Global Dietary Supplements Testing Market Size, Share Analysis Report By Test Type (Identity/Authentication, Potency, Contaminants, Microbiological, Adulteration, Stability And Shelf Life, Allergen And GMO, Label Claim Verification, Functional Claims, Others), By Product Tested (Botanicals, Phytochemicals, Vitamins And Minerals, Amino Acids And Proteins, Specialty Supplements, Others), By Region and Companies - Industry Segment Outlook, Market Assessment, Competition Scenario, Trends and Forecast 2026-2035
- Published date: Mar 2026
- Report ID: 182470
- Number of Pages: 313
- Format:
-
keyboard_arrow_up
Quick Navigation
Report Overview
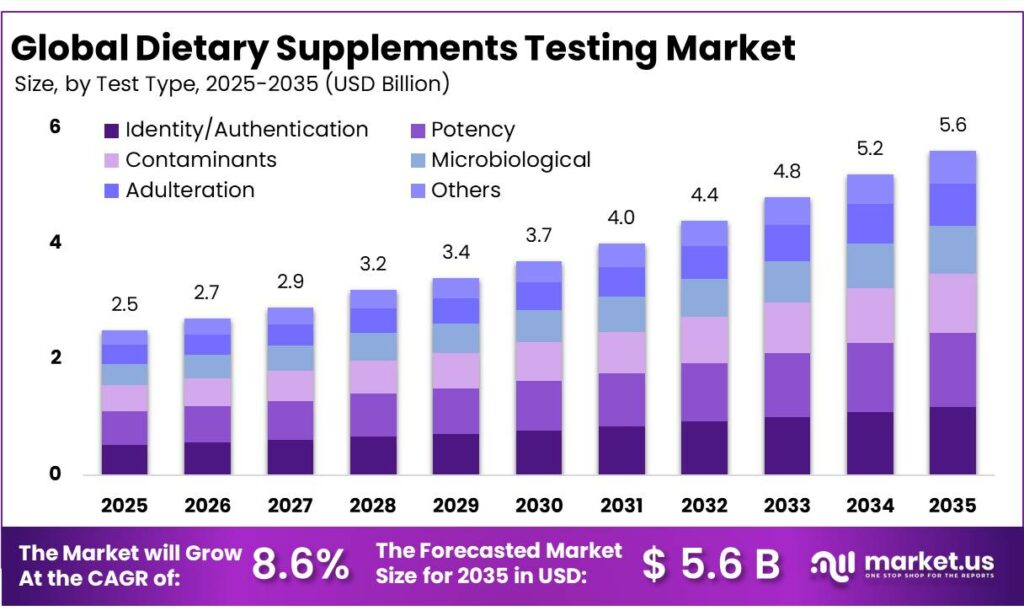
The Global Multivitamin Gummies Market size is expected to be worth around USD 5.6 Billion by 2035, from USD 2.5 Billion in 2025, growing at a CAGR of 8.6% during the forecast period from 2026 to 2035. In 2025, North America held a dominant market position, capturing more than a 41.3% share, holding USD 1.0 Billion revenue.
The multivitamin gummies segment sits at the intersection of dietary supplementation, functional foods, and consumer-friendly delivery systems. From an industry standpoint, gummies have moved beyond a children’s format into a mainstream adult dosage form because they combine convenience, taste, and compliance.
NIH notes that multivitamin/mineral supplements are already used by about one-third of U.S. adults and by one-quarter of children and adolescents, while multivitamin/mineral products represented 14% of all supplement purchases and 38% of vitamin-and-mineral supplement sales in the United States in 2019. NIH also reports that U.S. dietary supplement sales reached $55.7 billion in 2020, including $21.2 billion for vitamin/mineral-containing supplements and $8.0 billion for multivitamin/multimineral products, which indicates a large and established base from which gummy formats continue to gain share.
The industrial scenario remains favorable because gummies solve a practical adherence problem while fitting premium positioning trends in wellness. Mérieux NutriSciences observed that gummies, designated by USP as chewable gels, have become an “extremely popular” supplement dosage form and that under 10% of all new product launches in the prior year involved gummy delivery systems, showing meaningful innovation activity without suggesting category saturation.
At the same time, the wider supplement environment remains highly active: the Council for Responsible Nutrition stated in October 2024 that 75% of Americans use dietary supplements, 42% take them for overall health and wellness, 30% for immune health, 27% for energy, and 69% value personalized regimens. Median monthly spending among supplement users also increased from $48 in 2023 to $50 in 2024, indicating durable willingness to pay.
Demand is also being supported by a clear nutritional need base. WHO states that more than 2 billion people worldwide are deficient in key vitamins and minerals, while 42% of children under 5 and 40% of pregnant women globally are affected by anaemia. This backdrop supports continued interest in convenient preventive nutrition formats, especially where consumers seek easy daily routines rather than conventional tablets.
Government and public-health initiatives are also reinforcing the category’s long-term structure. WHO’s 24 October 2025 guideline on fortifying edible oils and fats with vitamins A and D and the World Health Assembly resolution on safe and effective food fortification both signal sustained policy support for micronutrient improvement. On the regulatory side, FDA’s dietary supplement compliance program dated 18 August 2025 reaffirmed inspections under 21 CFR Part 111 and sample-based enforcement to ensure acceptable quality and reduce exposure to adulterated or misbranded products.
Key Takeaways
- Multivitamin Gummies Market size is expected to be worth around USD 5.6 Billion by 2035, from USD 2.5 Billion in 2025, growing at a CAGR of 8.6%.
- Potency held a dominant market position, capturing more than a 23.6% share.
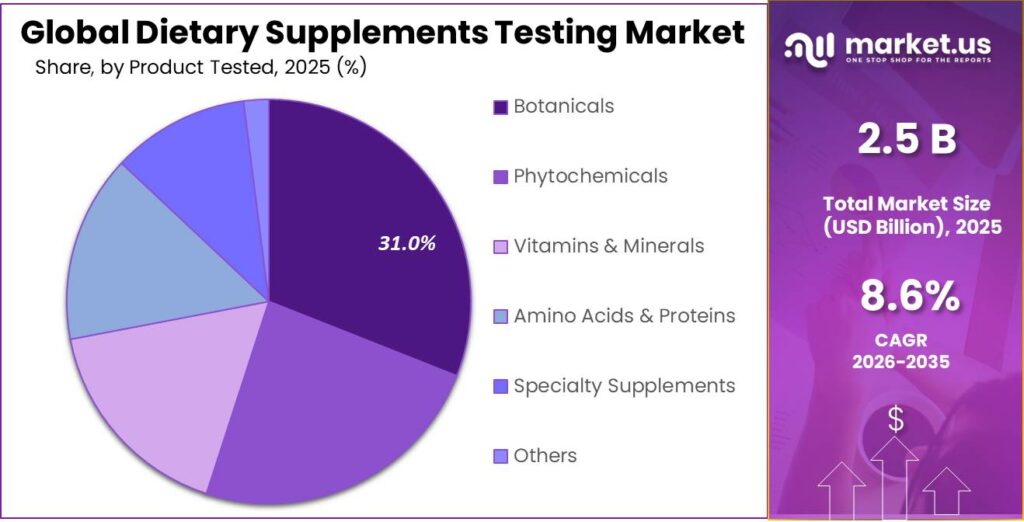
- Botanicals held a dominant market position, capturing more than a 31.5% share.
- North America emerged as the dominating region, accounting for 41.3% share and approximately USD 1.0 billion.
By Test Type Analysis
Potency testing leads with 23.6% share driven by strict label accuracy needs
In 2025, Potency held a dominant market position, capturing more than a 23.6% share. This leadership reflects the core role potency testing plays in verifying that dietary supplements contain the exact amount of active ingredients stated on the label. Regulatory frameworks such as those enforced by the U.S. Food and Drug Administration under Good Manufacturing Practices require manufacturers to ensure identity, purity, strength, and composition of their products, making potency testing a non-negotiable step in production.
By Product Tested Analysis
Botanicals lead with 31.5% share as herbal demand stays strong
In 2025, Botanicals held a dominant market position, capturing more than a 31.5% share. This strong position comes from the growing consumer shift toward plant-based and traditional health solutions. Herbal supplements such as turmeric, ashwagandha, ginseng, and green tea extracts are widely used for immunity, stress relief, and overall wellness. Because these products come from natural sources, their composition can vary depending on origin, harvesting, and processing. This makes testing very important to confirm identity, strength, and absence of contaminants.
Key Market Segments
By Test Type
- Identity/Authentication
- Potency
- Contaminants
- Microbiological
- Adulteration
- Stability & Shelf Life
- Allergen & GMO
- Label Claim Verification
- Functional Claims
- Others
By Product Tested
- Botanicals
- Phytochemicals
- Vitamins & Minerals
- Amino Acids & Proteins
- Specialty Supplements
- Others
Emerging Trends
Rising focus on transparency and mislabeling is shaping advanced testing trends
One of the most noticeable trends in dietary supplements testing is the growing focus on transparency and the detection of mislabeling. Consumers today are more aware and cautious about what they consume, especially after multiple reports highlighting quality concerns. Recent findings suggest that nearly 45% of supplements face mislabeling risks, which clearly shows the gap between product claims and actual content.
Governments and public health agencies are also encouraging better labeling practices and stricter quality checks. This shift is leading to increased demand for third-party testing and certification. Consumers now prefer products that are tested and verified by independent labs, which is pushing companies to invest more in quality assurance. Over time, transparency is not just becoming a regulatory requirement but also a competitive advantage for supplement brands.
Shift toward personalized nutrition is increasing demand for advanced testing
Another key trend is the rise of personalized nutrition, which is changing how supplements are developed and tested. Instead of general multivitamins, consumers are now looking for products tailored to their specific health needs. This shift is supported by advancements in biomarker and diagnostic testing. In 2025, personalized supplement plans based on health data and nutrient deficiencies are becoming more common, increasing the need for precise and reliable testing methods.
At the same time, global health concerns are driving this trend further. The World Health Organization reports that more than 2 billion people worldwide suffer from micronutrient deficiencies, which is pushing demand for targeted supplementation.
Drivers
Rising consumer usage is pushing stronger quality testing across supplements
One of the biggest driving factors for dietary supplements testing is the steady rise in consumer usage and awareness. As more people include vitamins, minerals, and herbal products in their daily routine, the need to ensure safety and correct dosage becomes very important. According to the Council for Responsible Nutrition, about 75% of U.S. adults used dietary supplements in 2024, showing how common these products have become in everyday health habits. At the same time, the National Institutes of Health reported that dietary supplement sales in the U.S. reached $55.7 billion in 2020, which reflects a large and growing consumer base.
Government regulations and safety programs are strengthening testing demand
Another strong driver is the growing role of government regulations and safety initiatives. Authorities across countries are tightening rules to make sure supplements meet proper quality standards. The U.S. Food and Drug Administration enforces Good Manufacturing Practices under 21 CFR Part 111, which requires companies to test supplements for identity, purity, strength, and composition. This makes testing a compulsory step in production rather than a choice.
On a global level, the World Health Organization has highlighted that more than 2 billion people worldwide suffer from micronutrient deficiencies, increasing the need for safe and effective supplementation.
Restraints
High cost and complex testing processes limit wider adoption
One of the key restraining factors in the dietary supplements testing market is the high cost and technical complexity involved in testing procedures. Testing supplements is not as simple as basic quality checks, especially for products containing multiple active ingredients or botanical extracts. Advanced methods like chromatography, mass spectrometry, and DNA-based testing require skilled professionals and expensive equipment.
According to the U.S. Food and Drug Administration, manufacturers must comply with strict Good Manufacturing Practices under 21 CFR Part 111, which include detailed testing for identity, purity, strength, and composition. These requirements increase operational costs, especially for small and mid-sized companies. The National Institutes of Health reported that the dietary supplement industry reached $55.7 billion in sales in 2020, showing strong demand, but not all companies operate at the same financial scale.
Variability in raw materials makes standard testing difficult
Another major challenge is the natural variability of raw materials, especially in herbal and botanical supplements. Unlike synthetic products, plant-based ingredients can differ in composition based on geography, climate, soil, and harvesting methods. This makes it harder to maintain consistent quality and requires more detailed and repeated testing.
The World Health Organization has highlighted that herbal medicines are widely used globally, but their quality can vary significantly if not properly controlled. Additionally, WHO states that more than 2 billion people globally face micronutrient deficiencies, increasing dependence on supplements while also raising the need for accurate and consistent formulations.
Opportunity
Expanding global demand for supplements is opening new testing opportunities
One major growth opportunity in the dietary supplements testing market comes from the rapid expansion of supplement consumption worldwide. As more people focus on preventive healthcare, the demand for supplements continues to rise, which directly increases the need for proper testing. Studies show that the global dietary supplements market was valued at around USD 152 billion in 2021 and is expected to grow significantly in the coming years, reflecting strong consumption trends.
At the same time, health concerns are pushing this demand even further. According to the World Health Organization, more than 2 billion people worldwide suffer from micronutrient deficiencies, which highlights the need for reliable supplementation.
Innovation in formulations and personalized nutrition is boosting testing demand
Another key opportunity lies in the shift toward innovative and personalized supplement products. Consumers today are not just buying basic vitamins; they are choosing customized blends, plant-based products, and targeted solutions for immunity, gut health, and energy. This trend is increasing the complexity of supplement formulations, making testing more important than ever. Data shows that the dietary supplements market is expected to reach around USD 209.52 billion in 2025, showing strong year-on-year growth driven by innovation and lifestyle changes.
Regional Insights
North America dominates with 41.3% share driven by strong supplement consumption and strict regulatory framework
In the Dietary Supplements Testing Market, North America emerged as the dominating region, accounting for 41.3% share and approximately USD 1.0 billion in value. The United States plays a central role, where the U.S. Food and Drug Administration oversees product safety and compliance under the Dietary Supplement Health and Education Act. The FDA estimates that the U.S. dietary supplement market exceeds $60 billion and includes over 100,000 products, highlighting the scale and complexity of the industry.
Consumer usage is also significantly high in the region. Data from the Centers for Disease Control and Prevention shows that 57.6% of U.S. adults used dietary supplements in recent years, reflecting strong adoption across different age groups. This widespread usage increases the need for accurate testing related to potency, purity, and contamination.

Key Regions and Countries Insights
- North America
- US
- Canada
- Europe
- Germany
- France
- The UK
- Spain
- Italy
- Rest of Europe
- Asia Pacific
- China
- Japan
- South Korea
- India
- Australia
- Rest of APAC
- Latin America
- Brazil
- Mexico
- Rest of Latin America
- Middle East & Africa
- GCC
- South Africa
- Rest of MEA
Key Players Analysis
Eurofins Scientific is one of the leading players in dietary supplements testing, supported by its strong global laboratory network and wide analytical capabilities. In 2025, the company reported revenues of around €7,296 million, reflecting a 5% year-on-year growth driven by organic expansion and acquisitions. The company operates thousands of laboratories worldwide, focusing on food, pharmaceutical, and supplement testing.
Bureau Veritas is a well-established global TIC (Testing, Inspection, and Certification) company with a strong presence in food and supplement testing. In 2024, the company reported revenues of approximately USD 6.34 billion and operated with 79,000 employees across 1,500+ offices and laboratories in 140 countries. Its scale allows it to support large supplement manufacturers with quality assurance, safety testing, and certification services. The company’s global reach and regulatory expertise make it a key player in ensuring product compliance and safety in the dietary supplements industry.
Top Key Players Outlook
- Eurofins Scientific
- Mrieux Nutri Sciences
- Intertek Group PLC
- Bureau Veritas
- SGS SA
- ALSLimited
- TVSD
- Neogen Corporation
- BSCG Banned Substances Control GROUP
- Auriga Research dietarysupplementtestinglab
Recent Industry Developments
Eurofins Scientific has built a strong position in the dietary supplements testing sector through its large global lab network and advanced analytical capabilities. As of 2025, the company reported total revenues of €7,296 million, growing from €6,951 million in 2024, showing steady expansion supported by food and supplement testing demand.
Mérieux NutriSciences has built a solid position in the dietary supplements testing sector by focusing on food safety, quality, and scientific validation services. As of 2025, the company operates a network of over 140 laboratories and employs more than 10,000 professionals globally, allowing it to support supplement manufacturers with testing for contaminants, microbiology, and nutrient verification across multiple markets.
Report Scope
Report Features Description Market Value (2025) USD 2.5 Bn Forecast Revenue (2035) USD 5.6 Bn CAGR (2026-2035) 8.6% Base Year for Estimation 2025 Historic Period 2020-2024 Forecast Period 2026-2035 Report Coverage Revenue Forecast, Market Dynamics, Competitive Landscape, Recent Developments Segments Covered By Test Type (Identity/Authentication, Potency, Contaminants, Microbiological, Adulteration, Stability And Shelf Life, Allergen And GMO, Label Claim Verification, Functional Claims, Others), By Product Tested (Botanicals, Phytochemicals, Vitamins And Minerals, Amino Acids And Proteins, Specialty Supplements, Others) Regional Analysis North America – US, Canada; Europe – Germany, France, The UK, Spain, Italy, Rest of Europe; Asia Pacific – China, Japan, South Korea, India, Australia, Singapore, Rest of APAC; Latin America – Brazil, Mexico, Rest of Latin America; Middle East & Africa – GCC, South Africa, Rest of MEA Competitive Landscape Eurofins Scientific, Mrieux Nutri Sciences, Intertek Group PLC, Bureau Veritas, SGS SA, ALSLimited, TVSD, Neogen Corporation, BSCG Banned Substances Control GROUP, Auriga Research dietarysupplementtestinglab Customization Scope Customization for segments, region/country-level will be provided. Moreover, additional customization can be done based on the requirements. Purchase Options We have three licenses to opt for: Single User License, Multi-User License (Up to 5 Users), Corporate Use License (Unlimited User and Printable PDF)  Dietary Supplements Testing MarketPublished date: Mar 2026add_shopping_cartBuy Now get_appDownload Sample
Dietary Supplements Testing MarketPublished date: Mar 2026add_shopping_cartBuy Now get_appDownload Sample -
-
- Eurofins Scientific
- Mrieux Nutri Sciences
- Intertek Group PLC
- Bureau Veritas
- SGS SA
- ALSLimited
- TVSD
- Neogen Corporation
- BSCG Banned Substances Control GROUP
- Auriga Research dietarysupplementtestinglab










