can automatically analyze retinal images within minutes and improve screening coverage.
Government agencies and healthcare institutions are also supporting digital ophthalmology initiatives to reduce preventable blindness. Furthermore, rising diabetes prevalence among aging populations and younger adults is accelerating demand for automated retinal diagnostics. These factors collectively strengthen market expansion opportunities for AI-driven retinal screening technologies worldwide.
Trending Factors
One of the key trends in the AI-driven retinal screening device market is the growing integration of autonomous AI systems into primary healthcare and community screening programs. Hospitals and healthcare providers are increasingly deploying AI-enabled retinal imaging devices that can independently detect diabetic retinopathy without direct ophthalmologist intervention.
This trend is helping healthcare systems improve patient throughput and reduce specialist workload. Research studies have demonstrated that AI-assisted retinal screening can significantly improve operational efficiency while maintaining high diagnostic accuracy.
Another emerging trend is the adoption of cloud-based AI platforms and portable retinal cameras in rural healthcare settings. In India, multicentric validation studies of AI-driven diabetic retinopathy screening systems reported sensitivity rates of approximately 92% and specificity rates near 88% for disease detection.
Healthcare organizations are also focusing on explainable and ethical AI models to improve physician trust and regulatory compliance. Furthermore, integration of multimodal AI and large language models into ophthalmology workflows is expanding clinical decision support capabilities. These technological advancements are improving accessibility, affordability, and scalability of retinal disease screening across global healthcare systems.
Restraining Factors
Despite significant technological progress, the AI-driven retinal screening device market faces several restraints associated with regulatory, infrastructure, and clinical adoption challenges. One major limitation is the requirement for high-quality retinal images for accurate AI interpretation. Poor image quality caused by inadequate equipment handling, cataracts, or insufficient patient cooperation can reduce diagnostic reliability. This creates operational difficulties, particularly in low-resource healthcare facilities.
Another important restraint is the limited availability of standardized reimbursement policies for AI-based retinal screening procedures in several countries. Many healthcare providers remain hesitant to invest in advanced AI systems without clear financial incentives or long-term reimbursement frameworks. In addition, concerns related to data privacy, cybersecurity, and algorithmic bias continue to affect adoption rates.
Clinical validation requirements also remain stringent because retinal screening directly influences blindness prevention decisions. Regulatory authorities require extensive multicenter testing before approval, increasing commercialization timelines and development costs.
Furthermore, healthcare professionals may resist autonomous AI systems due to concerns regarding diagnostic accountability and workflow disruption. These barriers can slow large-scale implementation, especially in developing healthcare systems with limited digital infrastructure and trained personnel.
Opportunity
The expanding unmet need for early diabetic retinopathy diagnosis in underserved and rural populations presents a major opportunity for the AI-driven retinal screening device market. According to CDC estimates, diabetic retinopathy prevalence continues to increase alongside rising diabetes incidence globally. Early diagnosis can reduce severe vision loss risk by up to 95% through timely treatment and monitoring.
Many countries face shortages of ophthalmologists and retina specialists, particularly in remote regions. AI-powered retinal screening devices offer an opportunity to decentralize eye care services through primary clinics, pharmacies, and telemedicine networks. Portable retinal cameras integrated with cloud-based AI platforms can enable large-scale screening programs at lower operational costs.
Government healthcare initiatives focused on digital health transformation and blindness prevention are expected to create favorable growth conditions. In India and several emerging economies, AI-based retinal screening systems are being evaluated for national diabetes screening programs because they can improve accessibility in resource-constrained settings.
In addition, technological advancements in multimodal AI, smartphone-based retinal imaging, and explainable AI models are expected to create new commercial opportunities for device manufacturers, healthcare providers, and digital health companies over the coming years.
Regional Analysis
In 2025, North America dominated the AI-Driven Retinal Screening Device Market, accounting for over 41.3% of the global market share and generating approximately US$ 0.9 billion in revenue. The regional market growth is primarily supported by the strong presence of advanced healthcare infrastructure, widespread adoption of artificial intelligence technologies, and increasing prevalence of diabetic retinopathy and other retinal disorders across the United States and Canada.
The growing aging population and rising awareness regarding early detection of eye diseases have further accelerated the demand for AI-powered retinal screening solutions in the region. Additionally, favorable reimbursement policies, high healthcare spending, and increasing investments in digital health technologies have encouraged healthcare providers to integrate automated retinal screening systems into clinical workflows.
The presence of leading market players and continuous technological advancements in AI-based diagnostic imaging are also contributing to market expansion. Moreover, government initiatives promoting preventive healthcare and teleophthalmology services are enhancing accessibility to retinal screening, particularly in remote and underserved areas.
Academic collaborations and research activities focused on improving diagnostic accuracy and workflow efficiency are expected to strengthen the regional market outlook. As a result, North America is anticipated to maintain its leading position throughout the forecast period, supported by ongoing innovation and increasing adoption of AI-enabled ophthalmic diagnostic devices.
Key Regions and Countries
North America
- The US
- Canada
Europe
- Germany
- France
- The U.K.
- Italy
- Spain
- Russia & CIS
- Rest of Europe
Asia Pacific
- China
- India
- Japan
- South Korea
- ASEAN
- Australia & New Zealand
- Rest of Asia Pacific
Middle East & Africa
- GCC
- South Africa
- Rest of Middle East & Africa
Latin America
- Brazil
- Mexico
- Rest of Latin America
Key Players Analysis
The AI-Driven Retinal Screening Device Market is characterized by the presence of several established medical imaging companies and emerging artificial intelligence-focused healthcare firms competing through technological innovation, regulatory approvals, and strategic collaborations. Key players such as Topcon Corporation, Canon Medical Systems Corporation, NIDEK Co., Ltd., and Optomed Plc are strengthening their market position by integrating AI algorithms into retinal imaging systems for the early detection of diabetic retinopathy, glaucoma, and age-related macular degeneration.
AI-specialized companies including Digital Diagnostics Inc. and Eyenuk Inc. are gaining significant traction due to their FDA-cleared autonomous screening platforms, which improve diagnostic efficiency and reduce dependency on ophthalmologists. Strategic partnerships between device manufacturers, hospitals, and telehealth providers are further accelerating market penetration, particularly in underserved and remote regions.
Companies are increasingly focusing on portable and cloud-connected retinal screening devices to expand point-of-care diagnostics. In addition, investments in deep learning algorithms, real-time image analysis, and workflow automation are enhancing screening accuracy and operational efficiency. Competitive intensity is expected to increase as players prioritize product innovation, geographic expansion, and regulatory compliance to strengthen their global market presence.
Market Key Players
- Eyenuk, Inc.
- Digital Diagnostics, Inc.
- Retina-AI Health, Inc.
- AEYE Health, Inc.
- Optomed plc
- Verily Life Sciences (a Google company)
- Forus Health Pvt. Ltd.
- Medios (RetinaLyze System)
- iHealthScreen Inc.
- AiDx Medical Inc.
- DeepMind (Google-owned)
- Visulytix Ltd.
- Guangxi Realway Medical
- Novasight
- PlenOptika, Inc.
- Others
Recent Developments
- Digital Diagnostics, Inc.(January 2025) – Digital Diagnostics deepened its collaboration with primary care and retail health chains in the U.S. to scale its LumineticsCore autonomous AI platform, focusing on integrating the system into everyday diabetes workflows so that eye screening becomes a routine, point-of-care service rather than a separate specialist visit. This push positioned the company as a reference partner for health systems that want to operationalize guideline-level diabetic retinopathy screening without adding new ophthalmology capacity.
- Eyenuk, Inc.(March 2025)- Eyenuk secured about 25 million dollars in funding, bringing its total capital raised to roughly 43 million dollars, with the round led by AXA IM Alts and joined by several existing healthcare investors. The company signaled that the new capital would be used to extend its EyeArt autonomous AI platform to additional retinal and systemic diseases and accelerate commercialization across new geographies.
- Optomed plc(October 2025)- Optomed promoted its Aurora IQ handheld fundus camera with embedded AI analysis, positioning the device as a mobile solution that can capture wide‑field, non‑mydriatic retinal images and run AI‑based grading at the point of care. This product push strengthened Optomed’s role as the hardware backbone for partners’ algorithms, including collaborations with several AI vendors to deliver turnkey retinal screening kits.
Report Scope
Report Features Description Market Value (2025) US$ 2.2 Billion Forecast Revenue (2035) US$ 7.1 Billion CAGR (2026-2035) 12.6% Base Year for Estimation 2025 Historic Period 2020-2024 Forecast Period 2026-2035 Report Coverage Revenue Forecast, Market Dynamics, Competitive Landscape, Recent Developments Segments Covered By Component (Hardware, Software) By Type (Diabetic Retinopathy Screening Devices, Glaucoma Detection Devices, Age-related Macular Degeneration (AMD) Screening Devices, Others) By End-User (Hospitals, Ophthalmic Clinics, Ambulatory Surgical Centers, Diagnostic Centers, Others) Regional Analysis North America – The US, Canada; Europe – Germany, France, U.K., Italy, Spain, Russia & CIS, Rest of Europe; Asia Pacific – China, India, Japan, South Korea, ASEAN, Australia & New Zealand, Rest of Asia Pacific; Middle East & Africa – GCC, South Africa, Rest of Middle East & Africa; Latin America – Brazil, Mexico, Rest of Latin America Competitive Landscape Eyenuk, Inc., Digital Diagnostics, Inc., Retina-AI Health, Inc., AEYE Health, Inc., Optomed plc, Verily Life Sciences (a Google company), Forus Health Pvt. Ltd., Medios (RetinaLyze System), iHealthScreen Inc., AiDx Medical Inc., DeepMind (Google-owned), Visulytix Ltd., Guangxi Realway Medical, Novasight, PlenOptika, Inc., Others, Customization Scope Customization for segments, region/country-level will be provided. Moreover, additional customization can be done based on the requirements. Purchase Options We have three licenses to opt for: Single User License, Multi-User License (Up to 5 Users), Corporate Use License (Unlimited User and Printable PDF)  AI-Driven Retinal Screening Device MarketPublished date: May 2026add_shopping_cartBuy Now get_appDownload Sample
AI-Driven Retinal Screening Device MarketPublished date: May 2026add_shopping_cartBuy Now get_appDownload Sample -
-
- Eyenuk, Inc.
- Digital Diagnostics, Inc.
- Retina-AI Health, Inc.
- AEYE Health, Inc.
- Optomed plc
- Verily Life Sciences (a Google company)
- Forus Health Pvt. Ltd.
- Medios (RetinaLyze System)
- iHealthScreen Inc.
- AiDx Medical Inc.
- DeepMind (Google-owned)
- Visulytix Ltd.
- Guangxi Realway Medical
- Novasight
- PlenOptika, Inc.
- Others










